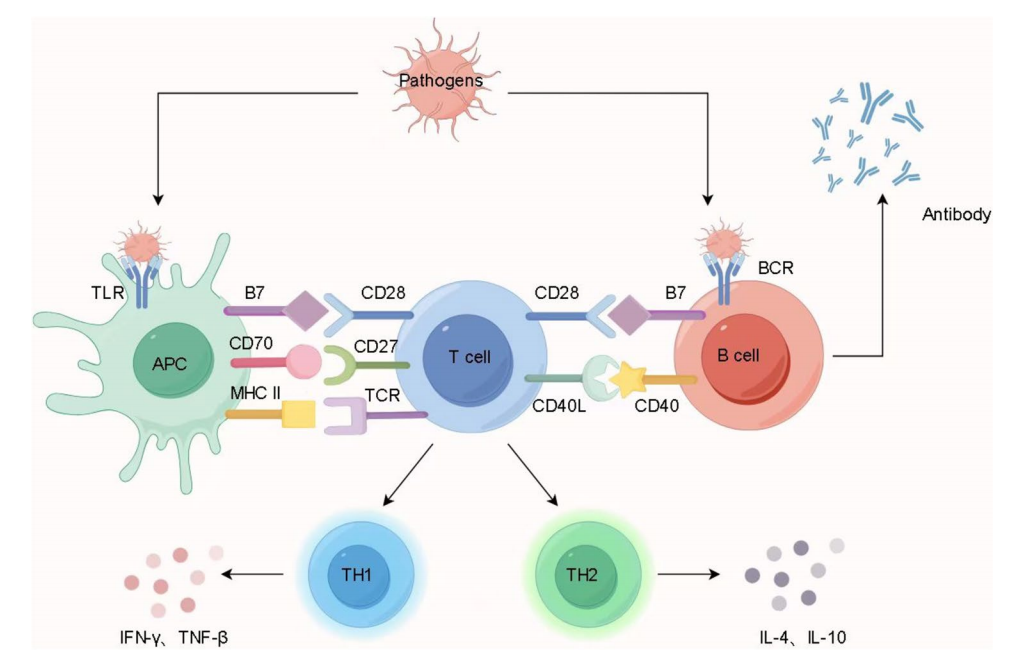
CD27 is a member of the tumor necrosis factor receptor superfamily 7 (TNFRSF7). It is a costimulatory immune checkpoint receptor expressed on the surface of T cells, NK cells, and B cells. This receptor binds to and is activated by the ligand CD70/CD27L expressed by B cells. CD27/CD70 mediates the activation and expansion of antigen-specific T cells, thereby providing immune surveillance for B cells. CD27/CD70 increases the production of interferon-γ (IFN-γ) by NK cells to promote proliferation and cytotoxicity, thus affecting the innate immune system.
(Data source: Song XF, et al. Cardiovasc Drugs Ther. 2025)
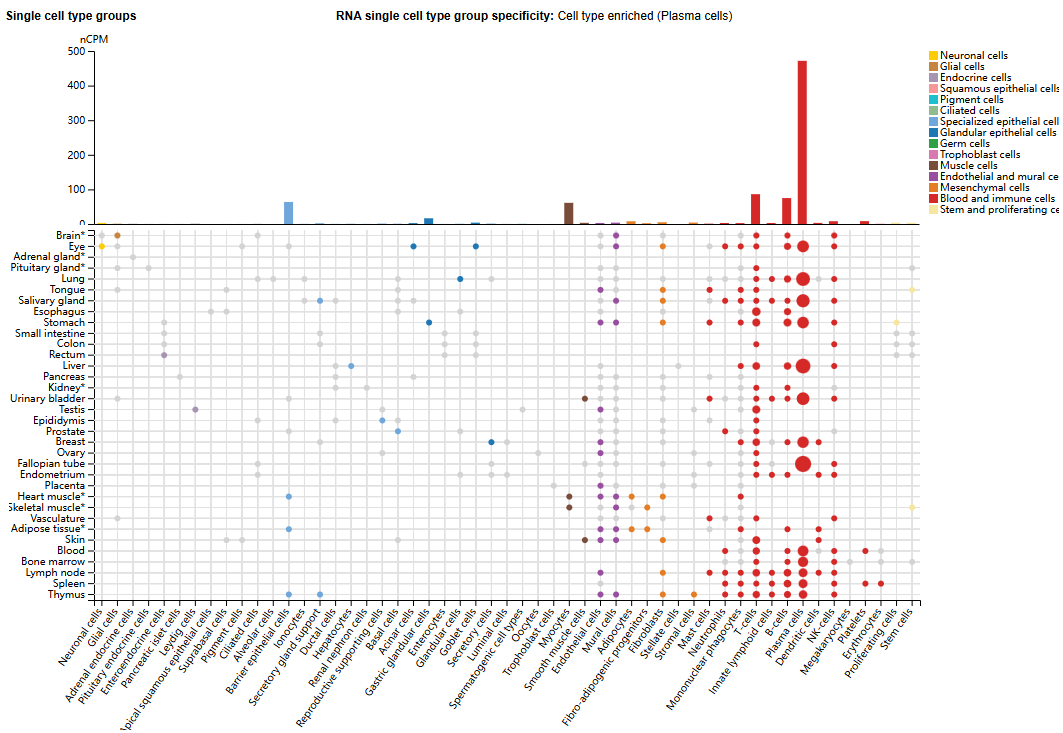
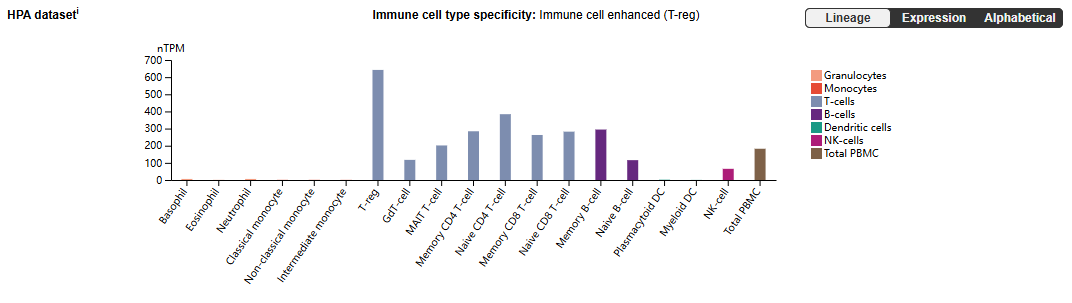
CD27 expression distribution
CD27 is mainly expressed in immune cells such as plasma cells, T cells (CD4+T and CD8+T cells), B cells, DC cells, and NK cells. It is also expressed in intestinal epithelial cells, gastric gland cells, and cardiomyocytes.
(Data source: uniprot)
Structure and ligands of CD27
CD27 is a type I transmembrane protein composed of 260 amino acids, including a signal peptide, an extracellular region, a transmembrane region, and an intracellular region. CD27 exists on the cell surface as a disulfide-linked homodimer.
The extracellular region contains three TNFR-Cys repeats, which are responsible for binding to the ligand CD70. The intracellular region is rich in the TRAF-binding motif (PxQxT), which is responsible for pro-survival and co-stimulatory signaling, rather than apoptosis induction.
The natural ligand of CD27 is CD70, which is expressed rather strictly and only temporarily on activated immune cells, including T cells, B cells, dendritic cells (DCs) and NK cells.
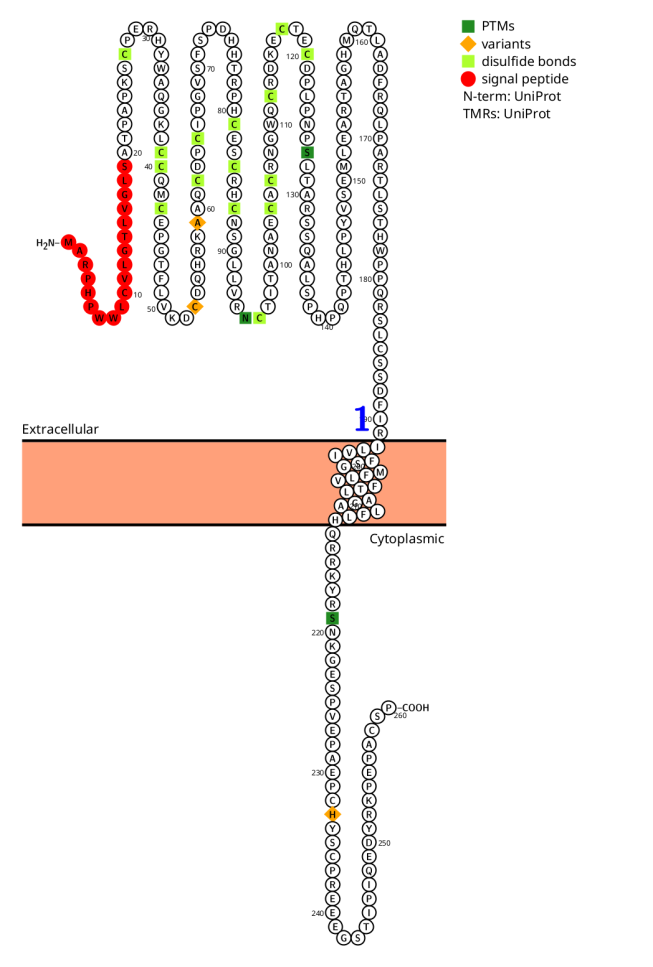
(Data source: protter)
The role of CD27 in immune cells
The role of CD27 in T cells
CD27, as a co-stimulatory molecule, provides a second signal (co-stimulatory signal) for T cell activation . Studies have shown that within the CD4+ T cell population, CD4+ T cells can differentiate into various helper T (TH) cell subsets, such as TH1, TH2, TH17, and Treg, playing different roles in the immune system. The CD27-mediated second signal can also promote the proliferation and survival of naïve human CD4+ T lymphocytes in vitro.
Upon binding to its ligand CD70, CD27 activates T cells via both classical and non-classical NF-κB pathways, upregulating anti-apoptotic molecules (BCL-XL) and cytokine receptors (IL-2Rα, IL-12Rβ), thereby enhancing CD8+ T cell survival and cytokine sensitivity. CD27 co-stimulation promotes antigen-specific CD8+ T cell proliferation, enhances cytotoxic function, and increases cytokine (IFN-γ, TNF, IL-2) production. CD27 signaling promotes the transformation of effector T cells into memory T cells by maintaining IL-7Rα expression; however, persistent CD27 stimulation can lead to T cell exhaustion and impaired memory function. Enhanced CD27 co-stimulation promotes the recall response and secondary proliferation of memory CD8+ T cells, while CD27 deficiency delays virus-specific T cell responses and reduces viral clearance capacity.
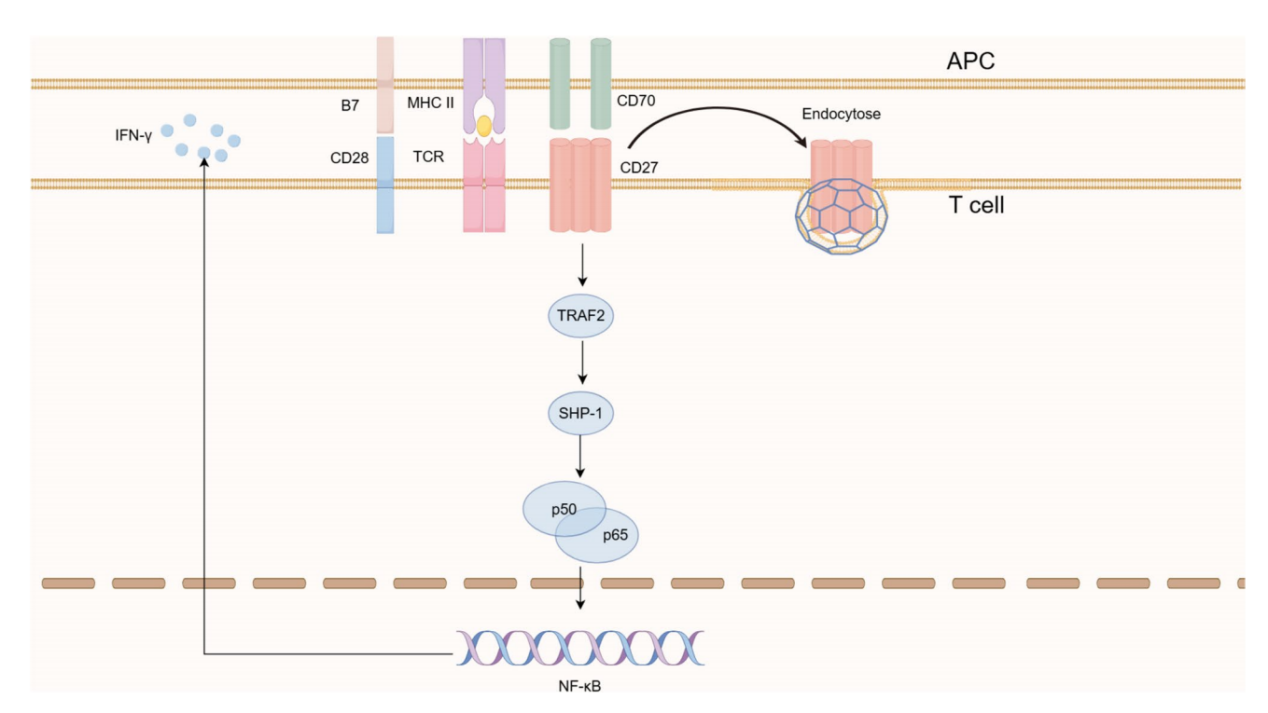
(Data source: Song XF, et al. Cardiovasc Drugs Ther. 2025)

(Data source: Grant EJ, et al. Curr Opin Virol. 2017)
The role of CD27 in B cells
CD27 is a member of the TNF receptor superfamily. Its ligand CD70 provides a co-stimulatory signal for B cell activation, promoting IgG production and plasma cell differentiation. In the germinal center dark zone, B cells proliferate and upregulate CD27 expression, and CD27 expression levels are positively correlated with the frequency of somatic high-frequency mutations. CD27-CD70 interaction is crucial for GC (germinal center) formation; CD27 deficiency leads to reduced GC volume, a significant decrease in CD27+ memory B cells, and impaired T-dependent antibody responses. CD27dull is an early low-mutation subset, while CD27bright is a highly mutated subset selected by GC; both work together to maintain the flexibility and stability of immune memory. CD27 is not only a surface marker but also a core functional molecule regulating GC responses, affinity maturation, and the generation of long-lived plasma cells.
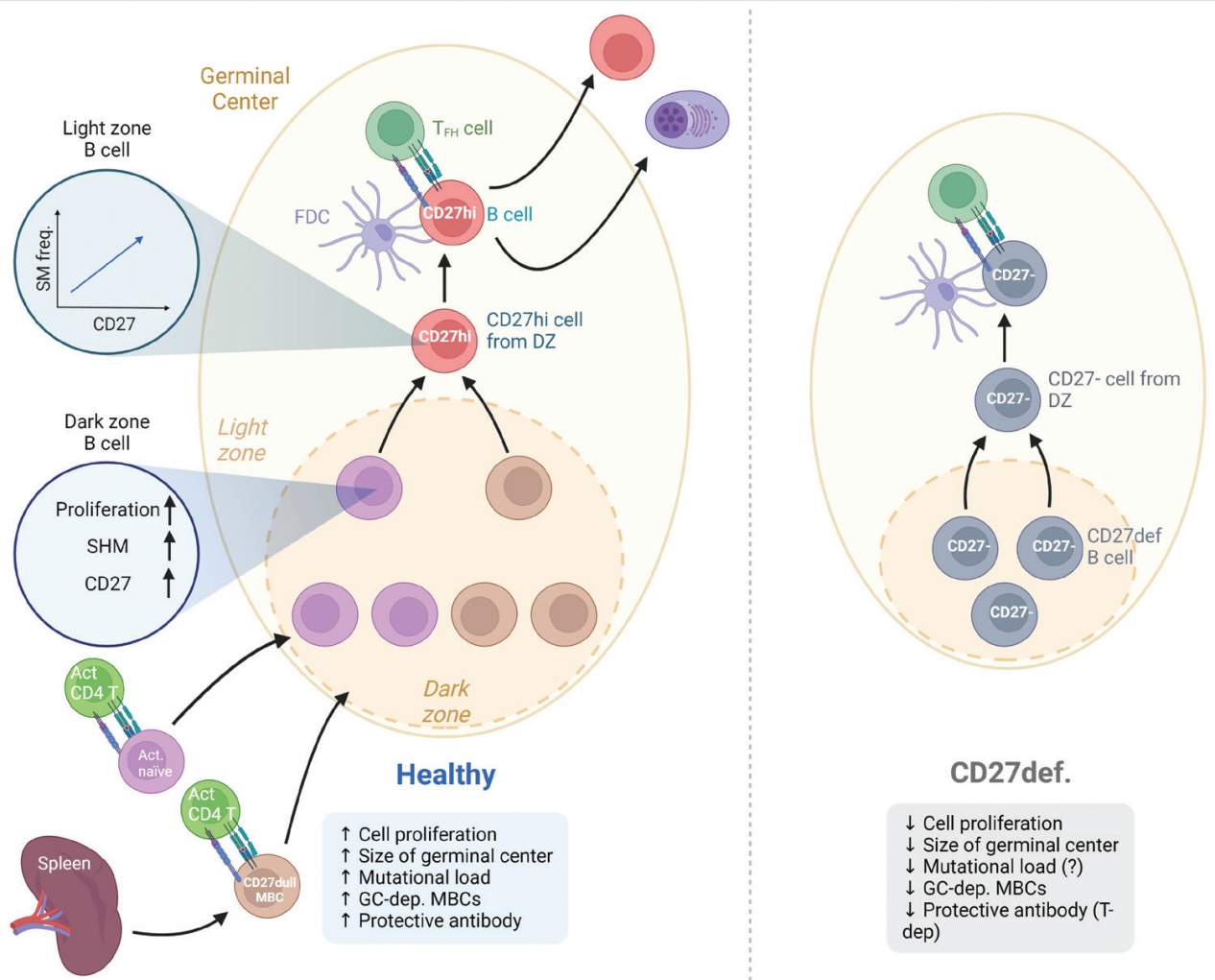
(Data source: Grimsholm O, Clin Exp Immunol. 2023)
CD27-targeted therapy
Given the central role of CD27 in activating and maintaining anti-tumor immune responses, it has become a highly promising target in the field of tumor immunotherapy, and there are currently a number of antibody drugs in clinical development and preclinical research.
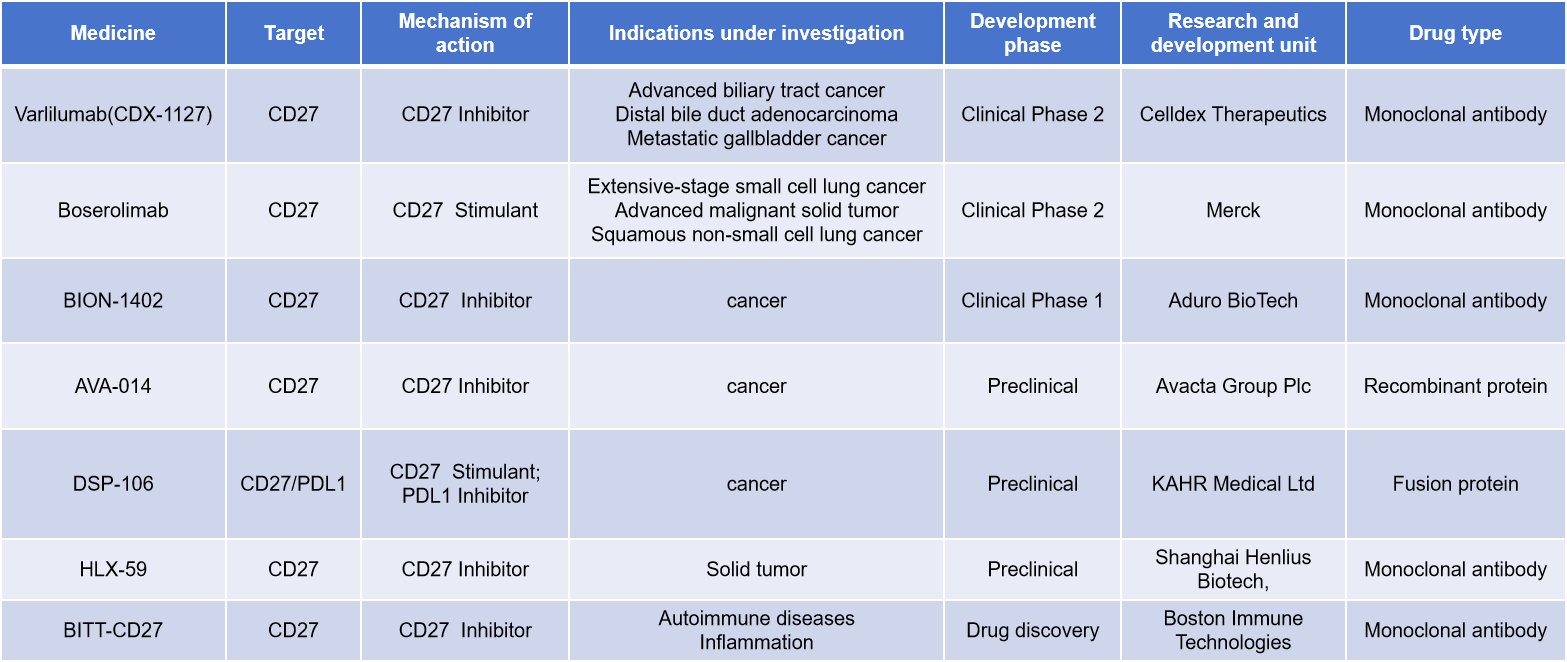
(Data source: Synapse)
Varlilumab (CDX-1127) is a monoclonal antibody targeting CD27, currently in phase 2 clinical trials. The RiVa trial (NCT03307746) evaluated the combination therapy of varlilumab and rituximab (a tumor-depleted CD20 antibody) in patients with relapsed/refractory B-cell non-Hodgkin lymphoma (B-NHL). Gene set enrichment analysis (GSEA) showed no enrichment of immune cell activation gene tags in the rituximab monotherapy group, while the combination therapy group exhibited significant enrichment of immune cell activation gene tags, confirming that varlilumab can induce in vivo immunostimulatory activity in the B-cell lymphoma tumor microenvironment. Although the combination therapy had a good safety profile, the overall clinical response was limited (overall response rate 15.4%). Studies have shown that CD27 agonists can overcome the immunosuppressive tumor microenvironment and drive clinically significant antitumor activity, but their efficacy depends on sufficient intratumoral target expression and a pre-existing "immune-primed" tumor state. Therefore, accurate patient screening is key to realizing the clinical value of such agonists.

(Data source: Buermann LE, et al. Clin Cancer Res. 2025)
Boserolima b (MK-5890) is an anti-CD27 monoclonal antibody. A phase 2 clinical trial (NCT06829199) was conducted in 2025 to treat early-stage triple-negative breast cancer patients with boserolima b in combination with the PD-1 antibody pembrolizumab and chemotherapy. Data showed that in 41 evaluable patients, the objective response rate reached 51%, the disease control rate reached 80%, and the median progression-free survival (PFS) was 11.1 months.
