CD52 (Campath-1 antigen) is a 12-amino acid glycoprotein anchored to glycosylphosphatidylinositol. Its soluble form, sCD52, regulates T cell activation and macrophage phagocytosis by binding to the Siglec-10/G receptor and transmitting inhibitory signals.
CD52 expression distribution
CD52 expression is tissue-specific. It is mainly expressed in the epididymis of male tissues, primarily in reproductive support cells. It is also expressed on the cell surface of immune cells, and is also expressed in small amounts in mature lymphocytes, natural killer (NK) cells, eosinophils, neutrophils, monocytes/macrophages, and dendritic cells (DCs).
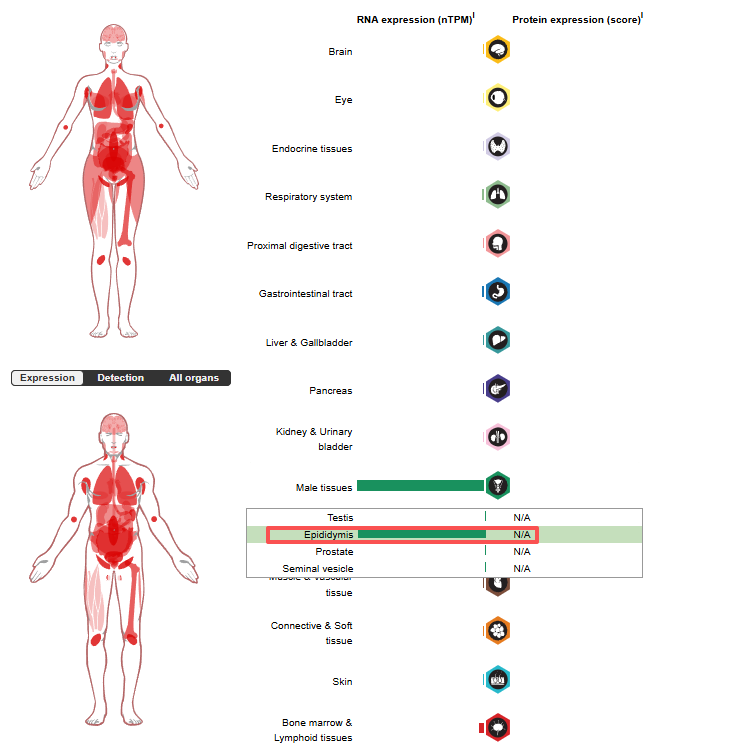
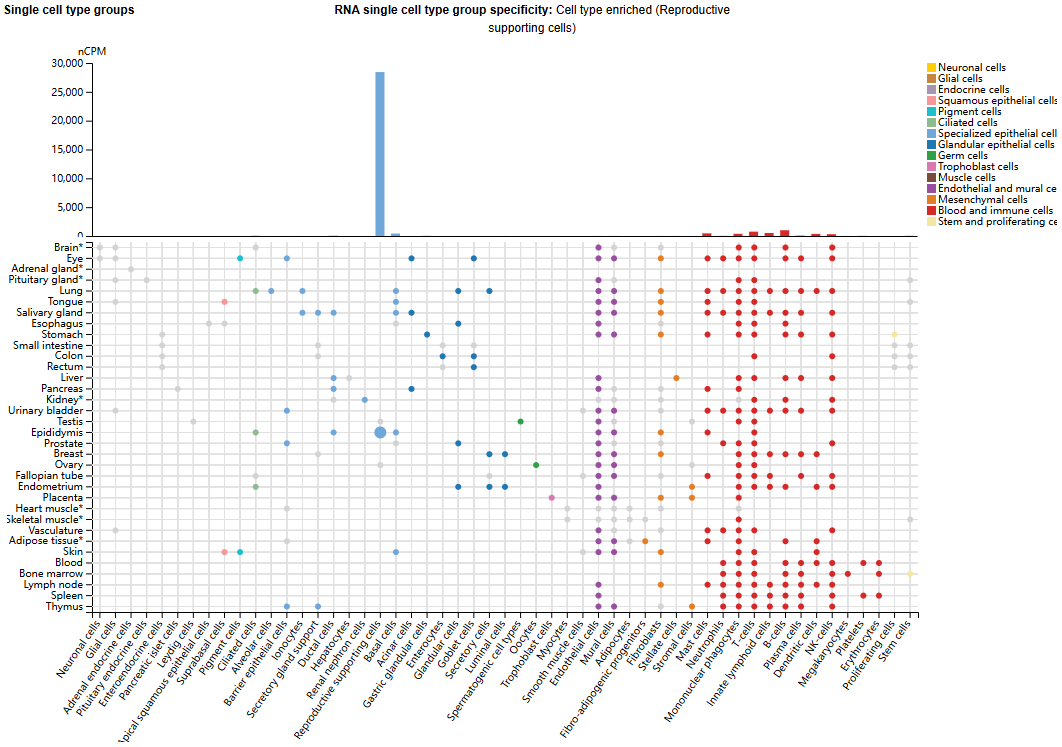
(Data source: uniprot)
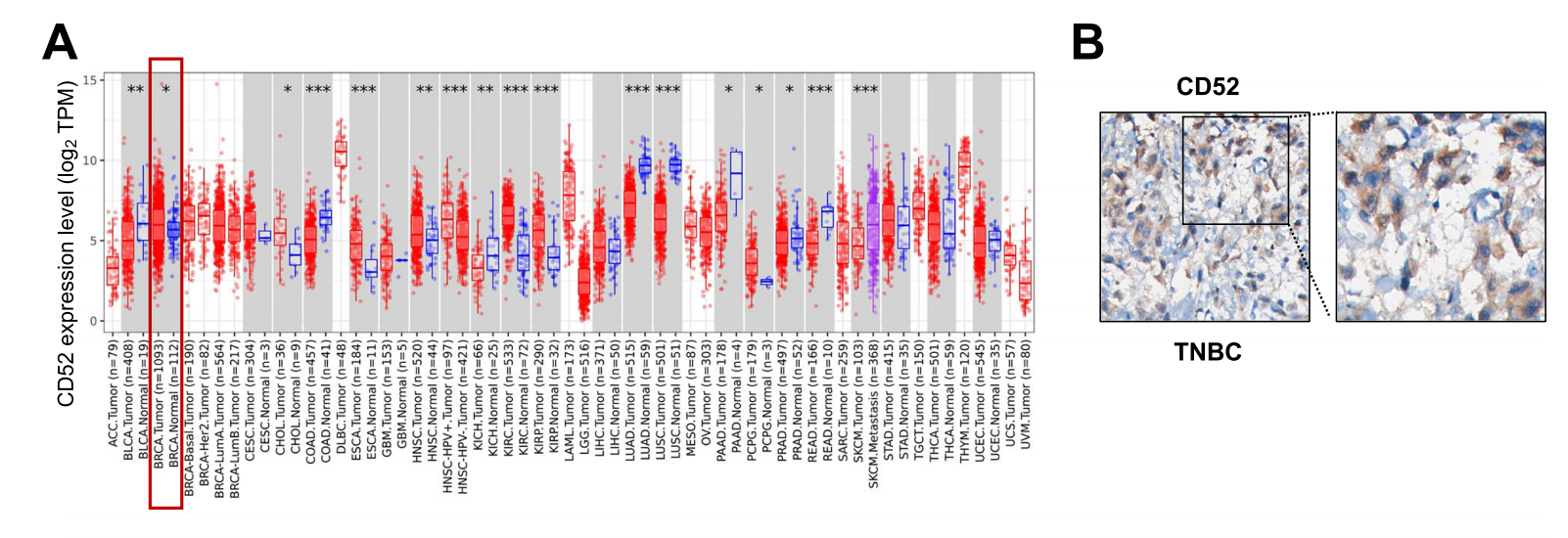
(Data source: Qin X, et al. NPJ Breast Cancer. 2026)
CD52 Structure
CD52 is a lipid-anchored glycosylated protein composed of 61 amino acids, located on human chromosome 1p36 and encoded by two exons . The mature protein has an extremely short peptide chain of only 12 amino acids , and is anchored to the cell membrane via a GPI anchor, rather than a transmembrane domain. Because the GPI anchor is easily cleaved by phospholipases, cell-bound CD52 is released as soluble CD52 (sCD52) via phospholipase C.
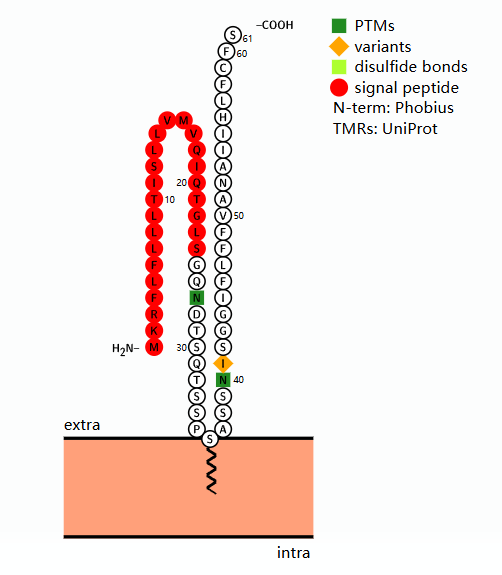
(Data source: protecter)
The role of CD52 in immune regulation
T cell suppression: CD52 is released from its GPI anchor via phospholipase. sCD52 binds to Siglec-10 on the surface of effector T cells via its sialylated sugar chain, leading to phosphorylation of the intracellular ITIM motif of Siglec-10. This recruits phosphatases SHP-1/SHP-2, inhibiting the phosphorylation of downstream signaling molecules Lck and ZAP-70 of the T cell receptor (TCR), thereby negatively regulating T cell activation.
Promoting Immune Evasion: CD52 transmits inhibitory signals by binding to the Siglec-G (mouse)/Siglec-10 (human) receptors on the surface of macrophages; the intracellular domain of Siglec-G contains the ITIM motif, recruiting phosphatases SHP-1/SHP-2 to inhibit macrophage phagocytosis. Cancer cells can utilize CD52 as a "don't eat me" signal to evade macrophage clearance, similar to the CD47-SIRPαpathway.
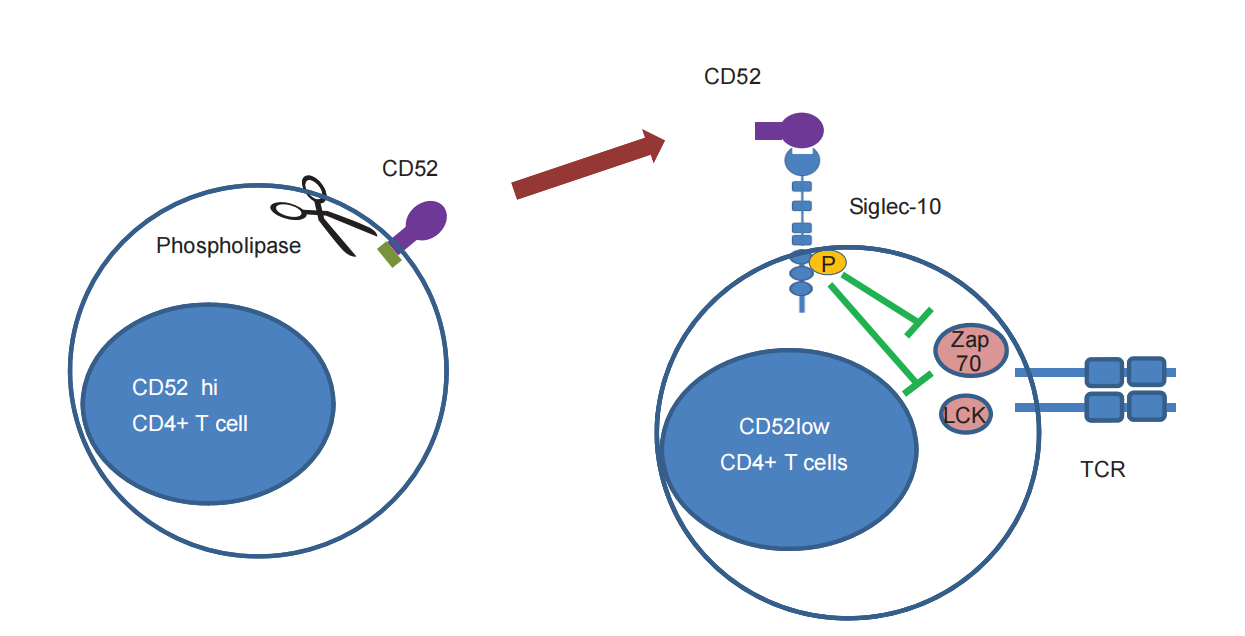
(Data source: Toh BH, et al. Cell Mol Immunol. 2013)
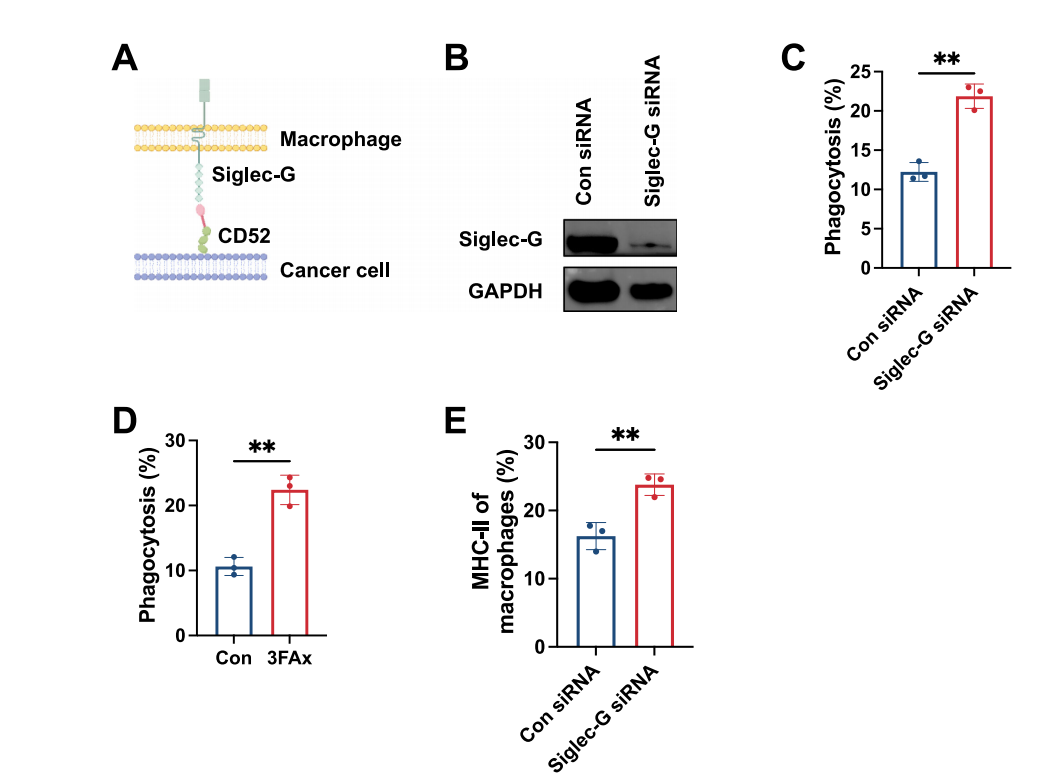
(Data source: Qin X, et al. NPJ Breast Cancer. 2026)
CD52-targeted therapy
A lemtuzumab is a monoclonal antibody targeting CD52, jointly developed by Bayer and Sanofi. It was approved by the FDA in 2001 for relapse-remission multiple sclerosis, chronic lymphocytic leukemia, multiple sclerosis, and CD22-positive B-cell acute lymphoblastic leukemia.
ALLO-647 is a proprietary anti-CD52 monoclonal antibody developed by Allogene Therapeutics. In August 2025, Allogene Therapeutics decided to terminate development of ALLO-647. Previously, a participant died in the company's Phase II ALPHA3 study. The investigation confirmed that the event was caused by ALLO-647 in the lymph node disseminated intravenous fluid (CLDF) regimen, not cema-cel itself. The participant developed disseminated adenovirus infection due to immunosuppression on day 54 after infusion, ultimately leading to liver failure and death. Allogene has completely halted ALLO-647-related studies and, after consultation with the FDA, has reverted to the standard cyclophosphamide + fludarabine (FC) regimen.
