Platelet glycoprotein VI (GPVI) is a collagen receptor involved in collagen-induced platelet adhesion and activation, playing a crucial role in platelet procoagulant activity and subsequent thrombin and fibrin formation. This procoagulant function may contribute to arterial and venous thrombosis. This signaling pathway involves the FcRγ chain, Src kinase (possibly FYN or LYN) and SYK, the adaptor protein LAT, and leads to the activation of PLCG2. It is a therapeutic target for arterial thrombotic complications.
GP6 expression distribution
GP6 is mainly expressed in platelets, nephron cells, ductal cells, and neurons, and is expressed in small amounts in monocytes, NK cells, and T cells.
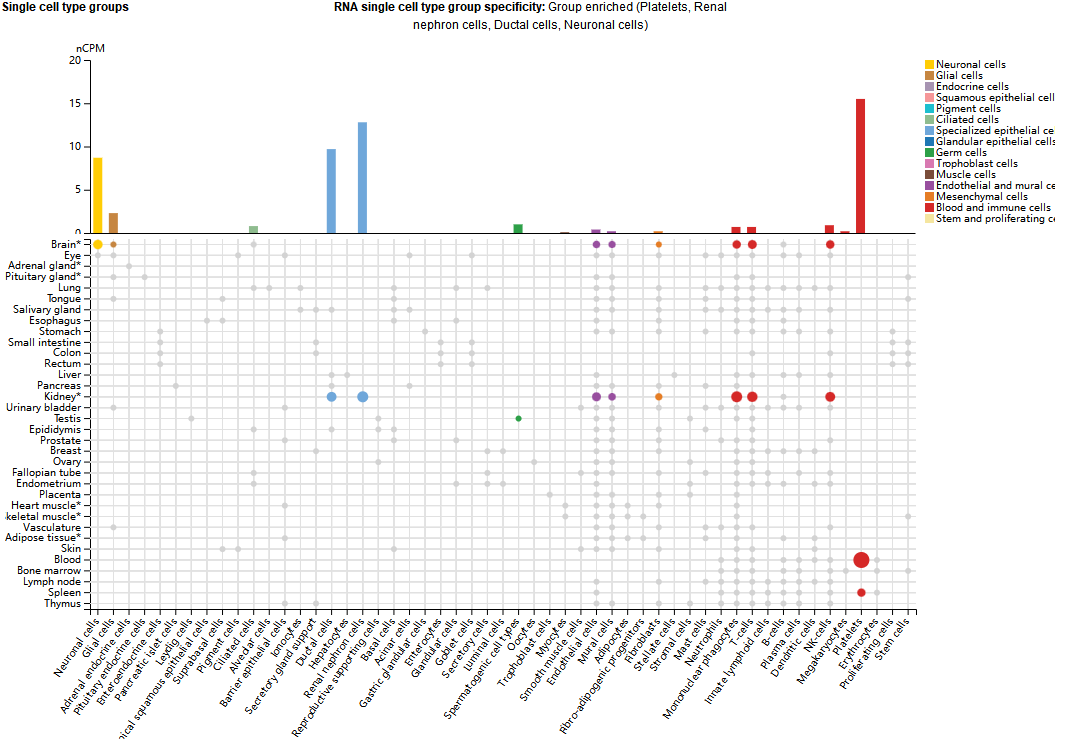
(Data source: uniprot)
GP6 structure and signal transduction regulation
GP6 is a type I transmembrane protein composed of 339 amino acids. It includes an extracellular Ig-like domain, D1 and D2 domains, a glycosylated stalk, a single transmembrane region, and a short intracellular tail. The cytoplasmic tail has a proline-rich region that binds to the Src homology 3 domain of a non-receptor tyrosine kinase. GPVI binds to collagen through its extracellular D1 domain. GPVI typically exists in monomeric form, while the D2 domain induces dimerization.
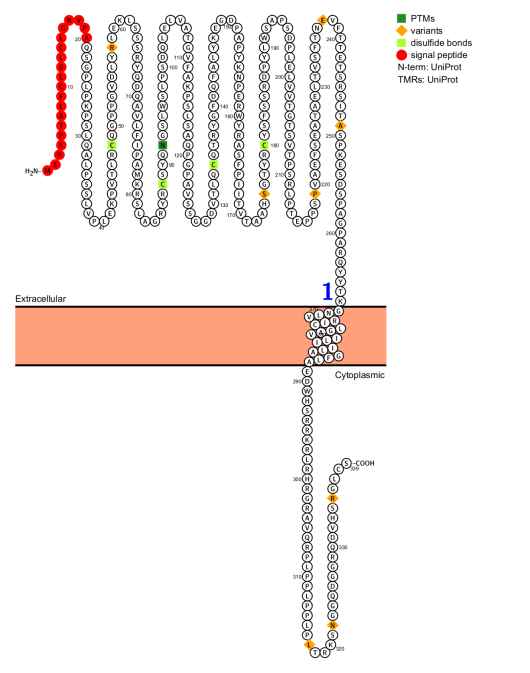
(Data source: protecter)
GPVI forms a complex with FcR-γ chains in the membrane via salt bridges formed across the transmembrane region and interactions within the intracellular tail. The FcR-γ chains are homodimers, each containing an immunoreceptor tyrosine activation motif (ITAM). Crosslinking of GPVI induces phosphorylation of conserved ITAM tyrosine residues by Src family kinases (sfk) Fyn, Lyn, and Src. Recruiting Syk to the phosphorylated ITAM initiates downstream signaling, leading to PLCγ2 activation and platelet aggregation.

(Data source: Li R, et al. Drug Discov Today. 2025)
The role of GP6 in thrombosis
GPVI activity is regulated through two mechanisms: dimerization and receptor aggregation, to effectively activate platelets. Platelets express multiple collagen receptors. Following vascular injury under blood flow conditions, the interactions of GPIbα-von Willebrand factor (VWF), integrin α2β1-collagen, and GPVI-collagen jointly mediate platelet adhesion. While GPIbα - VWF is not essential under high shear stress, integrin α2β1 can stabilize collagen binding and promote GPVI dimerization and aggregation, thereby amplifying GPVI activation and synergistically supporting platelet aggregation and thrombus formation. As a central collagen receptor, GPVI activates the FcRγ chain-dependent pathway, triggering calcium flow and integrin αIIbβ3 activation, promoting strong platelet adhesion and thrombus formation. Furthermore, under extremely high shear stress, the interaction between GPVI and fibrin significantly contributes to thrombus stability. GPVI plays a dual role in thrombus formation by promoting platelet aggregation and coagulation.
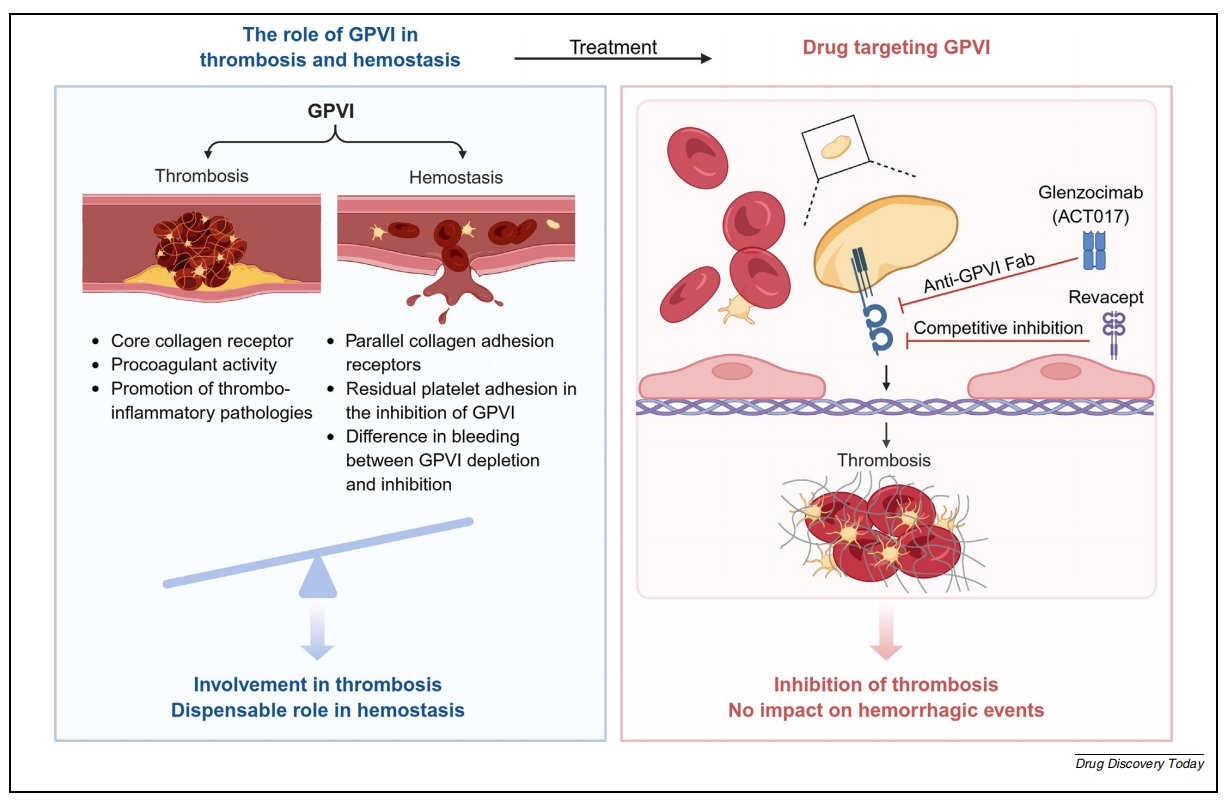
(Data source: Li R, et al. Drug Discov Today. 2025)
Targeted therapy for GP6
Targeted therapies for GP6 primarily include glenzocimab antibodies and Revacept fusion proteins. Glenzocimab (ACT017) is a humanized Fab fragment designed based on a mouse monoclonal antibody, mainly used to treat acute ischemic stroke with pulmonary embolism. In vitro studies using human platelets have demonstrated its effective inhibition of collagen-induced platelet aggregation. Glenzocimab binds to the D2 domain of GPVI, blocking dimerization and ligand binding. This leads to effective inhibition of receptor aggregation.
Revacept is an Fc fusion protein produced by fusing the extracellular domain of human GPVI with the human Fc domain, forming a 150 kDa homodimer with a specific hinge region. It mimics extracellular GPVI and competes with membrane-bound GPVI for ligand binding. GPVI-Fc effectively inhibits platelet adhesion to fixed collagen and thrombus formation. In a mouse carotid artery injury model, administration of 1–2 mg/kg of GPVI-Fc reduced strong platelet adhesion and aggregation. In a phase I clinical trial, researchers evaluated the safety, tolerability, and pharmacokinetic/potential dynamics (PK/PD) of Revacept in 30 healthy male subjects. Safety assessments showed that Revacept did not significantly prolong bleeding time, had no effect on platelet count or coagulation parameters, and no anti-drug antibodies were detected. Furthermore, no serious adverse events were observed during the study.
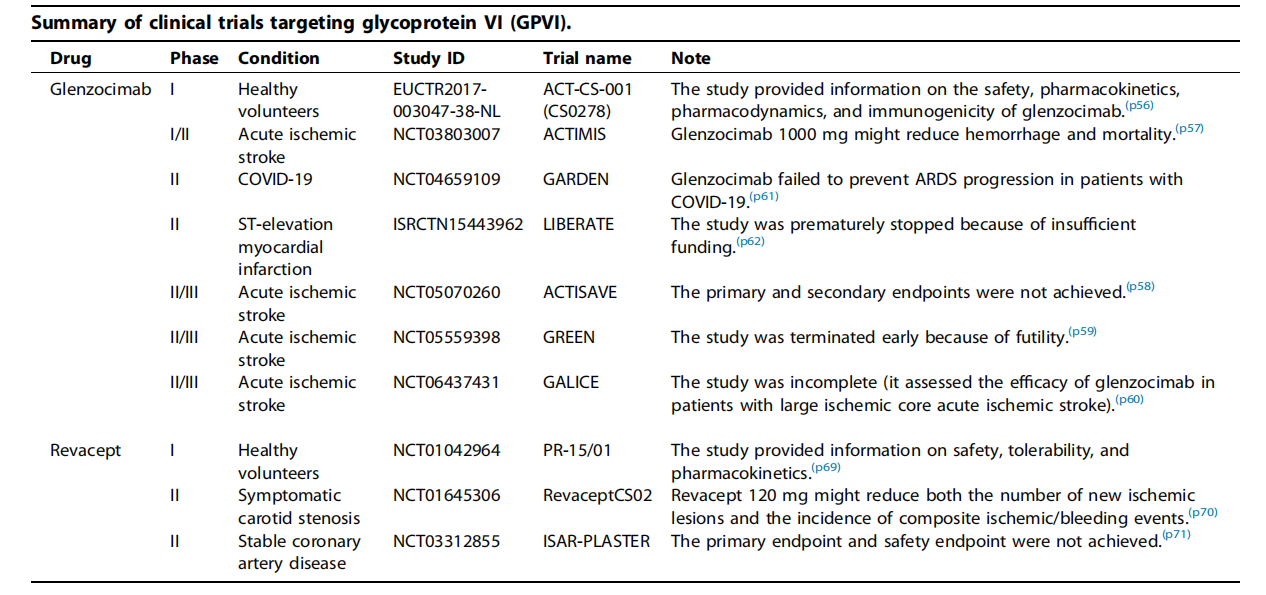
(Data source: Li R, et al. Drug Discov Today. 2025)

In addition to the above, other methods can inhibit the GPVI signaling pathway, which requires the interaction of SFKs and the FcR-γ chain with the intracellular tail of GPVI. SFKs phosphorylate the ITAM tyrosine residues of the FcR-γ chain upon GPVI activation, leading to further downstream signaling. Blocking the interaction of GPVI with SFKs or the FcR-γ chain results in signal inhibition. GPVI is cleaved by ADAM10 before reaching the transmembrane region, a process known as shedding. Extracellular GPVI is released from the platelet surface, leaving behind a truncated GPVI consisting of a transmembrane region and an intracellular region. This cleaved structure no longer possesses signaling activity because it cannot aggregate with ligands. ADAM10 - induced shedding is mediated by Tspan5 and 33. SFKs phosphorylate the ITAM sequence of the FcR-γ chain upon GPVI activation. This induces downstream signaling events. G6b-B binds to GPVI and the intracellular phosphatase SHP2. Linking SHP2 to FcR-γ can promote the dephosphorylation of the ITAM sequence and inhibit further downstream signal transduction.

(Data source: Slater A, et al. Eur Heart J Cardiovasc Pharmacother. 2024)
