Integrin α-IIb, also known as ITGA2B, ITGAB, GP2B, and CD41; and integrin β-3, also known as ITGB3, GP3A, and CD61; ITGB3 and ITGAV bind to form integrin α-V/β-3, which are receptors for cytoskeletal proteins, fibronectin, laminin, matrix metalloproteinase-2, osteopontin, osteomodulin, prothrombin, thromboretin, and von Willebrand factor. ITGB3 binds to ITGA2B to form integrin α-IIb/β-3; integrin α-IIb/β-3 (ITGA2B: ITGB3) is a receptor for fibronectin, prothrombin, fibrinogen, plasminogen, and thromboretin. It recognizes the RGD sequence among various ligands. It recognizes the HHLGGGAKQAGDV sequence in the fibrinogen γ chain. Upon activation, integrin α-IIb/β-3 induces platelet/platelet interactions by binding to soluble fibrinogen. Integrin α-IIbβ3 is also a receptor for the erythrocyte-specific ICAM4 ligand, which is involved in heterologous cell-cell adhesion between erythrocytes and activated platelets.
Integrin αIIbβ3 is primarily expressed in platelets and megakaryocytes, and its bidirectional signaling is crucial for platelet function, hemostasis, and arterial thrombosis. The bidirectional signaling of integrin αIIbβ3 also plays an important role in cancer progression by regulating the interaction between integrin αIIbβ3 and the fibrinogen/αvβ3 complex on the tumor cell surface and/or the release of vascular endothelial growth factor ( VEGF ) from activated platelets.

(Data source: Peng L, et al. Front Immunol. 2026)
Structure of integrin αIIbβ3
Integrin αIIbβ3 is a heterodimeric calcium-dependent glycoprotein receptor. ITGA2B (α subunit) and ITGB3 (β subunit) are both transmembrane glycoproteins that form a complex through non-covalent interactions. These two subunits consist of three distinct structural regions: an extracellular domain (ECD), a transmembrane domain (TMD), and a short cytoplasmic tail (CT).
The α subunit consists of an α heavy chain and a β light chain linked by disulfide bonds. The extracellular region consists of four domains: a heptaloid β-propeller domain, a thigh domain, and two lower leg domains.
The β subunit is a cysteine-rich single-chain protein. Its extracellular region contains seven complex domains: a βI-like/βA domain, a plexin-signalin-integrin (PSI) domain, four cysteine-rich epidermal growth factor (EGF)-like modules, and a β-tail domain (βTD). The βA domain, located at the foremost end, is the core of ligand binding. It contains a crucial metal-dependent adhesion site (MIDAS) that interacts directly with the RGD sequence on ligands such as fibrinogen via divalent cations (e.g., Ca²⁺, Mg²⁺).
αIIbβ3 has three different conformational states: a bent-closed (inactive) form, a stretched-closed (intermediate) form, and a stretched-open (active) form.

(Data source: Wu J, et al. J Cell Mol Med. 2026)
Bidirectional signal transduction of integrin αIIbβ3
Integrin αIIbβ3 in inside-out signal transduction
Vascular injury or agonists (such as ADP and thrombin) stimulate platelet surface receptors (such as GPCRs and GPVI), triggering signaling events such as increased intracellular calcium ions and activation of protein kinase C (PKC). Downstream effector molecule Rap1-GTP is activated, recruiting the adaptor protein RIAM and the key activating protein Talin to the cell membrane. Talin binds to the intracellular tail of the β3 subunit, disrupting the salt bridge connection between αIIb and the intracellular segment of β3; Kindlin-3 co-binds, jointly inducing receptor transmembrane dissociation and conformational changes in the extracellular domain.
The αIIbβ3 receptor transitions from a low-affinity state to a high-affinity state, enabling it to bind fibrinogen and vWF with high affinity, thus initiating platelet aggregation.

Outward-to-inward signal transduction of integrin αIIbβ3
Following ligand binding to the extracellular domain of integrin αIIbβ3, integrin αIIbβ3 aggregation promotes Src activation via autophosphorylation. Caloplasmin cleaves the cytoplasmic tail of integrin β3, causing partially activated Src to dissociate from the integrin β3 tail. Src phosphorylation supports the activation of various enzymes and signaling proteins, such as FAK, Syk kinase, RhoGAP, Rac-GEFs, RhoGEFs, and PI3K. Gα13, talin, kindlin, tensin, and vinculin provide the necessary link between the cytoplasmic tail of integrin β3 and actin. Kindlin can directly couple integrin β3 to the actin cytoskeleton via the ILK/PINCH/parvin complex.

(Data source: Huang J, et al. J Hematol Oncol. 2019)
Signal transduction of integrin αIIbβ3 in tumors
Integrin αIIbβ3 binds to cell membrane receptors via extracellular ligands, extracellular matrix (ECM) components, or transforming growth factor-β1 (TGF-β1), activating FAK, Src, PI3K, KRAS, and other intracellular molecules. Through signaling pathways such as MAPK and AKT/mTOR, it ultimately regulates downstream molecules such as c-Myc and NF-κB, thereby promoting malignant phenotypes such as tumor invasion and metastasis, angiogenesis, immunosuppression, and cell proliferation.
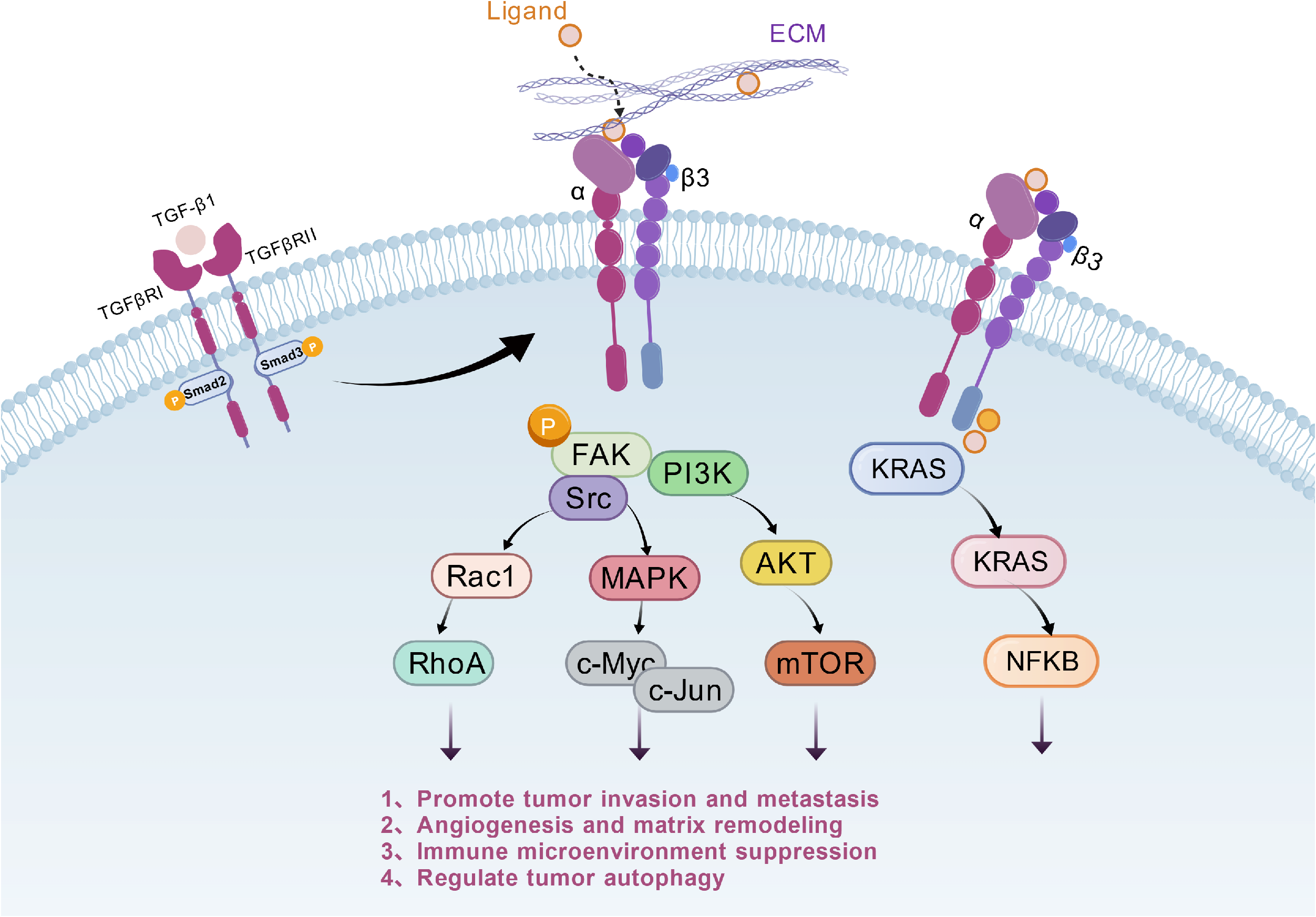
(Data source: Peng L, et al. Front Immunol. 2026)
Targeted therapy of integrin αIIbβ3
Abciximab is an antibody Fab fragment developed by Johnson & Johnson that targets αIIbβ3. The steric hindrance created by abciximab's binding to integrin αIIbβ3 prevents the interaction of fibrinogen and other ligands with integrin αIIbβ3, thus interfering with platelet aggregation and thrombus formation. Abciximab has nearly equal affinity for blocking integrin αIIbβ3 or αvβ3. This drug was approved by the US FDA in 1994.
