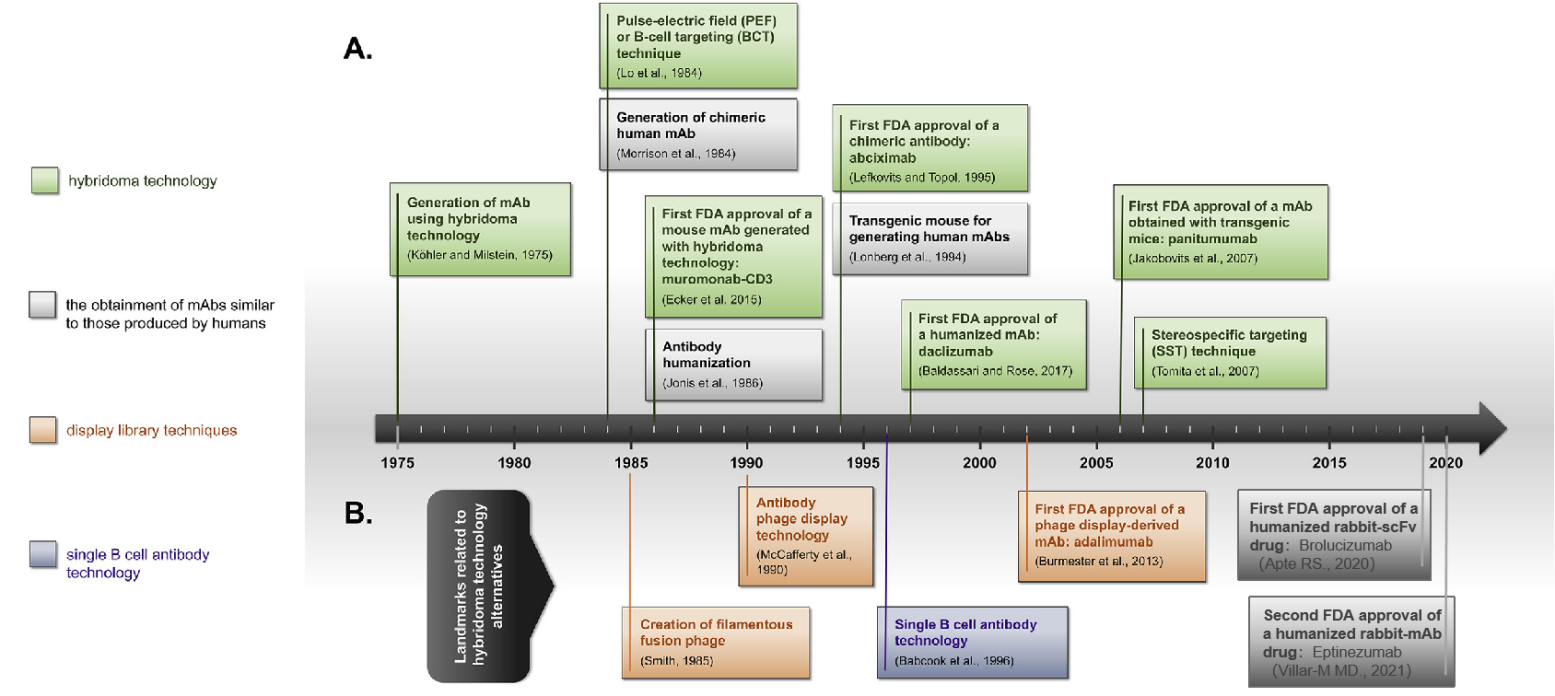
Since the first report of monoclonal antibody (mAb) development using hybridoma cell lines in 1975, to the approval of the first murine monoclonal antibody for human therapy in 1986, it became evident that engineering modifications could enhance the therapeutic efficacy of monoclonal antibodies. This realization sparked the construction of a series of novel antibodies: chimeric antibodies, humanized antibodies and fully human antibodies produced in transgenic mice (aimed at reducing the immunogenicity of rodent antibodies). Subsequent efforts focused on enhancing effector functions, controlling half-life, improving tumor and tissue accessibility, enhancing physical properties (such as stability), and more efficient (lower cost) engineering production.
(Moraes J. Z, et al. Current Research in Immunology. 2021)
One of the advantages of recombinant antibodies is their ability to undergo engineered modifications. With the primary amino acid sequence of an antibody, researchers can diversify and create a variety of antibody forms. Antibody engineering innovations have provided opportunities for designing diverse antibodies.

(Lou H, et al. Cancer Commun (Lond). 2022)
We have extensive experience in recombinant antibody engineering and expression. Leveraging mammalian cell expression platforms, combined with database analysis and molecular design, we can produce low-cost, high-yield engineered recombinant antibodies.