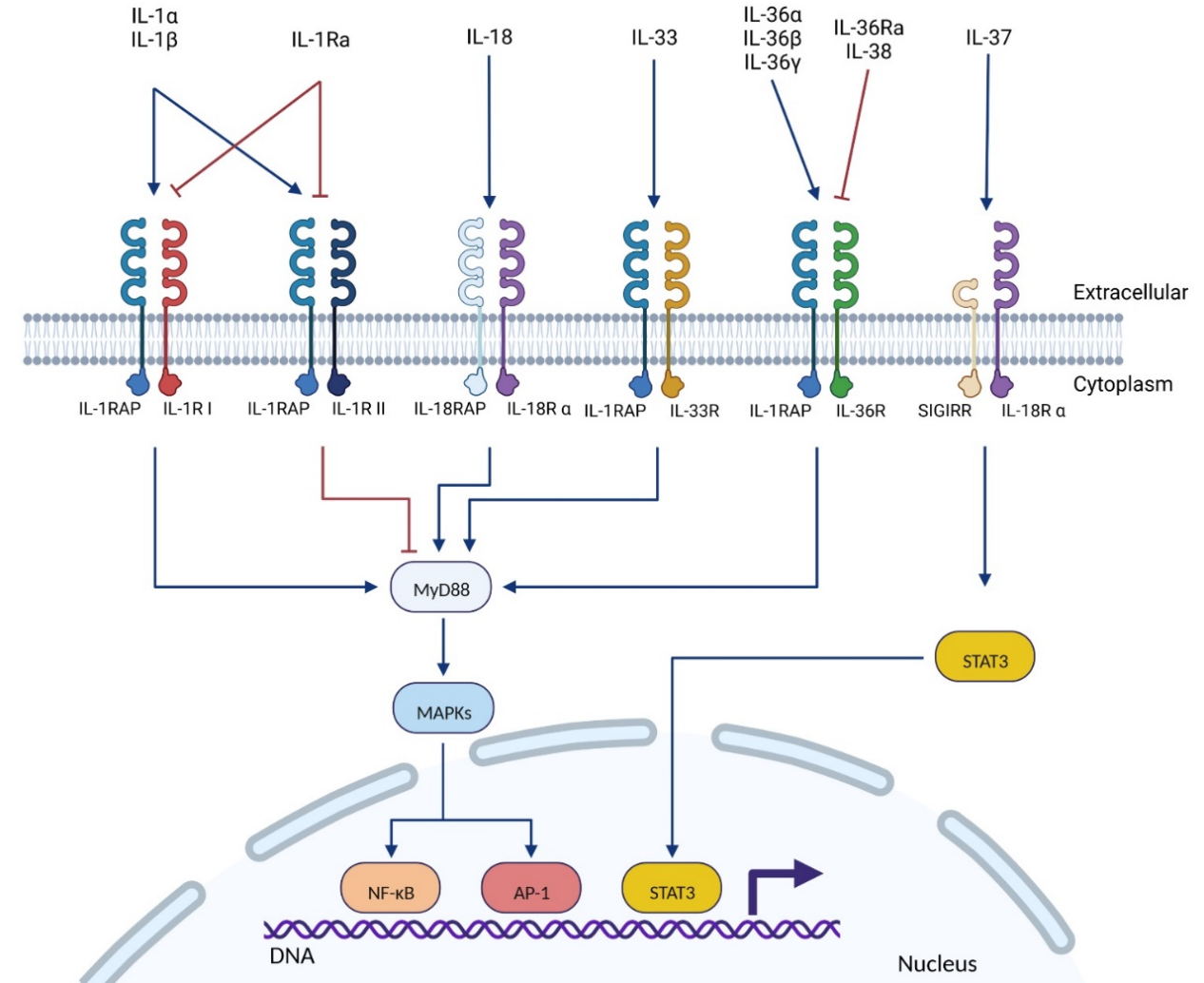
Interleukin-1 receptor accessory protein (IL-1RAP), also known as IL-1R3, is a member of the IL-1 receptor family. In normal tissues, IL-1RAP is mainly expressed in the liver, specifically in neutrophils, hepatocytes, and trophoblast cells. IL-1RAP is involved in three signaling pathways of the interleukin-1 family: interleukin-1 receptor (IL-1R), interleukin-33 receptor (IL-33R), and interleukin-36 receptor (IL-36R). These pathways exhibit pro-tumor activity. IL-1RAP upregulation has been found in several cancers, including hematologic malignancies (myeloid and lymphoid leukemias) and solid tumors (pancreatic, colorectal, or breast tumors).
(Data source: Frenay J, et al. Int J Mol Sci. 2022)
Structure of IL1RAP
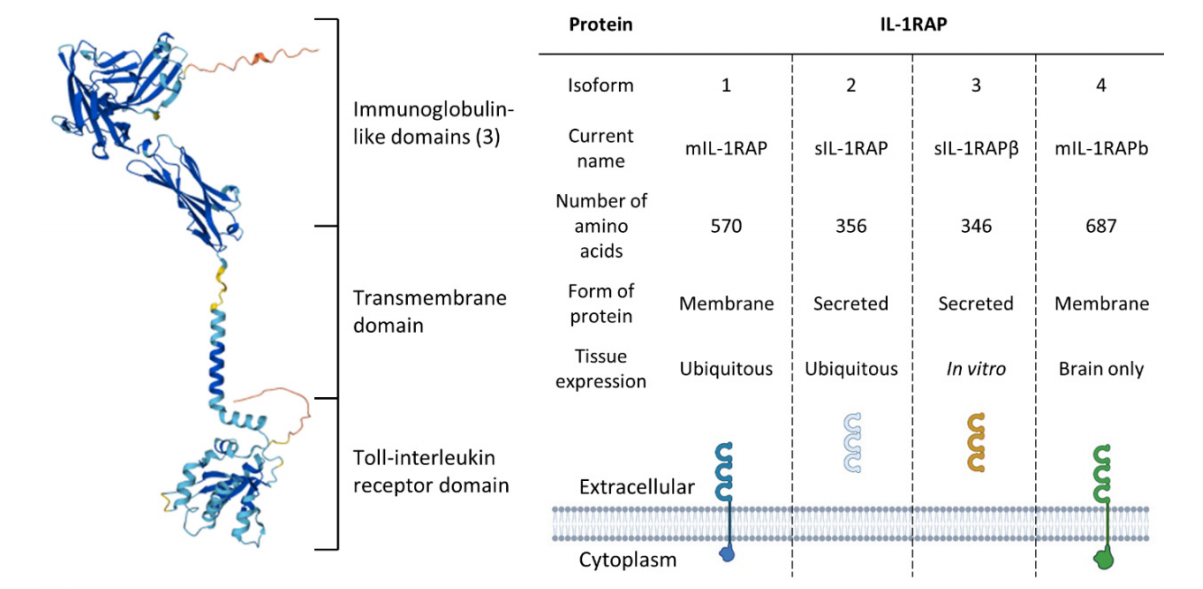
The IL-1RAP gene, encoded on chromosome 3q28, is a single-channel type I membrane protein composed of 570 amino acids. The IL-1RAP molecule consists of a triple immunoglobulin (Ig) sulfhydryl terminus (D1, D2, and D3) that recognizes β-cloverleaf cytokines, and a TIR C-terminal endogenous domain that activates the NF-κB and MAPK pathways. IL-1RAP acts as a co-receptor for IL-1RI, IL-1RII, IL-33R, and IL-36R. The TIR domain of IL-1RAP contains two key regions, both essential for IL-1 signaling. IL-1RAP exists in four splice variants: two soluble forms and two membrane forms.
(Data source: Frenay J, et al. Int J Mol Sci. 2022)
IL1RAP signaling pathway and regulation
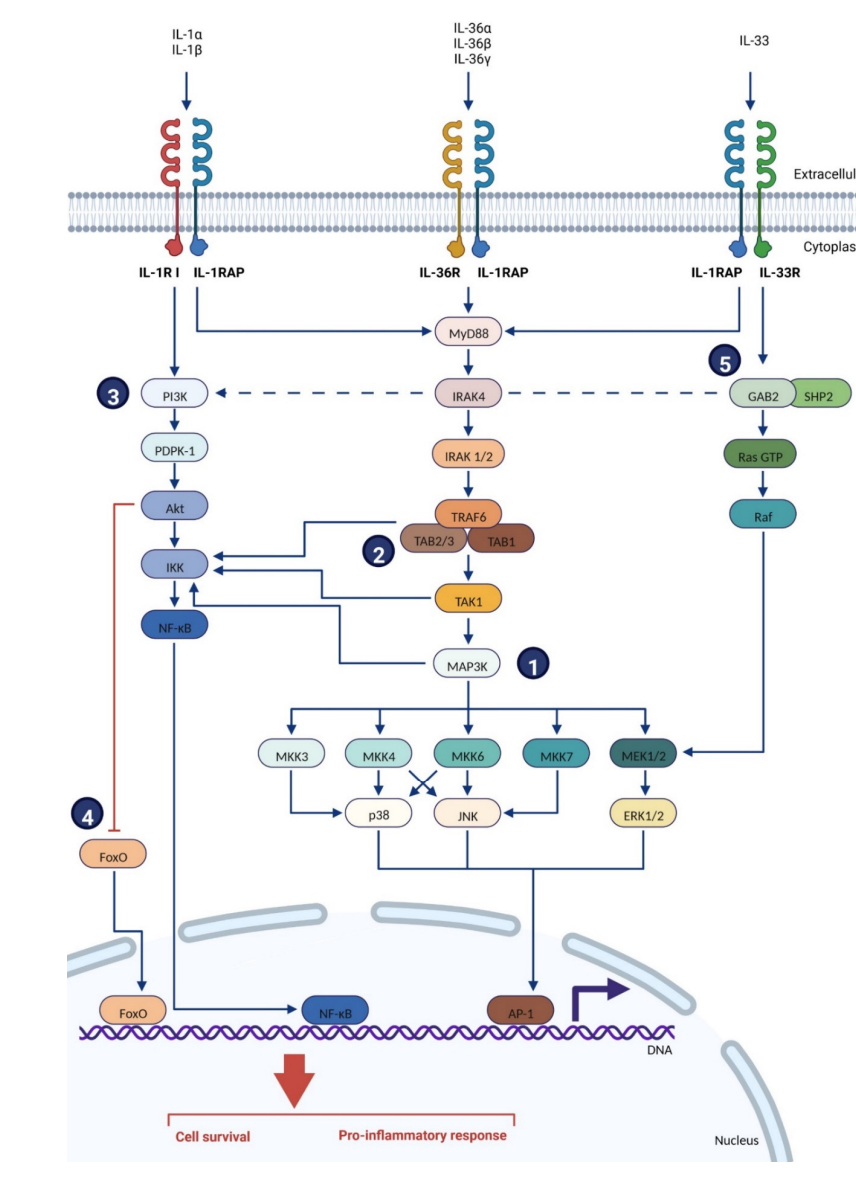
IL-1RAP is involved in three signaling pathways of the interleukin-1 family: IL-1R, IL-33R , and IL-36R. Cytokines bind to their major receptors, inducing the recruitment of IL-1RAP. The intracellular TIR (Toll/interleukin-1 receptor) domains of the major receptor and IL-1RAP are juxtaposed, which is essential for the recruitment and binding of several intracellular proteins and kinases, such as Toll-interacting protein (Tollip), myeloid differentiation factor 88 (MyD88), members of the IL-1R-associated kinase (IRAK) family, and TNF receptor-associated factor 6 (TRAF-6). These proteins subsequently trigger an intracellular signaling cascade, inducing the expression of NF-κB and AP-1-dependent pro-inflammatory cytokines, chemokines, and secondary mediators of the inflammatory response.
(Data source: Frenay J, et al. Int J Mol Sci. 2022)
The role of IL-1RAP in tumors
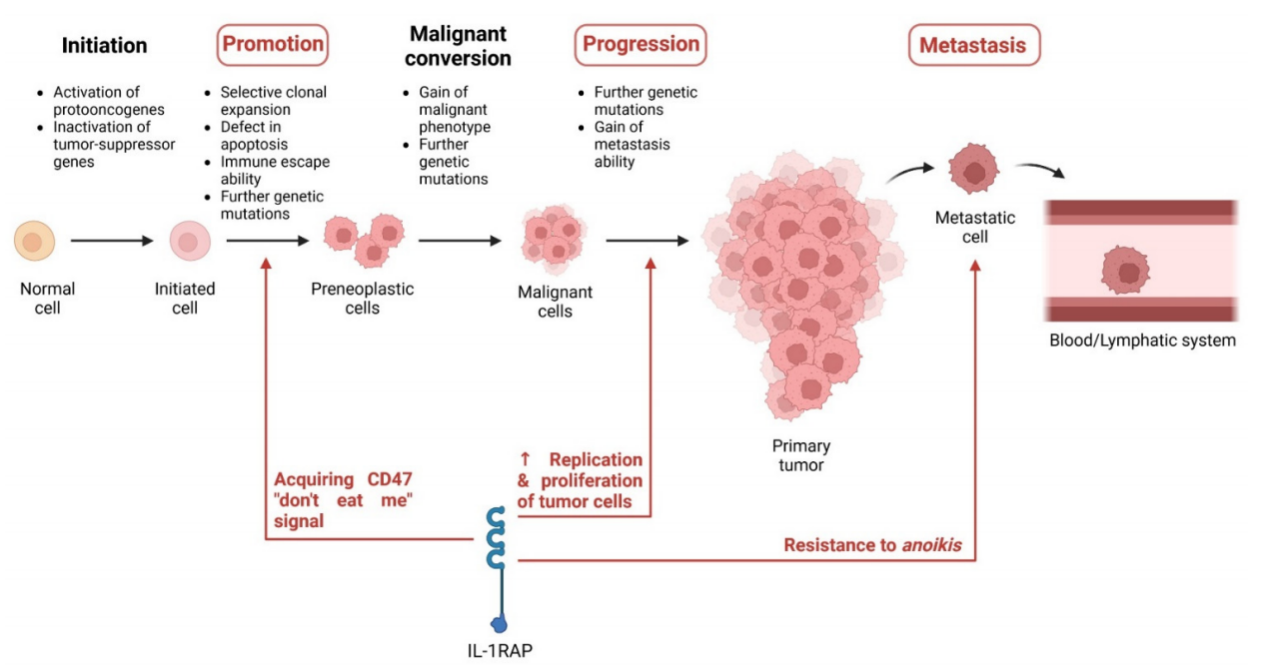
IL-1RAP plays a role in pro-inflammatory responses through the NF-κB and AP-1 pathways. IL-1RAP is highly expressed in hematologic malignancies and some solid tumors. IL-1RAP plays different roles in different tumors, including immune escape ability, tumor progression, and anoikis resistance during metastasis.
IL-1RAP plays an important role in the progression of cervical cancer. This IL-1RAP-mediated NF-κB activation induces the production of CD47 on the surface of tumor cells, thereby gaining the ability to escape the immune system by expressing CD47 (the “don’t eat me” signal) on the surface of tumor cells.
IL-1RAP is upregulated by protein tyrosine phosphatase receptor-δ (PTPRD) in glioma cells. At this time, the S and G2 cell cycle phases in glioma cells are increased, which suggests that PTPRD may promote the replication of glioma cells and tumor proliferation through the IL-1RAP pathway.
IL-1RAP has been identified as a factor that inhibits anoikis in Ewing sarcoma. In Ewing sarcoma, IL-1RAP maintains glutathione (GSH) levels through two pathways: binding to the xCT/CD98 transporter to enhance cystine uptake and activating CTH enzymes to promote de novo cystine synthesis.
(Data source: Frenay J, et al. Int J Mol Sci. 2022)
Targeted therapy for IL1RAP
Because IL-1RAP is highly expressed in tumor cells, blocking IL-1RAP is more effective than blocking major receptors (IL-1R, ST2, IL-1Rrp2) in inhibiting the production of pro-inflammatory cytokines (such as IFN-γ and IL-6). This suggests that IL-1RAP may be an effective therapeutic target for diseases affected by multiple members of the IL-1 family.
Cantargia is a leading company in developing therapeutics targeting IL1RAP and has established a knowledge and technology platform in this field. Cantargia has developed monoclonal and bispecific antibodies targeting IL1RAP, both currently in preclinical and clinical trials.
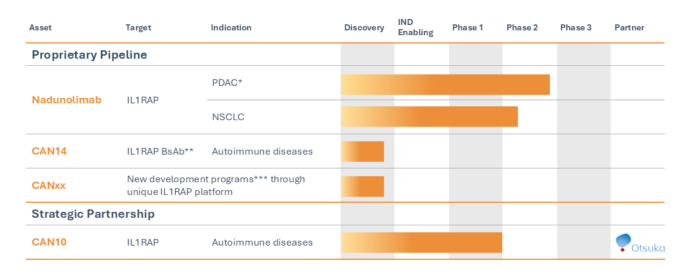
Nadunolimab (CAN04) destroys tumor cells through the ADCC effect by stimulating the immune system's natural killer cells. CAN04 also blocks the signaling of two forms of interleukin-1 (α and β), which promote tumor development and lead to resistance to chemotherapy.
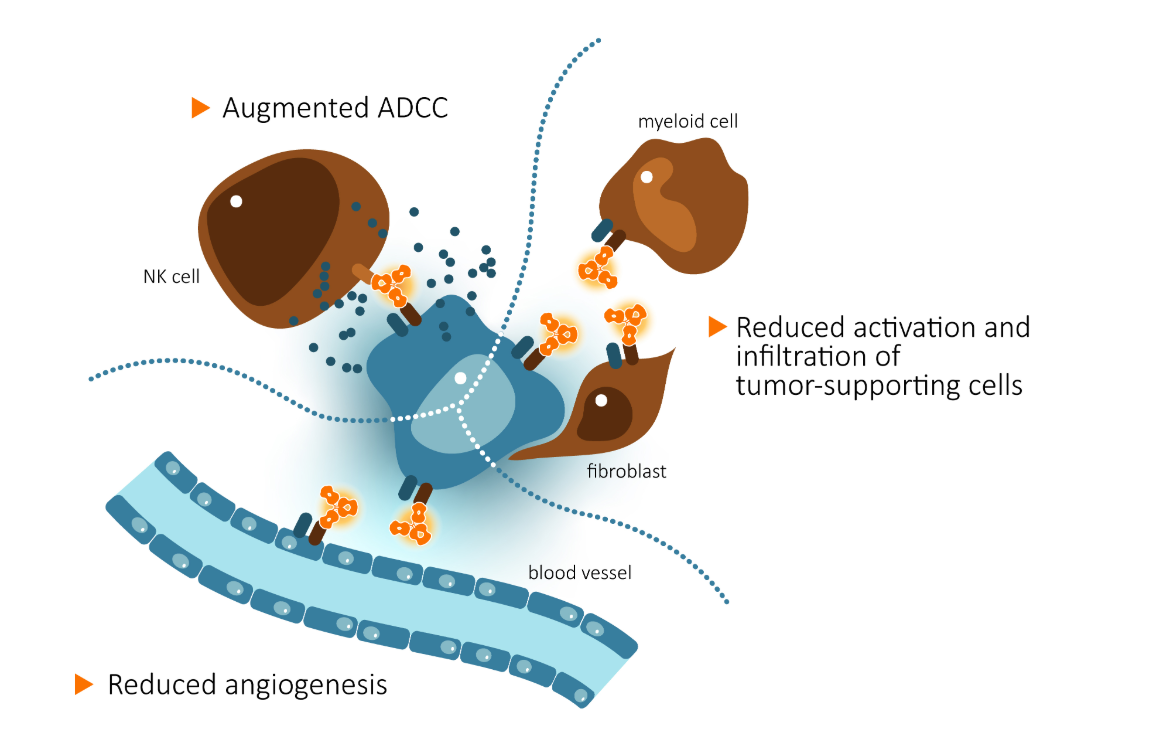
CAN04 is currently in Phase 1/2 clinical trials. CANFOUR, a Phase 1/2a trial, is CAN04's first clinical study. The Phase 2a portion of CANFOUR focuses on its use in combination with standard-of-care chemotherapy. CAN04 has been used in combination with gemcitabine and albumin-bound paclitaxel, as well as in combination with various platinum-based doublets, in pancreatic cancer (PDAC) and non-small cell lung cancer (NSCLC). Phase 1 results from CANFOUR showed that CAN04 has a high safety profile, and interim data from Phase 2a showed a synergistic effect between CAN04 and chemotherapy.
TRIFOUR is a phase Ib/II trial evaluating the efficacy of CAN04 in combination with carboplatin and gemcitabine for the treatment of triple-negative breast cancer (TNBC). TRIFOUR was conducted in Spain in collaboration with the Spanish Breast Cancer Research Group (GEICAM) and the Spanish Breast Cancer Collaborative Group.
The other two clinical trials, CAPAFOUR and CESTAFOUR, focus on the combined use of CAN04 and chemotherapy.
(Data source: Cantargia official website)
CAN10, also developed by anti-cantargia, is an anti-IL1RAP antibody for the treatment of autoimmune and inflammatory diseases. CAN10 possesses the unique ability to block not only IL-1 signaling but also IL-33 and IL-36 signaling. Simultaneous blocking of these three cytokines holds great potential for treating a variety of often heterogeneous autoimmune and inflammatory diseases. CAN10 has also been developed to minimize binding to effector cells. This means that, unlike nadunolimab (CAN04), CAN10 does not promote cell killing via ADCC. CAN10's first Phase 1 clinical trial (NCT06143371) is nearing completion. Treatment of healthy volunteers has been completed, while the trial in participants with psoriasis is ongoing. Results are being continuously reported to the market, and no safety issues have been observed to date at different dose levels.
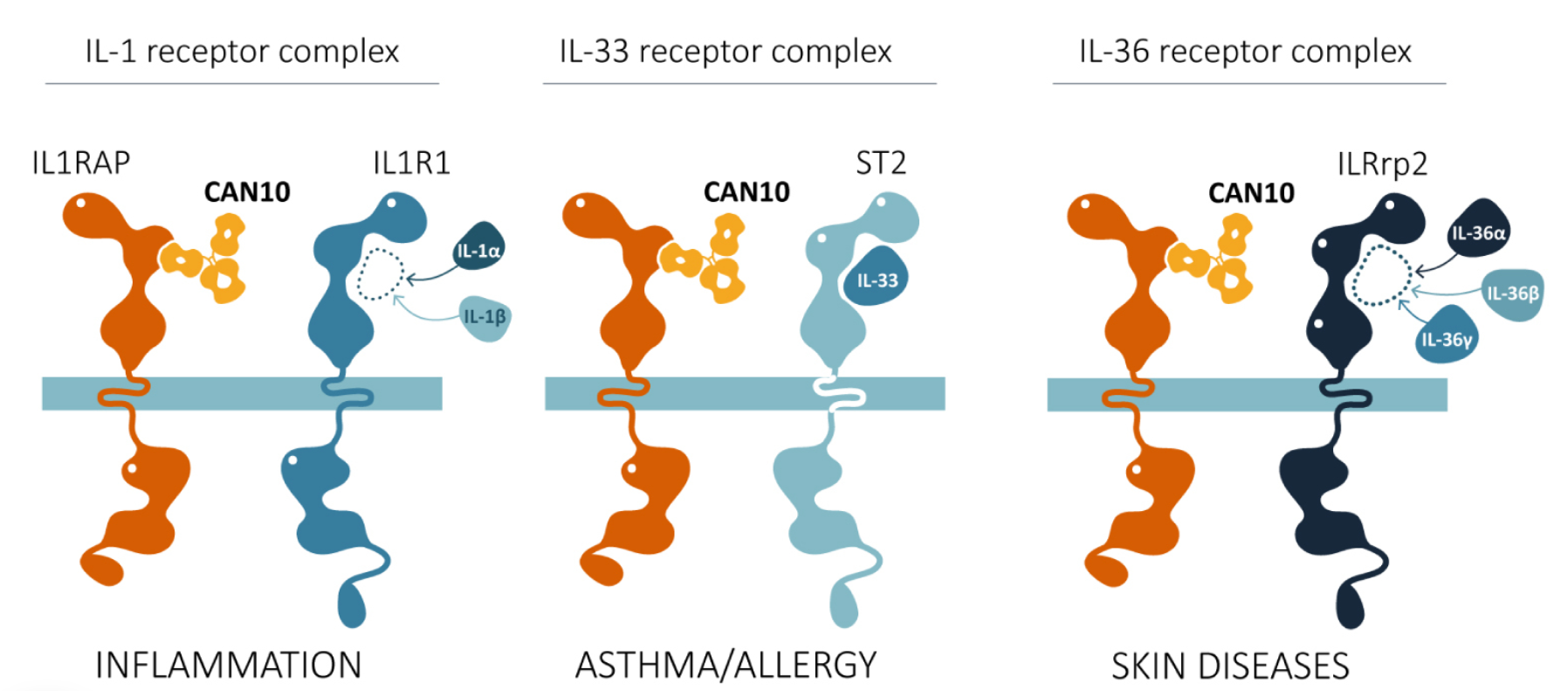
(Data source: Cantargia official website)
