Interleukin IL-20 belongs to the IL-20 subfamily of the IL-10 family. The IL-10 family mainly includes IL-10, IL-19, IL-22, IL-24, IL-26, IL-28, and IL-29; among them, IL-19, IL-20, IL-22, IL-24, and IL-26 belong to the IL-20 subfamily. They all play important roles in regulating immune responses, promoting inflammatory responses, and maintaining tissue homeostasis. Interleukin IL-20 is a pro-inflammatory and pro-angiogenic cytokine mainly secreted by monocytes and keratinocytes. For example, it affects keratinocyte proliferation, leading to psoriasis. Furthermore, it induces the production of tumor necrosis factor (TNF)-α and IL-6 through monocytes and stimulates the expression of growth factors in CD8+ T cells. As a pleiotropic cytokine, IL-20 is mainly involved in immune regulation, inflammatory responses, and tissue remodeling processes, playing an important role in skin homeostasis, bone metabolism, and the tumor microenvironment.

(Data source: Goleij P, et al. Int Immunopharmacol. 2025)
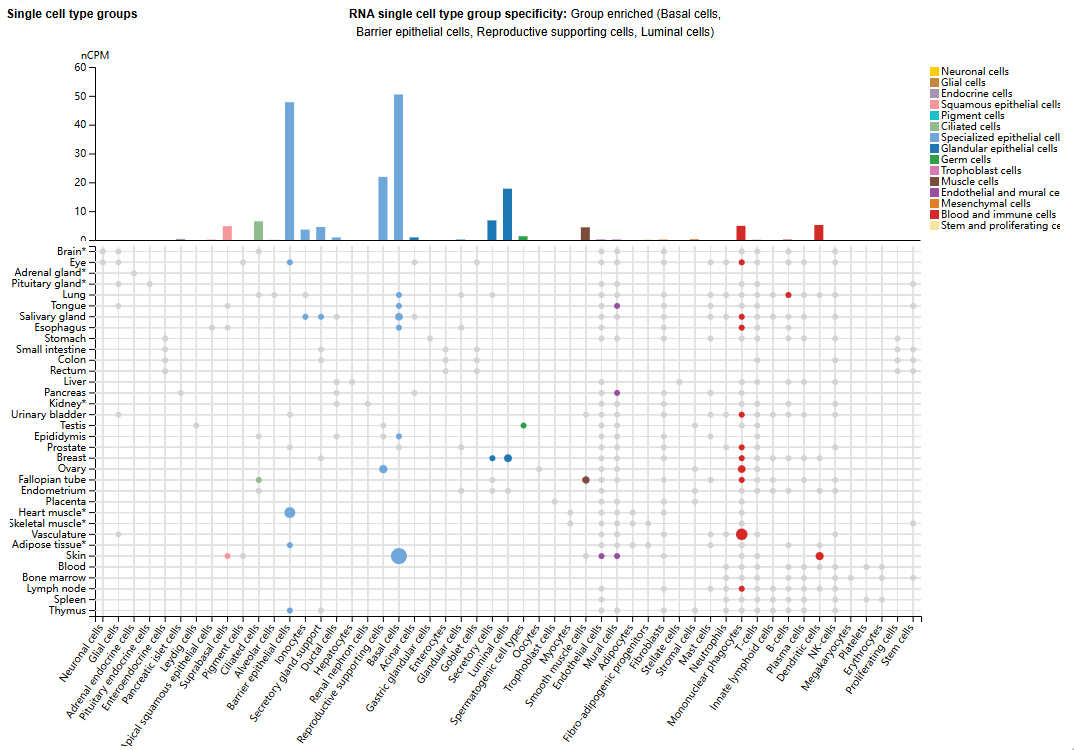
IL20 expression distribution
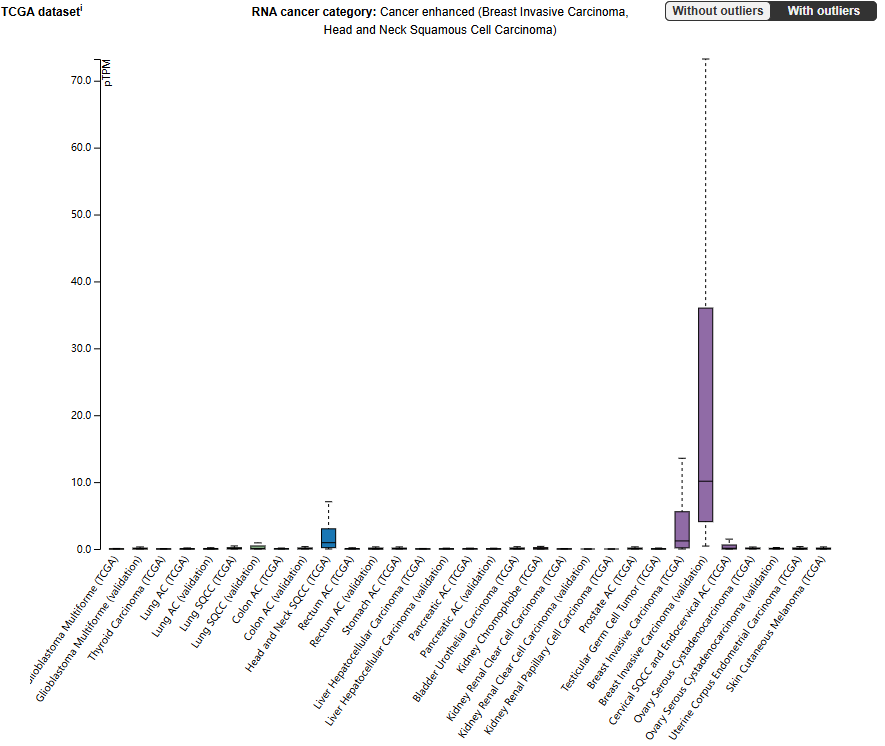
IL20 is mainly expressed in basal cells, barrier epithelial cells, germ support cells, and luminal cells. It is also expressed in macrophages and tumor dendritic cells. In tumor cells, it is mainly expressed in invasive breast cancer and squamous cell carcinoma of the head and neck.
(Data source: uniprot)
The structure of IL20 and its receptor
IL-20 is a secreted protein composed of 176 amino acids. Structurally, the IL-20 protein contains a typical four-helix bundle structure, a conserved feature of IL-10 family members, which helps maintain its stability and biological activity. IL-20 family receptors are typically composed of heterodimeric complexes formed by different receptor subunits.
The functional receptor for IL-20 consists of two subunits: IL-20Rα (ligand recognition chain) and IL-20Rβ (signal transduction chain). Both subunits belong to the type I cytokine receptor family. IL-20 binds with high affinity to the IL-20Rα/β complex, initiating downstream signaling. IL-20 also has a type II cytokine receptor, IL-22RA1, which binds to IL-20 together with IL-20RB.

(Data source: Maggisano V, et al. Int J Mol Sci. 2025)
The role of IL20 in disease
IL20 plays an important role in inflammation regulation and has effects in liver disease, psoriasis, rheumatoid arthritis and inflammatory bowel disease (IBD), some neurodegenerative diseases, and cancer.
Its role in liver diseases
In the inflammatory environment of the liver, IL-20 phosphorylates and activates the heterodimeric receptor complexes IL-20R1/IL-20R2 and IL-22R1/IL-20R2. Upon binding to its receptors, IL-20 activates JAK1 and TYK2, recruits and phosphorylates the transcription factor STAT3, which enters the nucleus in dimer form and binds to target genes, regulating the transcription of downstream genes. During bacterial infection of the liver, IL-20 activates the ERK1/2 and p38 MAPK signaling pathways via IL-20R2/IL-22R1, stimulating the expression of the NRF2 target enzyme NQO1, promoting the degradation of IκBζ, and inducing liver damage. Simultaneously, stimulated immune cells release large amounts of IL-1β, which further induces IL-20 gene expression through a MAPK and NF-κB-dependent mechanism.


(Data source: Wang K, et al. Heliyon. 2024)
The role of IL20 in psoriasis
IL-20 is highly expressed in patient skin lesions, promoting excessive proliferation of keratinocytes, inflammatory infiltration and angiogenesis, and is one of the core driving factors of the disease.
The role of IL20 in bone diseases
IL-20 plays a crucial role in the regulation of osteoclastogenesis and osteoblastogenesis, and is upregulated in various bone-related diseases. IL-20 is also important in the development of inflammation-mediated bone loss diseases such as rheumatoid arthritis (RA), osteoporosis, cancer-induced bone destruction, and fractures. In rheumatoid arthritis, IL-20 participates in joint destruction by promoting synovial cell activation, osteoclast differentiation, and bone erosion.

(Data source: Wang HH, et al. J Biomed Sci. 2018)
The role of IL20 in neurodegenerative diseases
IL-20 plays a crucial role in the pathogenesis of neurodegenerative diseases due to its multiple functions in inflammation, tissue repair, and immune regulation. These cytokines trigger various immune processes through IL-20 receptor complex signaling. IL-20 family cytokines affect microglia activation; dysregulation of microglia activation exacerbates neuronal damage. IL-20 is associated with elevated pro-inflammatory markers in glial cells, promoting neurodegeneration.

(Data source: Goleij P, et al. Int Immunopharmacol. 2025)
The role of IL20 in cancer
Members of the IL-20 subfamily are primarily pro-inflammatory cytokines that provide an environment that promotes and supports cancer growth, playing a crucial role in tumor development. These cytokines influence tumorigenesis by regulating cancer characteristics, including dysregulation of proliferation, resistance to cell death, induction of angiogenesis, invasiveness, and metastasis.
IL-20 is highly expressed in breast cancer tissues and promotes breast cancer growth in both in vitro and in vivo models, triggering the activation of signaling pathways involved in cell proliferation and anti-apoptotic signals such as Bcl-XL and Bad. IL-20 enhances the migration and invasion capabilities of cancer cells by increasing the production of MMP-9 and MMP-12 in breast and bladder cancer cells.

(Data source: Maggisano V, et al. Int J Mol Sci. 2025)
Targeted therapy for IL20
Fletikumab (NN8226, NNC0109-0012) is an IgG4 monoclonal antibody targeting IL-20 developed by Novo Nordisk for the treatment of psoriasis, rheumatoid arthritis, and other inflammatory diseases. It has been tested in clinical trials. In a Phase 1 clinical trial (NCT01038674), the drug demonstrated safety and good tolerability. In a Phase 2a clinical trial, neutralization of IL-20 was shown to be effective, particularly in rheumatoid factor (RF)-positive RA patients, further confirming the link between IL-20 and RF (NCT01282255). However, a Phase 2b clinical trial of this drug candidate failed to meet the primary efficacy endpoint of ACR20 (at 12 weeks) in RA patients who had an inadequate response to anti-TNFα (NCT01636817) or methotrexate (NCT01636843). There are several potential reasons for the failure of this anti-IL-20 antibody in clinical trials.
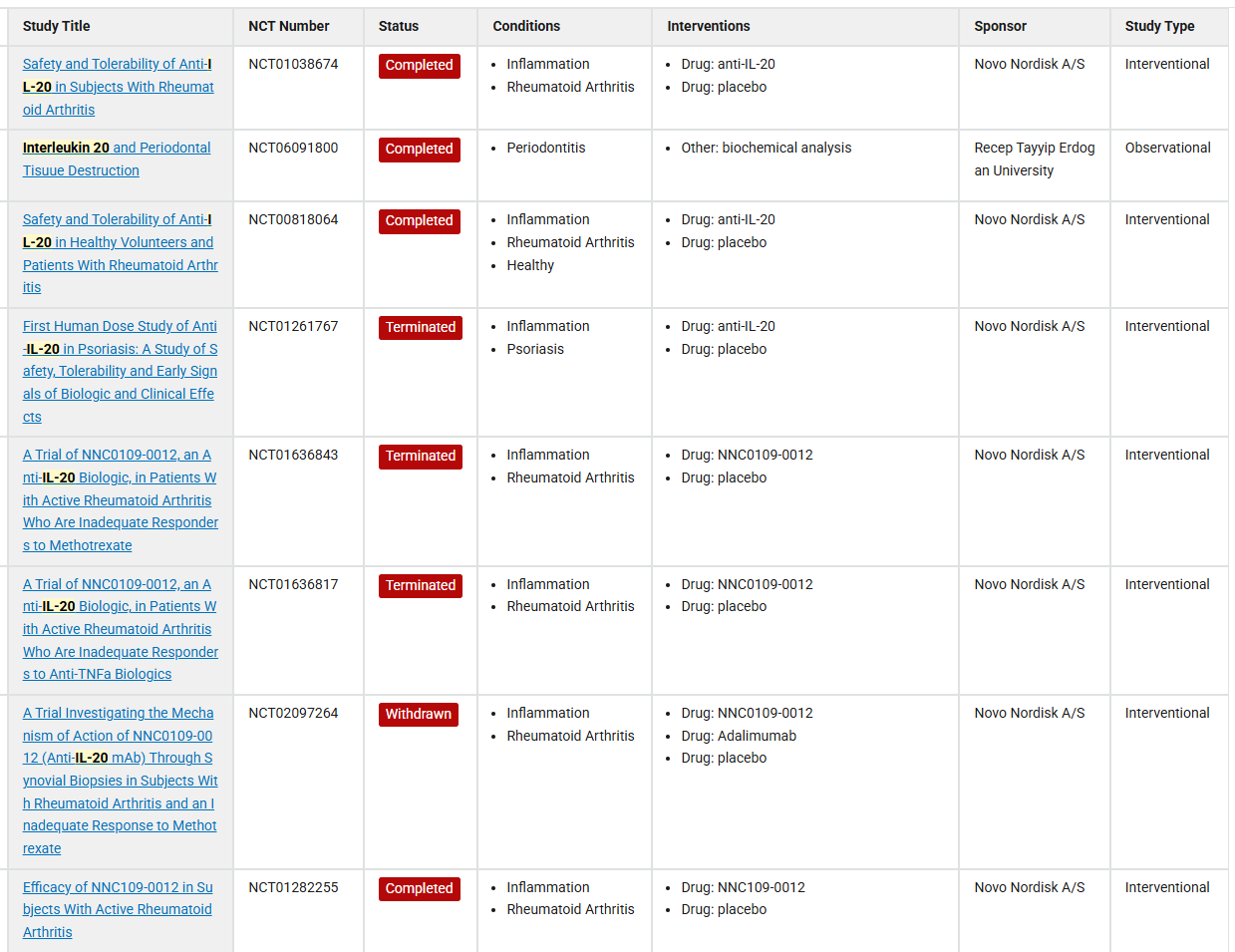
(Data source: clinicaltrials)
