
(DahlL,etal. SciAdv.2023)
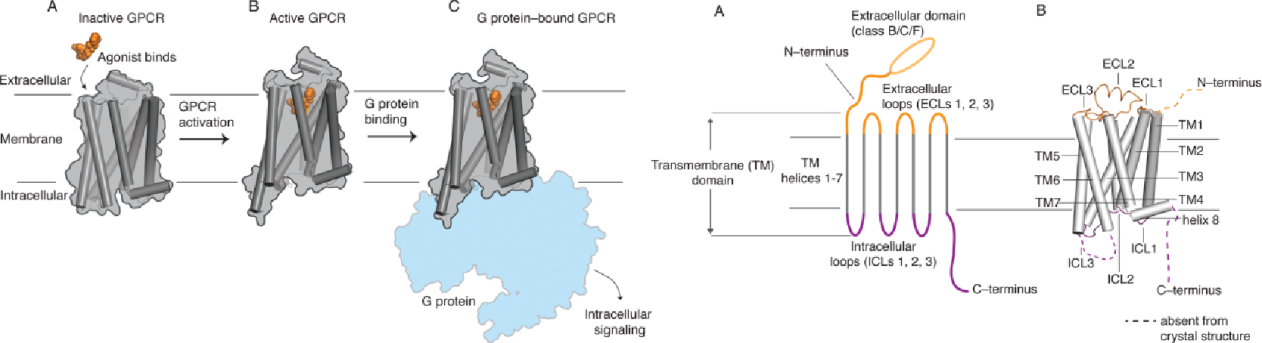
The G protein-coupled receptor (GPCR) superfamily is the largest family of human membrane proteins and drug targets, comprising approximately 750 membrane proteins. Their function primarily relies on their ability to change shape and transition between different conformations. They mediate numerous signaling pathways involved in cellular physiology and intercellular communication. Disruption of GPCR function or signal transduction is implicated in the pathophysiology of many disease states.
(LatorracaNR,etal. ChemRev.2017)
So, G protein-coupled receptors (GPCRs) are also targets for approximately 30% of approved therapeutic drugs. Although GPCRs represent one of the most targeted protein families in drug development, the dynamic nature of these membrane proteins poses challenges and limitations for the development of specific antibodies.
In May 2023, a research paper titled "Multiplexed selectivity screening of anti-GPCR antibodies" was published online in Advanced Science. This study employed a multiplexed immunoassay method to custom-test 407 anti-GPCR antibodies against a library of 215 GPCRs. The research provides insights into the immunogenicity of GPCR family proteins and the design and application of anti-GPCR antibodies as tool reagents and therapeutics, based on the data results.

Establishment of a Multiplexed Detection Method for Anti-GPCR Antibodies:
Researchers combined parallel expression of GPCRs with multiplexed protein capture analysis, using engineered tags to detect immunocaptured proteins and determine the relative quantities of GPCRs in solution. The data were then integrated into a network-based interface to explore the selectivity of each antibody for GPCR proteins. Of the 407 anti-GPCR antibodies tested, the majority were sourced from the Human Protein Atlas (HPA) program. The GPCR library was created by ectopic expression of 215 dual-site labeled GPCRs in 293F cells, each GPCR protein including an N-terminal Flag tag and a C-terminal 1D4 tag. Dodecylmaltoside (DM) was used as a detergent for washing, generating detergent micelles containing heterogeneous mixtures. Through multiplexed protein capture assays, the researchers determined which antibodies were selective in recognizing the intended GPCR targets.

(Experimental workflow and data analysis)
Discovery of Selective GPCR Antibodies:
In the selective binding screening of 407 antibodies against 205 GPCRs (although there were 215 GPCRs in the library, antibodies selected for 10 GPCRs did not pass QC testing and were only used as off-target candidates), 61% (248 out of 407) of the tested antibodies recognized their intended GPCRs. Of the remaining 159 antibodies, 9% (15 out of 159) enriched the intended targets along with at least one off-target, while an additional 20% (31 out of 159) of the remaining antibodies only bound to an unintended target, and 71% (113 out of 159) did not enrich any targets beyond the cutoff. The 248 highly selective antibodies corresponded to 154 unique GPCR targets.
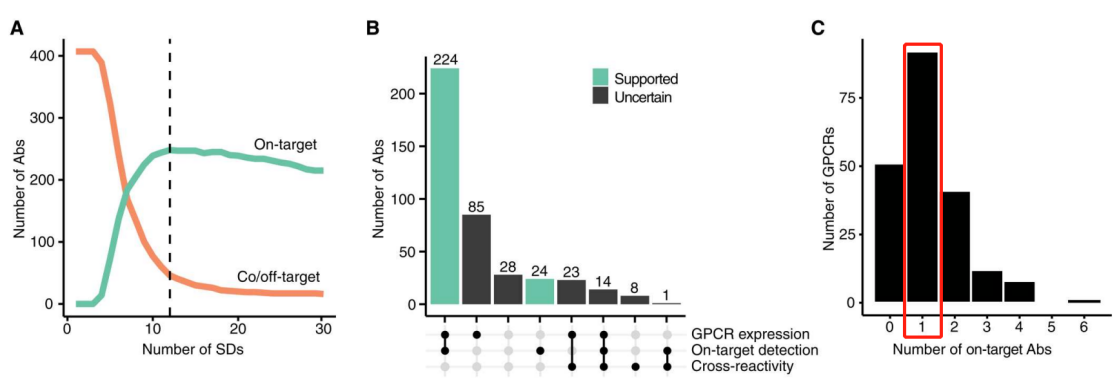
(Antibody Selectivity Thresholds and Summary Statistics)
Analysis of Different GPCR Sites:
Among the 205 GPCRs targeted by antibodies, 116 (57%) were targeted by two or more pairs of antibodies, allowing researchers to compare binding features with GPCRs and present different sites for selective and non-selective recognition of GPCRs.
For the rhodopsin family, the α subfamily member Adrenergic Receptor Alpha 2B (ADRA2B) was selectively recognized by all six antibodies, targeting three different regions, all located within the intracellular loop 3 (ICL3) of ADRA2B; the β subfamily member Gastrin-Releasing Peptide Receptor was selectively recognized by five out of six antibodies, targeting two different regions, one within the extracellular domain (ECD) and one within the extracellular loop 2 (ECL2); the δ subfamily member C-C Chemokine Receptor 7 (CCR7) was selectively recognized by one antibody, HPA060045, despite the use of three different antigens: one in the ECD, one in ECL2, and one corresponding to the receptor's intracellular C-terminal tail (CCR7's expression levels are lower than most receptors in the δ subfamily, thus classified as "uncertain," which may suggest that only antibodies with sufficiently high affinity for CCR7 can detect even lower levels of this GPCR); the γ subfamily member Follicle-Stimulating Hormone Receptor (FSHR) was selectively recognized by three antibodies, each targeting a different region within FSHR's large ECD.

(Rhodopsin Family Protein Recognition Site Map)
In the Glutamate Family, G Protein-Coupled Receptor Class C Group 5 Member D (GPRC5D) is selectively recognized by three out of four antibodies. The unsuccessful antibody HPA047203 was marked for recognizing the Extracellular Loop 2 (ECL2) region, while the selective antibodies targeted either its Extracellular Domain (ECD) (HPA071909) or the intracellular C-terminal tail (HPA064241 and HPA071739). HPA071739 cross-reacts with the related receptor GPRC5A; conversely, another antibody generated against the same antigen, HPA064241, exhibits high selectivity only for GPRC5D.

(GPRC5D Protein Recognition Site Ma)
For Corticotropin-Releasing Hormone Receptor 1 (CRHR1) tested with six antibodies, three are selectively recognized by CRHR1 and associated with an antigen representing Intracellular Loop 1 (ICL1).

(CRHR1 Protein Recognition Site Map)
Next, researchers analyzed epitope characteristics that lead to antibodies exhibiting specific or nonspecific binding, based on antigen structures predicted by AlphaFoldDB. According to the definition based on the low-predicted local distance difference test (pLDDT) scores, target antibodies are significantly enriched in regions predicted to be disordered in the protein. Additionally, defined by the average relative solvent accessible surface area (SASA), middle-target sites exposed to the surrounding environment are also enhanced.

(Distance Difference (pLDDT) and Relative Solvent Accessible Surface Area (SASA) Tests)
Conclusion:
After mapping the recognition sites based on polyclonal antibodies, specific monoclonal antibodies can be purified from the polyclonal mixture using the acquired sites. Following testing for specificity in expected determinations, the most suitable sites can be used to generate monoclonal antibodies. The paper also provides additional information on all antibodies and recognized sites, offering a design and screening strategy for guiding the development of GPCR target antibodies.

This literature vividly illustrates the limitations of anti-GPCR antibodies: ensuring stable specificity for GPCRs in different conformational states is crucial, requiring strong affinity characteristics. Combined with the scarcity of exposed sites on GPCR proteins, the development of antibodies is challenging. Rabbit-derived antibodies, with their smaller antigen recognition sites and the rabbit-specific high-affinity mature system, serve as an excellent immunological source.
