Compared to humans and mice (left), the B cell antibody sequences in rabbits (right) exhibit a preferential utilization of a limited subset of V (usually D) and J gene segments during the V(D)J gene rearrangement process. Although the initially generated antibody diversity is relatively restricted, rabbit B cells, upon egress from the bone marrow, migrate to gut-associated lymphoid tissue (GALT) where they undergo proliferation. They continue to undergo somatic hypermutation (SH) and gene conversion mediated by activation-induced cytidine deaminase (AID), resulting in mutations within the V(D)J genes. This significantly enhances antibody diversity and affinity. Consequently, rabbit monoclonal antibodies possess an enhanced ability to recognize phosphorylated peptides, carbohydrates, haptens, and small molecules. The lower right image depicts a tissue section of the rabbit cecum which serves as the primary site for rabbit GALT, proliferating B cells are stained green while non-proliferating B cells are stained red.
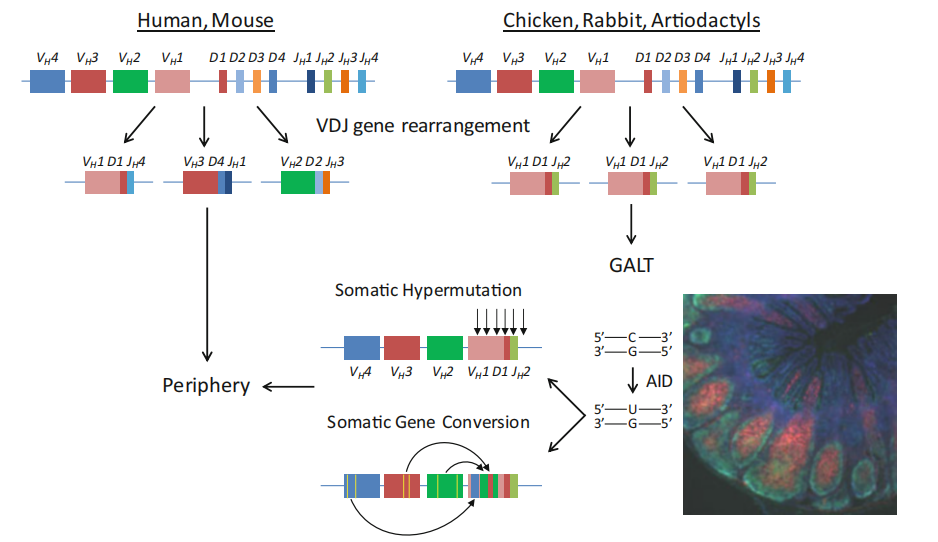
(Lanning DK, et al. Results Probl Cell Differ. 2015)
Compared to mice, rabbits exhibit a larger body size (with an average weight of experimental rabbits around 2.5 kg, while mice weigh approximately 25g), and the rabbit spleen is roughly 50 times bigger than that of a mouse. This facilitates the collection of a greater number of immune B cells and enables the production of a more diverse range of positive antibodies. Additionally, most mouse strains are inbred, which typically leads to less varied immune responses. Rabbits have simpler IgG subtype types (which aids in engineering modifications) and possess a more stable IgG structure. While mouse IgG primarily consists of five subclasses, rabbits lack IgG subclasses. Furthermore, rabbit IgG-κ1 constitutes approximately 90% of the light chain, and there exists an intrachain disulfide bond between C80 and C171 in the κ variable region, contributing to the stability of rabbit monoclonal antibodies.
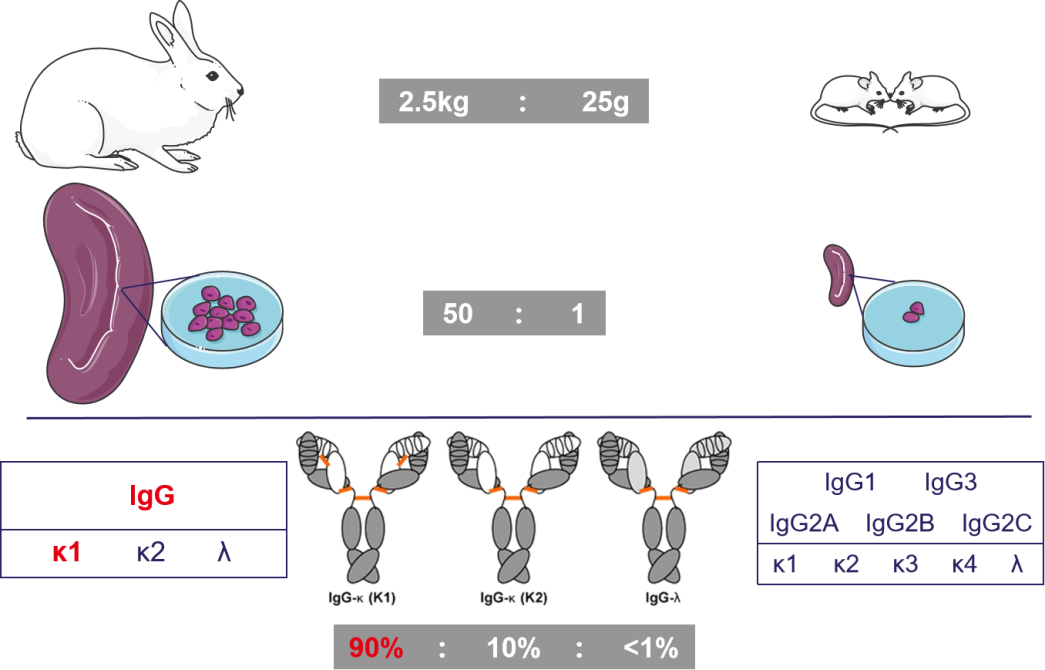
(Weber J, et al. Exp Mol Med. 2017)
Through the implementation of integrated optimization, we have successfully established a cutting-edge platform for the development of recombinant rabbit monoclonal antibodies from single B cells. This innovative platform has significantly expedited the screening process, traditionally requiring 1.5 months using hybridoma technology, to a remarkable timeframe of only 3-4 weeks. By harnessing the abundant resources of positive B cells, we have substantially enriched the diversity of potential antibody candidates. Furthermore, our rabbit-derived antibody molecules demonstrate unparalleled specificity and affinity, thereby providing robust support for your antibody development endeavors.
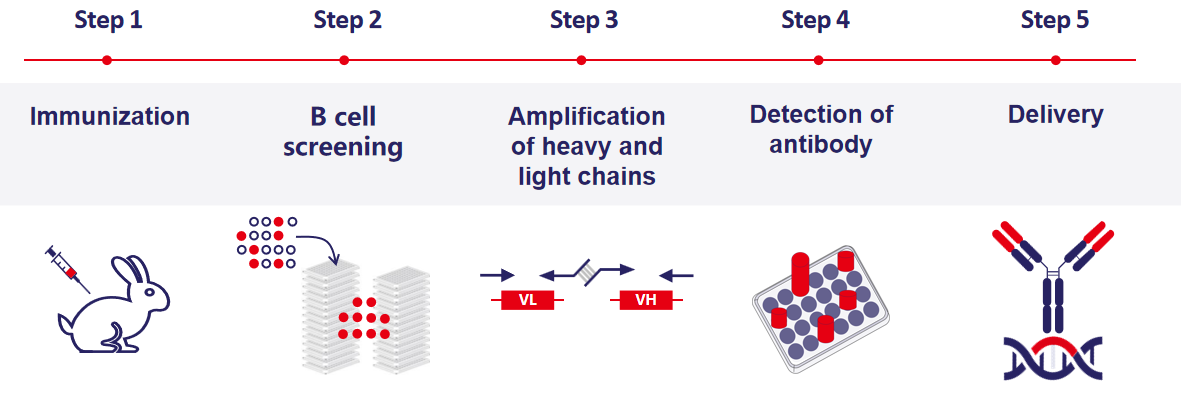
(Single B cell rabbit mab platform)