CHO cells, also known as Chinese hamster ovary cells, are a widely used cell line in the production of biopharmaceuticals. They were originally derived from the ovaries of Chinese hamsters, hence the name. Over the decades, CHO cells have been cultivated and manipulated for molecular biology and drug biotechnology, gaining significant popularity in recombinant antibody technology.


(Microscopic image of CHO transfected fluorescent protein)
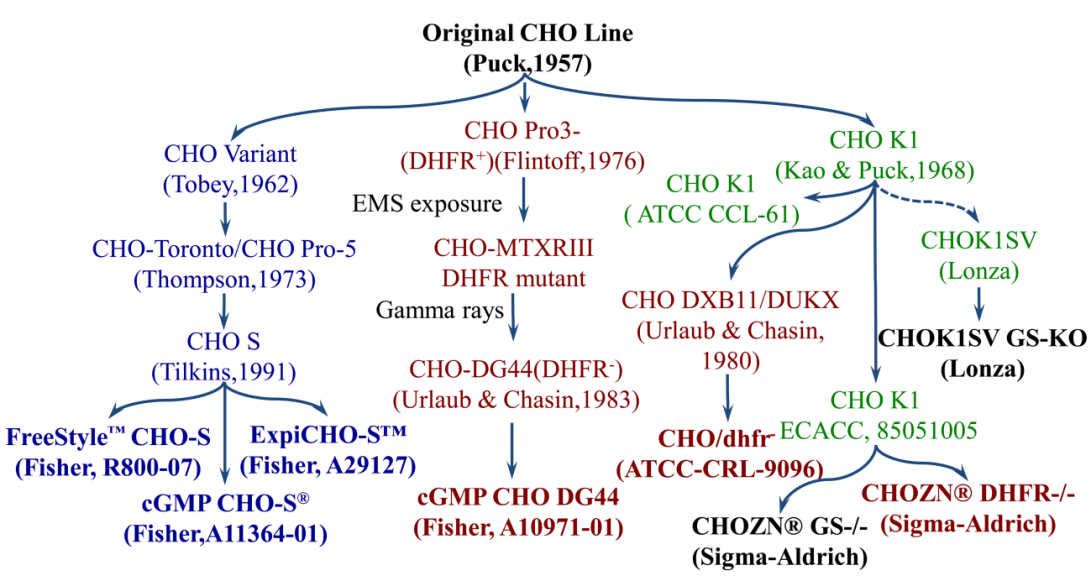
History of CHO Cells:
In 1957, T. Puck developed the first Chinese hamster ovary (CHO) cell line for cell genetics research. In the early 1980s, CHO cells gained increasing attention as host cells for the production of heterologous proteins. The original CHO cell line has been subcloned several times, and the most commonly used cell lines, CHO-k1, CHO DG44 and CHO-S are derived from different CHO lineages. CHO cells exhibit high diversity due to genetic and phenotypic instability, with different culture conditions leading to unique functional properties such as proliferation, glycosylation ability and levels of recombinant protein expression.
(Xu N, et al. Biochem Eng J. 2017)
CHO Cell Series:
CHO-S: In the early 1960s, Theodore Puck observed that a subset of CHO cells could grow as non-adherent suspension cultures. In 1973, the University of Toronto's Thompson tamed a CHO cell line suitable for suspension culture, enabling larger-scale cultivation in bioreactors. Subsequently, Gibco domesticated this cell line in CD CHO medium, establishing it under the name CHO-S. The adaptability of CHO cells to growth in suspension culture marked a paradigm shift in animal cell culture technology, ultimately paving the way for the large-scale production of recombinant protein therapeutics up to 20,000 L using stirred-tank reactors (STRs).
CHO-DG44: In 1983, Urlaub and Chasin constructed a CHO cell line, CHO-DXB11, completely lacking the DHFR locus. DHFR-deficient CHO cells cannot grow normally. Transfected cells, when treated with methotrexate (MTX), inhibit DHFR activity by competitively binding to the catalytic site of DHFR enzyme, preventing the conversion of dihydrofolate to the active tetrahydrofolate required for DNA synthesis. Gradual increases in MTX concentration in the culture medium of DHFR-deficient CHO cells, subjected to the transfection of vectors encoding the gene of interest (GOI) and DHFR, increase selection pressure on the cells. This leads to genomic amplification of DHFR and GOI, resulting in an increase in the number of transgene copies. Thus, surviving cell pools often exhibit enhanced cellular productivity of recombinant proteins.
CHO-K1: The unmodified wild-type CHO cells. Stored in ATCC since 1968, now known as CHO-K1 (ATCC CCL-61). A subclone isolated in 1985 is preserved in ECACC (85051005). Originally, CHO-K1 cells required serum supplementation in the culture process and grew as adherent cells. In 2002, Lonza domesticated CHO-K1 cells for suspension culture in serum-free medium, establishing the CHOK1 SV cell line, mainly used in combination with the GS selection system. In the GS selection system, the target gene and a single copy of the GS gene are delivered into the cells. Stable cells are selected in a medium lacking glycine, with additional addition of MSX to increase selection intensity, ultimately achieving the goal of selecting cell lines with high expression. In 2006, Merck domesticated CHO-K1 cells for suspension culture in chemically defined medium, resulting in the CHOZN CHO-K1 (Merck, 2006) cell line.
Advantages of CHO Cells:
⭕ Can undergo numerous post-translational modifications to obtain biotherapeutics, producing products almost identical to human quality, demonstrating excellent biocompatibility and drug activity.
⭕ Thrive well in both adherent and suspension cultures, exhibiting high productivity, making them an ideal choice for large-scale cultivation and GMP programs.
⭕ Many well-validated genetic tools available for optimizing CHO cells, from gene introduction to knockout, knockdown and post-translational silencing.
⭕ Remarkable number of approved products (used for approximately 50 biopharmaceutical drug productions approved in the EU and the US).
⭕ Applicable to serum-free, animal-free and protein-free culture conditions with better stability and safety.
We have developed a proprietary CHO cell antibody expression platform to meet the growing demand for recombinant antibody technology in pharmaceuticals, biotechnology, diagnostics and academia. This serum-free CHO cell transient expression system provides a faster and more economical antibody expression production. Additionally, it offers preliminary testing support for the generation of stable cell lines.