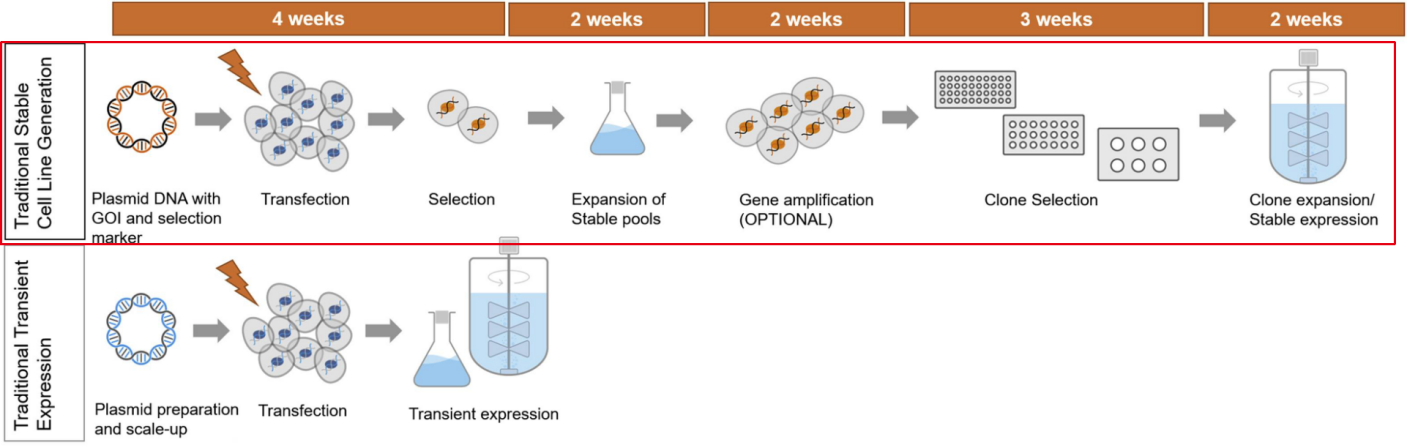
With the rapid development of protein/antibody biopharmaceuticals, immunodiagnostics and especially recombinant monoclonal antibodies, stable expression systems hold significant importance in biotechnological manufacturing. Unlike transient gene expression systems, stable cell line expression systems typically integrate foreign DNA into the host cell's chromosomes, ensuring stable expression and propagation as the cells proliferate.
(Luthra A, et al. Protein Expr Purif. 2021)
Expression Cell Lines:
Stable mammalian cell lines are the preferred choice for the production of recombinant proteins/antibodies, offering unique advantages. Currently, the most widely used mammalian cell lines in research or industry are human embryonic kidney cells and Chinese hamster ovary cells. For various reasons, CHO cells remain the preferred mammalian cell line for the production of recombinant protein therapeutics. Firstly, CHO cells can adapt and grow in suspension culture, making them an ideal choice for large-scale industrial cultivation. Secondly, CHO cells pose lower risks, as their susceptibility to human viruses is very low, reducing production losses and enhancing biosafety. Thirdly, CHO cells can grow in serum-free and chemically defined media, ensuring reproducibility between different batches of cell cultures. Lastly, CHO cells allow for post-translational modifications of recombinant proteins that are compatible with humans and biologically active. Specifically, the glycosylation of glycoproteins produced in CHO cells more closely resembles human glycosylation, lacking immunogenic α-galactose epitopes.
We independently developed and engineered high-expression CHO cell lines, optimizing the culture and selection systems to provide support and assurance for delivering satisfactory monoclonal cell lines.