Fibronectin (FN1), also known as CIG, is involved in cell adhesion, cell motility, opsonization, wound healing, and maintaining cell shape. Fibronectin is a key component of the extracellular matrix (ECM), and the FN1 gene plays an important role in angiogenesis, tissue repair, fibrosis, and cancer.
FN1 expression distribution
FN1 expression is tissue-specific, primarily found in the liver, placenta, and blood vessels. It is also expressed in trophoblasts, stellate cells, hepatocytes, smooth muscle cells, fibroblasts, and stromal cells. FN1 is a non-cellular extracellular matrix protein that is highly expressed in the stroma of many tumors (including head and neck squamous cell carcinoma), but is expressed very little in normal adult tissues.
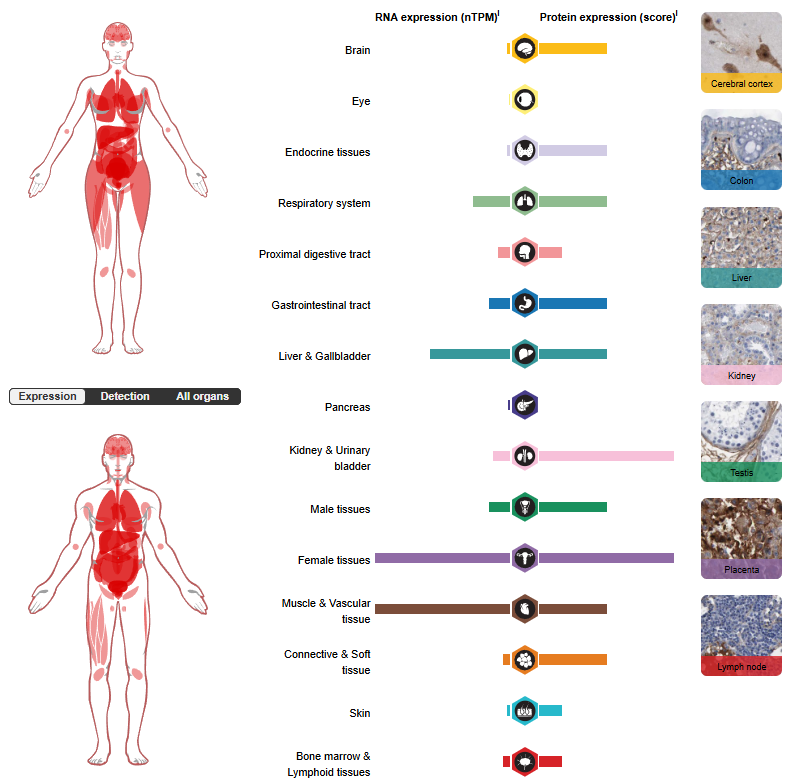
(Data source: uniprot)
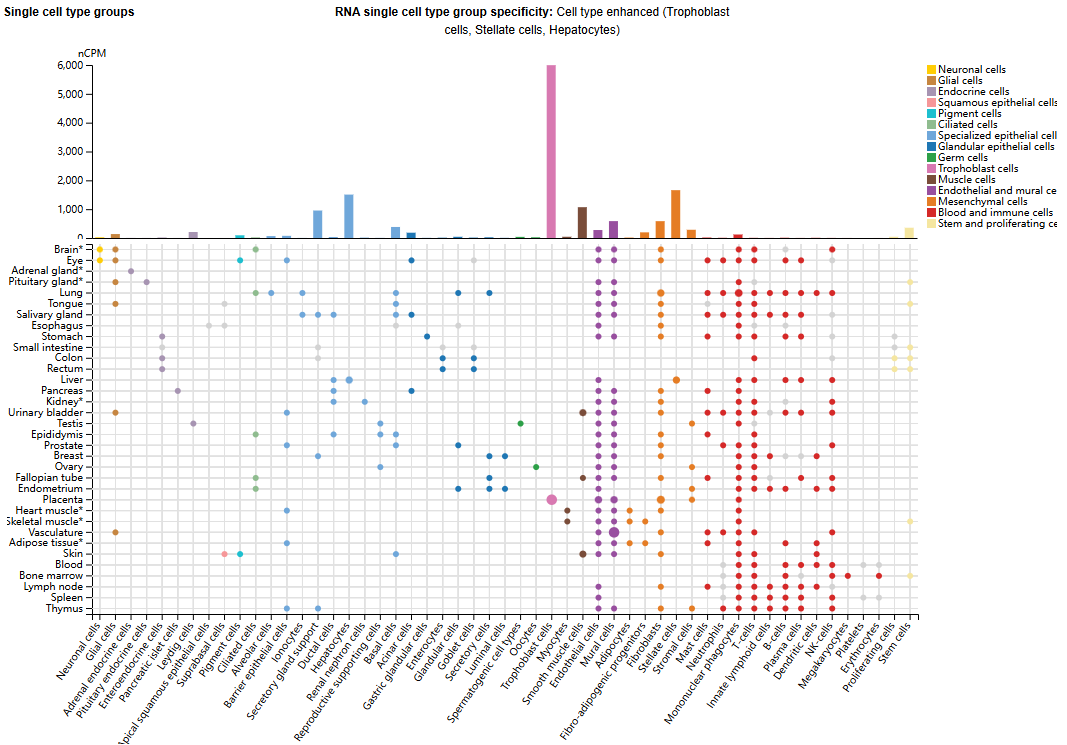
FN1 structure
FN1 consists of 2477 amino acids and is composed of two similar but different monomers of 220 and 250 kDa, connected by two disulfide bridges at the C-terminus of the protein. Each monomer consists of three different modules: type I (FNI), type II (FNII), and type III (FNIII).
The FNI module consists of 45 amino acids. It contains 12 FNI repeat sequences, 2 FNII repeat sequences, 15 constitutively expressed and 2 alternatively spliced (referred to as Extra Domains, EDA and EDB) FNIII repeat sequences, and a non-homologous variable (V) or type III linker region (IIICS) . The FNII module consists of 60 amino acids, including cysteine residues responsible for forming disulfide bridges within the domain. Each FNIII module consists of a shared sequence of approximately 90 residues arranged in seven β chains, forming two antiparallel β-sheets . The major part of the FN1 structure is composed of FNIII modules.
FN1 exists in plasma as a soluble dimer (pFN1) and as an insoluble portion of the fibrous network in the ECM (cFN). Plasma FN1 is secreted directly into the blood by hepatocytes in a soluble and inactive heterodimer form, with each subunit containing one or two IIICS segments, but lacking EDB and EDA modules.
Cellular FN1 consists of a series of heterogeneous subtypes, which are composed of different proportions of EDA and EDB domains and IIICS, and participate in the composition of the extracellular matrix in a tissue-specific manner.

(Data source: Spada S , et al. J Exp Clin Cancer Res. 2021)
Fibrous formation of FN1
During development, tissue homeostasis, and cancer, the role of FN1 in the composition of the extracellular matrix is primarily attributed to cFN1. Cellular FN1 dimers possess intrinsic and unique properties that enable the generation of extracellular matrix within tissues and organs; this is mainly due to their ability to generate fibrous FN1 matrix, a cell-dependent, dynamic, and complex process involving the multimodal and multidomain structure of FN1.
FN1 dimers maintain a compact conformation through disulfide bonds at the C-terminus of each subunit. FN1 binding to integrins induces conformational changes, causing the protein to unfold and bringing FN1 molecules closer together on the cell surface. This determines integrin aggregation and activation, accompanied by the recruitment of FAK, Src kinase, paxillin, and talin, generating an adhesion complex that activates actin filament polymerization. The actin cytoskeleton network binds to the cytoplasmic domain of integrins, forming a connection crucial for the initial generation of the FN1 matrix. RhoA GTPase-mediated cytoskeleton contractility and actin-myosin interactions induce cellular traction, resulting in FN1 conformational changes that open hidden FN1-FN1 binding sites—a necessary condition for FN1 filament formation.
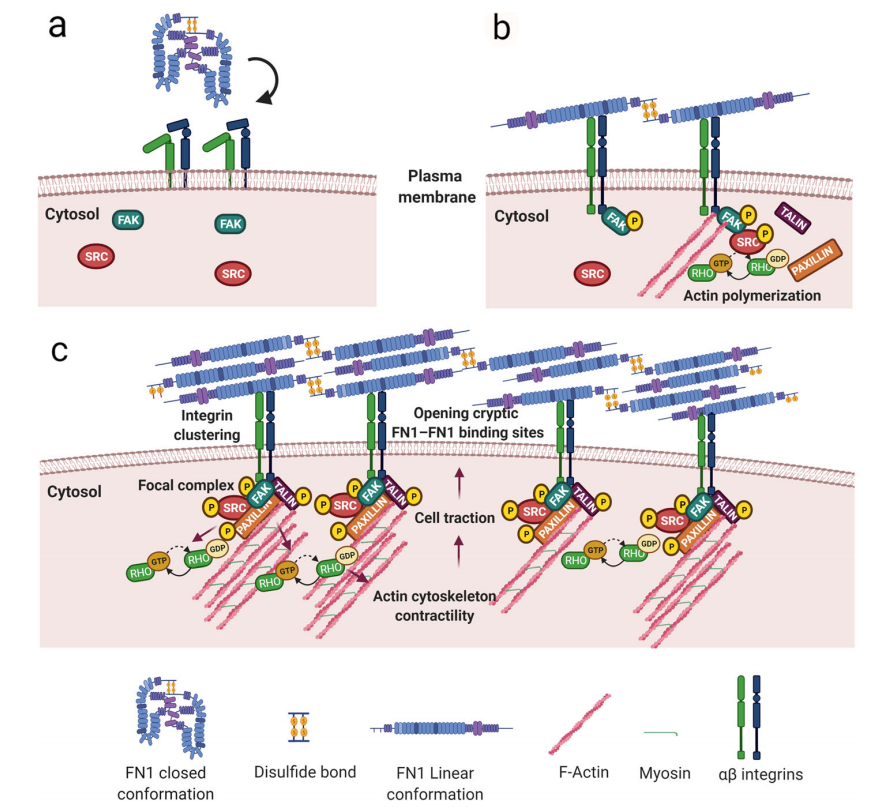
(Data source: Spada S , et al. J Exp Clin Cancer Res. 2021)
FN1 signaling pathway regulation and its role in tumor regulation
At the primary tumor site, cancer cells and cancer-associated fibroblasts (CAFs) secrete FN1. FN1 activates intracellular signaling mediated by integrins, TLRs, Wnt/β-catenin, and PI3K, leading to increased expression and secretion of FN1, MMPs, and TGFβ. Simultaneously, FN1 can migrate via exosomes. Invasive cancer cells can infiltrate and reach secondary organs, where immune-controlled tumor dormancy may occur. In metastatic proliferation, dormancy escape occurs in a permissive microenvironment with an extracellular matrix rich in FN1 and collagen, thus affecting resistance to treatment. The EDB isoform of FN1 has been used as a biomarker for cancer diagnostic imaging, while the FN1 pathway serves as a target for therapeutic applications.

(Data source: Aladal M, et al. Biomed Pharmacother. 2022)

(Data source: Spada S, et al. J Exp Clin Cancer Res. 2021)
Targeted therapy for FN1
Micvotabart (MICVO, formerly known as PYX-201) is an antibody-drug conjugate (ADC) that uniquely targets the extra domain B splice variant (EBD+FN) of fibronectin. MICVO possesses potential antitumor activity through three synergistic mechanisms.
MICVO is designed to bind to EDB+FN targets and release its payload within the tumor ECM to directly kill tumor cells; through bystander killing, the payload diffuses to neighboring tumor cells; tumor death and exogenous antibody fragments lead to T cell activation, triggering host immune activation and enhancing the treatment response.

MaMICVO (a mouse model of MICVO) and anti-PD-1 drugs synergistically enhance the efficacy of immunotherapy-resistant MOC2 tumors, highlighting the potential clinical benefit of MICVO in HNSCC patients who are unresponsive to immune checkpoint blockade.
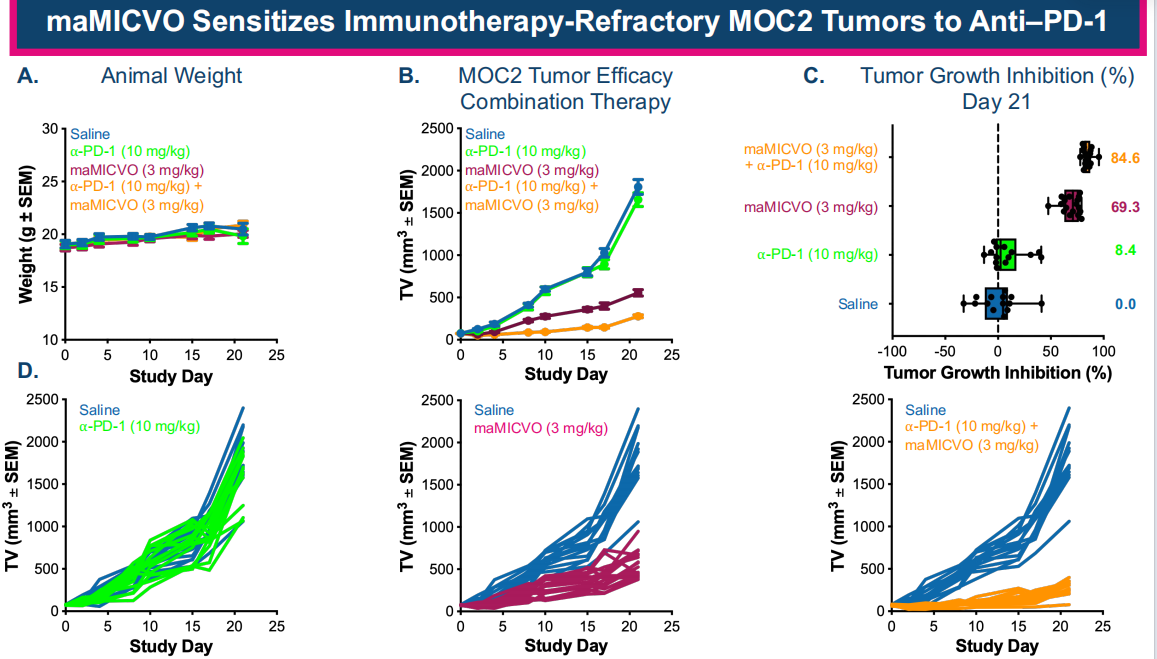
Pyxis Oncology is advancing the use of MICVO as a monotherapy (NCT05720117) and in combination with KEYTRUDA® (pembrolizumab) (NCT06795412) in a variety of solid tumors, with a focus on recurrent/metastatic head and neck squamous cell carcinoma.
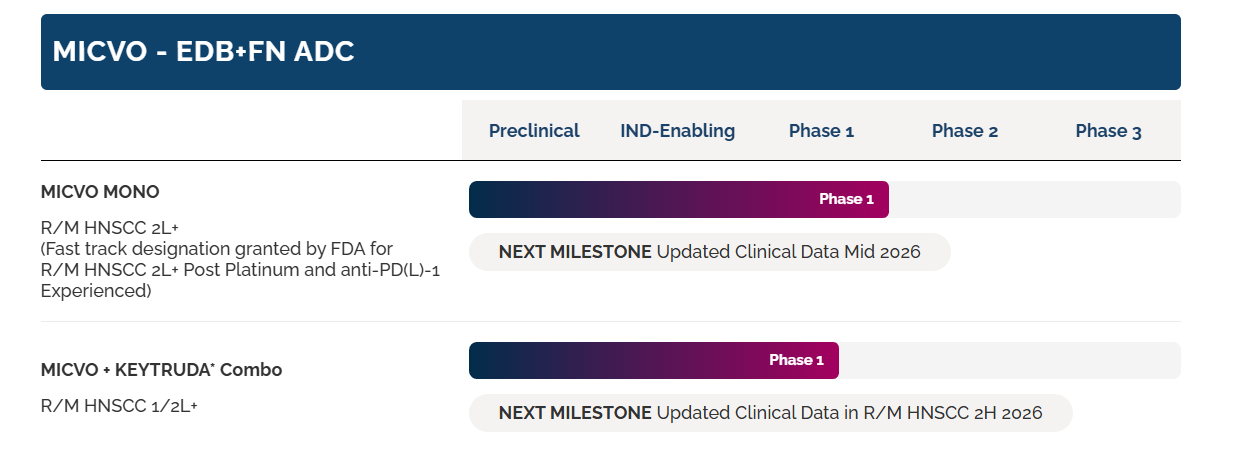
(Data source: Pyxis Oncology official website)
