Integrin alpha ITGA4 (CD49D), a key subunit of the integrin family, plays a role in the transport, adhesion, and migration of immune cells by forming functional heterodimers α4β1 (VLA-4) and α4β7. On activated endothelial cells, integrin VLA-4 triggers isomorphic aggregation of most VLA-4-positive leukocyte lineages. It may also be involved in interactions with cytolytic T cells of target cells. ITGA4 plays a central role in the pathogenesis of various autoimmune diseases, such as multiple sclerosis and inflammatory bowel disease, thus becoming an important target for therapeutic intervention.
ITGA4 expression distribution
ITGA4 is mainly expressed in megakaryocytes, erythrocytes, T cells, stem cells, and NK cells. In T cells, CD49D is primarily expressed in CD4+ and CD8+ T cells; compared to healthy controls, CLL patients show significantly increased expression of CD49D in both CD4+ and CD8+ T cells.
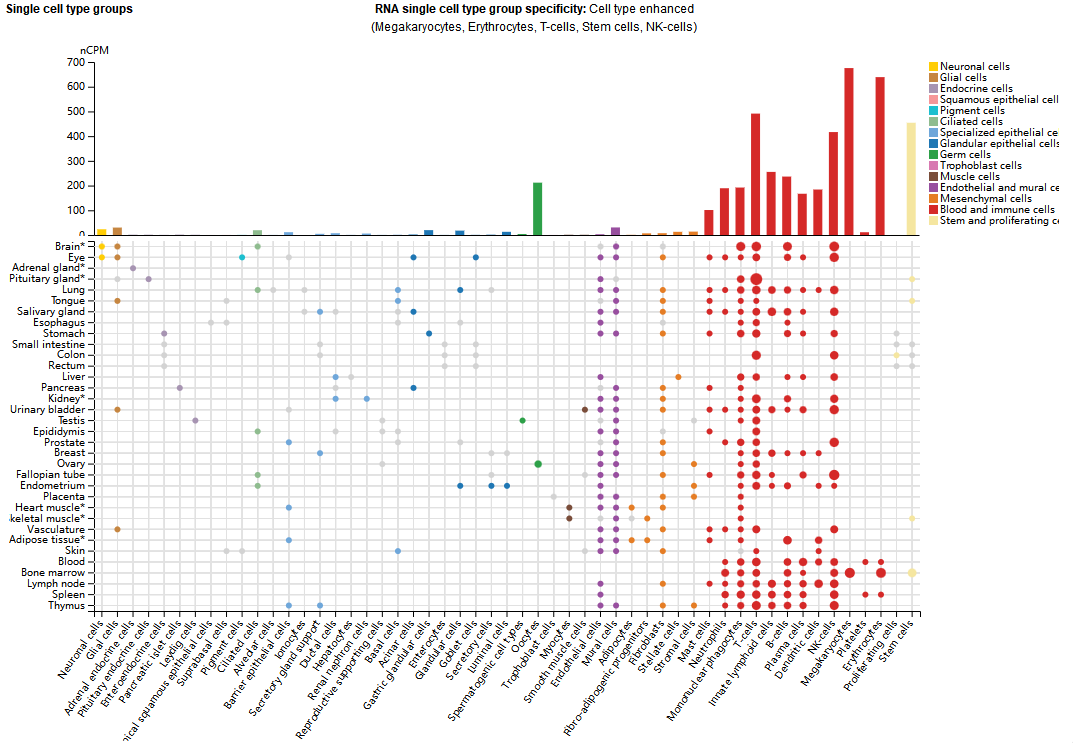

(Data source: uniprot)
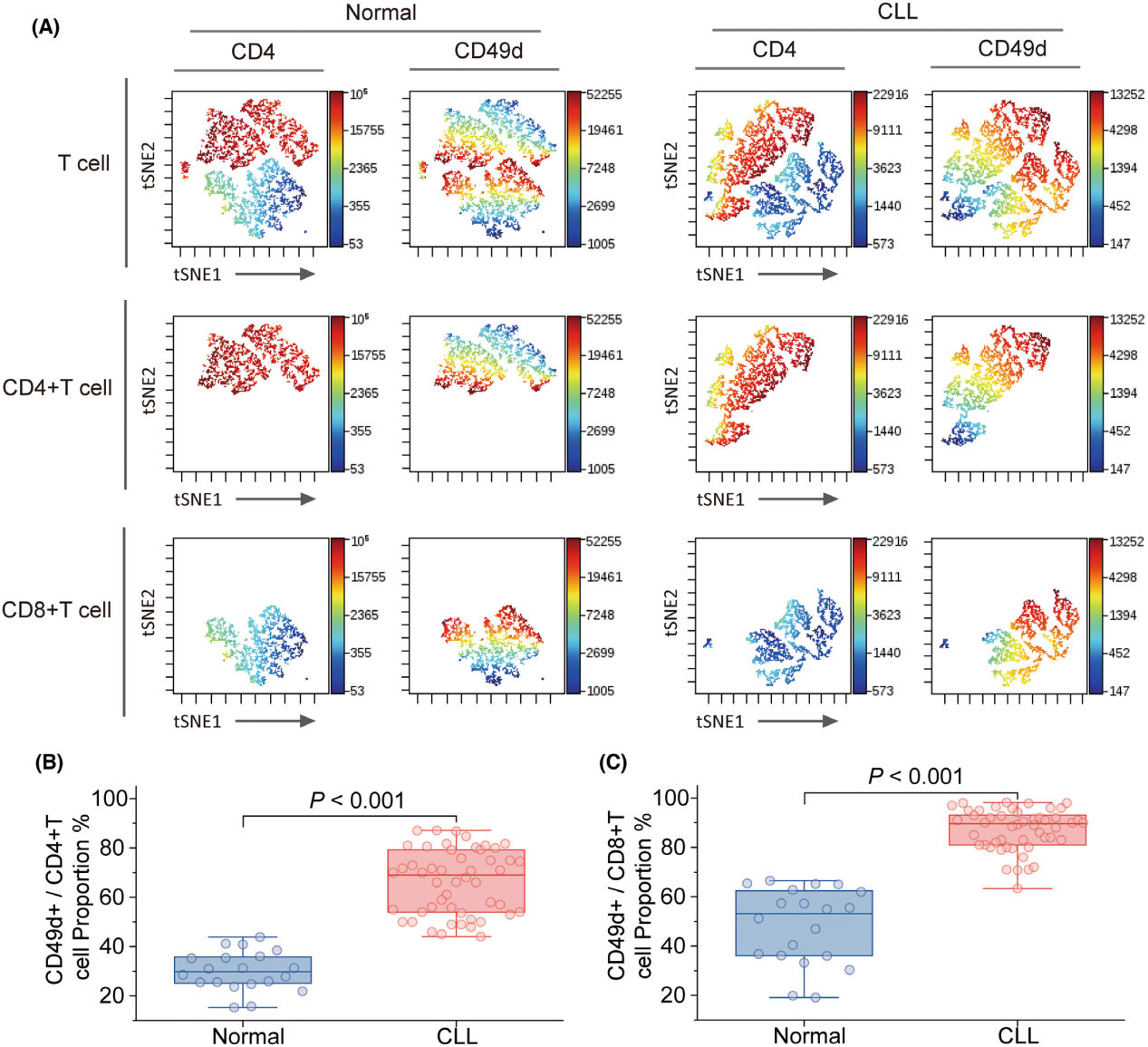
(Data source: Wang M, et al. Br J Haematol. 2025)
ITGA4 Structure
ITGA4 is a single-transmembrane type I membrane protein composed of 1032 amino acids, mainly consisting of a signal peptide, an extracellular domain, a transmembrane region, and an intracellular domain.
Precursor processing: ITGA4 initially synthesizes a precursor protein, which is then hydrolyzed by proteases to form heavy and light chains linked by disulfide bonds, eventually maturing into functional subunits.
Extracellular domain: The β-helical domain contains multiple FG-GAP repeat sequences that fold into a heptal β-propeller structure. This is the main region for ligand binding, responsible for recognizing and binding extracellular matrix molecules (such as the CS-1 fragment of fibronectin) or adhesion molecules on endothelial cells (such as VCAM-1 and MAdCAM-1).
Transmembrane domain: The single transmembrane α-helix not only acts as an anchoring agent but also participates in the interactions between subunits.
Cytoplasmic tail region: Relatively short, but contains key conserved domains. These domains contain binding sites for intracellular adaptor proteins such as Paxillin, Kindlin-3, and Talin. Phosphorylation modifications in this region (e.g., Ser1027) can finely regulate protein binding, thereby modulating signal transduction.
ITGA4 forms a heterodimer with the β1 subunit (CD29) through non-covalent bonding, and is therefore also known as α4β1 integrin (VLA-4).
The heterodimer α4β7 is formed by the binding of ITGA4 to the β7 subunit. Integrins VLA-4 and α4β7 are receptors for fibronectin; they recognize one or more domains within the CS-1 and CS-5 regions of fibronectin alternative splicing. They are also receptors for VCAM1; integrin α-4/β-1 recognizes the QIDS sequence in VCAM1. Integrin α-4/β-7 is also a receptor for MADCAM1, recognizing the LDT sequence in MADCAM1.
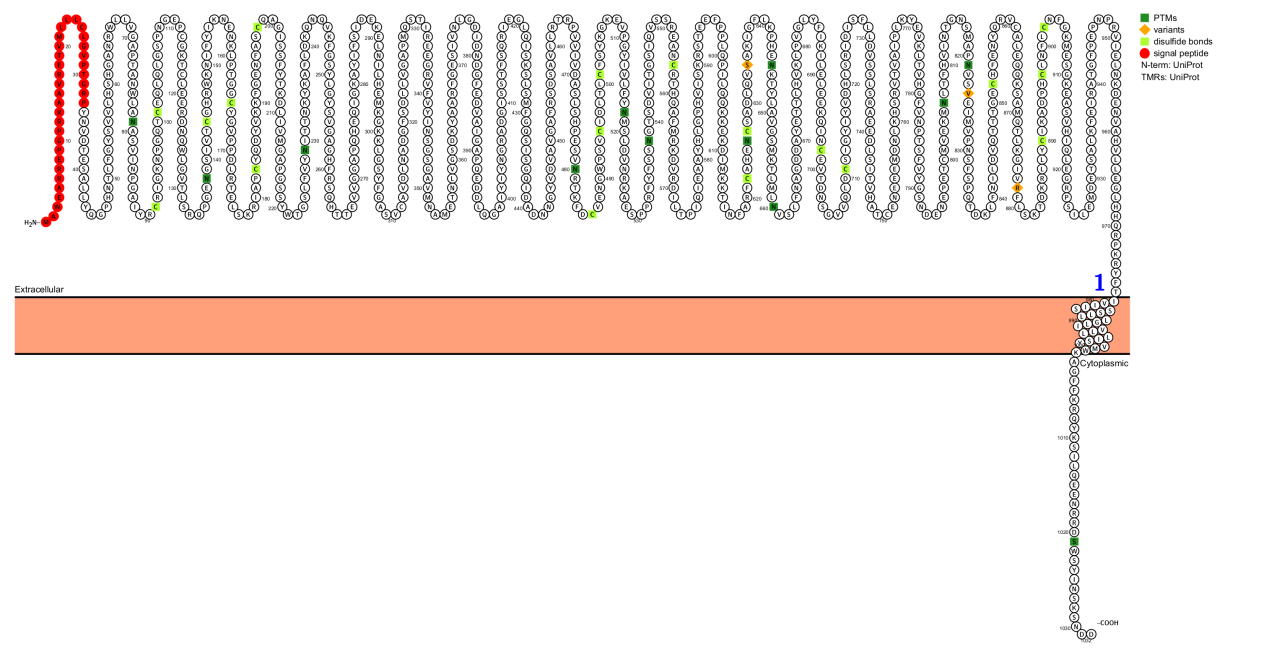
(Data source: protecter)
ITGA4 signal path and regulation
ITGA4 itself does not have enzymatic activity. Instead, it performs a unique bidirectional signal transduction function by forming heterodimers (usually paired with β1 or β7 subunits), linking the extracellular environment with the dynamics of the intracellular cytoskeleton.
Signal transmission“from the inside out”:
Chemokines or antigen stimulation activate the intracellular small GTPase Rap1 via G protein-coupled receptors or other immune receptors. Activated Rap1 recruits adaptor proteins such as Talin and Kindlin. Talin binds to the cytoplasmic tail of the ITGA4-βsubunit, causing the extracellular domain of the integrin to transition from a bent, low-affinity conformation to an extended, high-affinity conformation. This enables leukocytes to rapidly bind ligands expressed on the vascular endothelium (such as VCAM-1) in flowing blood, mediating leukocyte rolling, firm adhesion, and extravasation.
Signal transmission“from the inside out”:
Signal transduction occurs when integrin α4β1 interacts with VCAM-1. This binding activates focal adhesion kinase (FAK) and Src family kinases, forming a signaling complex that initiates multiple downstream cascade reactions. The PI3K/AKT pathway facilitates cytoskeleton remodeling and promotes cell migration; the MAPK/ERK pathway promotes cell survival by protecting leukocytes from apoptosis; and the Rho GTPase pathway regulates the formation of lamellar and filopodia, both of which support cell migration.

(Data source: De Matos MD, et al. Front Pharmacol. 2026)
Targeted therapy for ITGA4
Natalizumab is a monoclonal antibody targeting CD49D, approved for marketing in 2004 for diseases such as Crohn's disease, relapsing-remitting multiple sclerosis, and multiple sclerosis.
Abrilumab is a monoclonal antibody that blocks α4β7 integrin, developed by Amgen. In a phase 2b clinical trial in patients with moderate to severe ulcerative colitis (UC), the low-dose and high-dose groups achieved clinical responses of 13% and 12%, respectively, compared to 4% in the placebo group, meeting the trial's primary endpoint. Furthermore, these doses showed significantly higher rates of clinical response and mucosal healing compared to placebo.
Etrolizumab is an intestinal-selective monoclonal antibody that targets α4β7 and is currently undergoing phase III clinical trials worldwide for the treatment of inflammatory bowel disease (IBD).
Vedolizumab, developed by Takeda in Japan, is a monoclonal antibody approved by major global regulatory agencies, including the FDA, EMA, and NMPA, for the treatment of ulcerative colitis (UC) and Crohn's disease (CD). It preferentially binds to α4β7 integrin heterodimers expressed on T lymphocytes and blocks their interaction with mucosal vascular addressin cell adhesion molecule 1 (MAdCAM-1) on intestinal endothelial cells. By blocking α4β7 integrin, vedolizumab inhibits the migration of various immune cells to the intestine, particularly T lymphocytes, thereby reducing the excretion of pro-inflammatory cytokines and decreasing CD-related inflammatory responses. Vedolizumab has demonstrated efficacy, tolerability, and acceptable long-term safety in patients with Crohn's disease (CD). On February 19, 2026, Takeda announced positive data from the pivotal Phase 3 KEPLER trial, which showed that ENTYVIO® (vedolizumab) could provide clinical remission in patients aged 2 years and older with moderate to severe active ulcerative colitis (UC).

(Data source: Rubin DT, et al. Inflamm Bowel Dis. 2025)
