Service Support
Mabnus Biotech provided protein expression services for STIM1 and TFR1.

Background
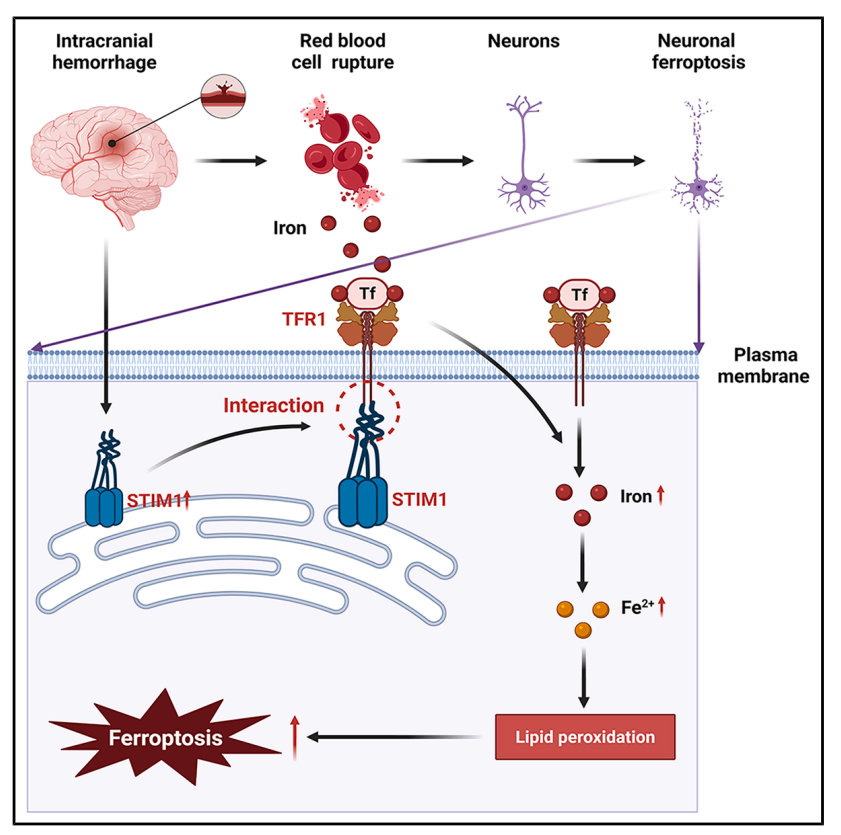
Intracerebral hemorrhage (ICH) accounts for 10%-15% of all stroke cases worldwide, making it the stroke subtype with the highest disease burden, with mortality and disability rates as high as 30%-80%. Treatment outcomes for ICH are unsatisfactory. Even with early minimally invasive evacuation of the primary hematoma, over 50% of survivors still suffer from moderate to severe neurological dysfunction, primarily due to persistent secondary brain injury (SBI) caused by the hematoma and its metabolites, leading to irreversible neuronal damage. Hematoma metabolite-induced ferroptosis is the main form of neuronal death after ICH and plays a crucial role in the progression of SBI. Given the often poor prognosis of ICH, there is a need to explore effective therapeutic targets. Matrix interaction molecule 1 (STIM1) is an important regulator of cellular calcium homeostasis, but its specific role in ICH remains unclear.

On February 17, 2026, the team led by Zhang Hongchen from Xijing Hospital published a study in Cell Rep Med entitled "The Ca2+ sensor STIM1 promotes neuronal ferroptosis by regulating iron homeostasis to exacerbate brain injury after intracerebral hemorrhage." This study found that STIM1 levels in neurons remained elevated after ICH, and elevated plasma levels in patients were associated with poor prognosis. Knocking out STIM1 in mice improved brain tissue and neuronal damage. Mechanistically, STIM1 primarily exacerbates neuronal damage by promoting ferroptosis. Importantly, in addition to regulating calcium signaling pathways, STIM1 also directly regulates iron homeostasis and promotes ferroptosis through its interaction with transferrin receptor 1 (TFR1). Finally, through virtual screening, S-IN-1 was identified as an inhibitor of the STIM1-TFR1 interaction, which can prevent neuronal ferroptosis and brain injury. These findings confirm the molecular function of STIM1 in regulating iron homeostasis, providing valuable insights and promising targets for ICH treatment.
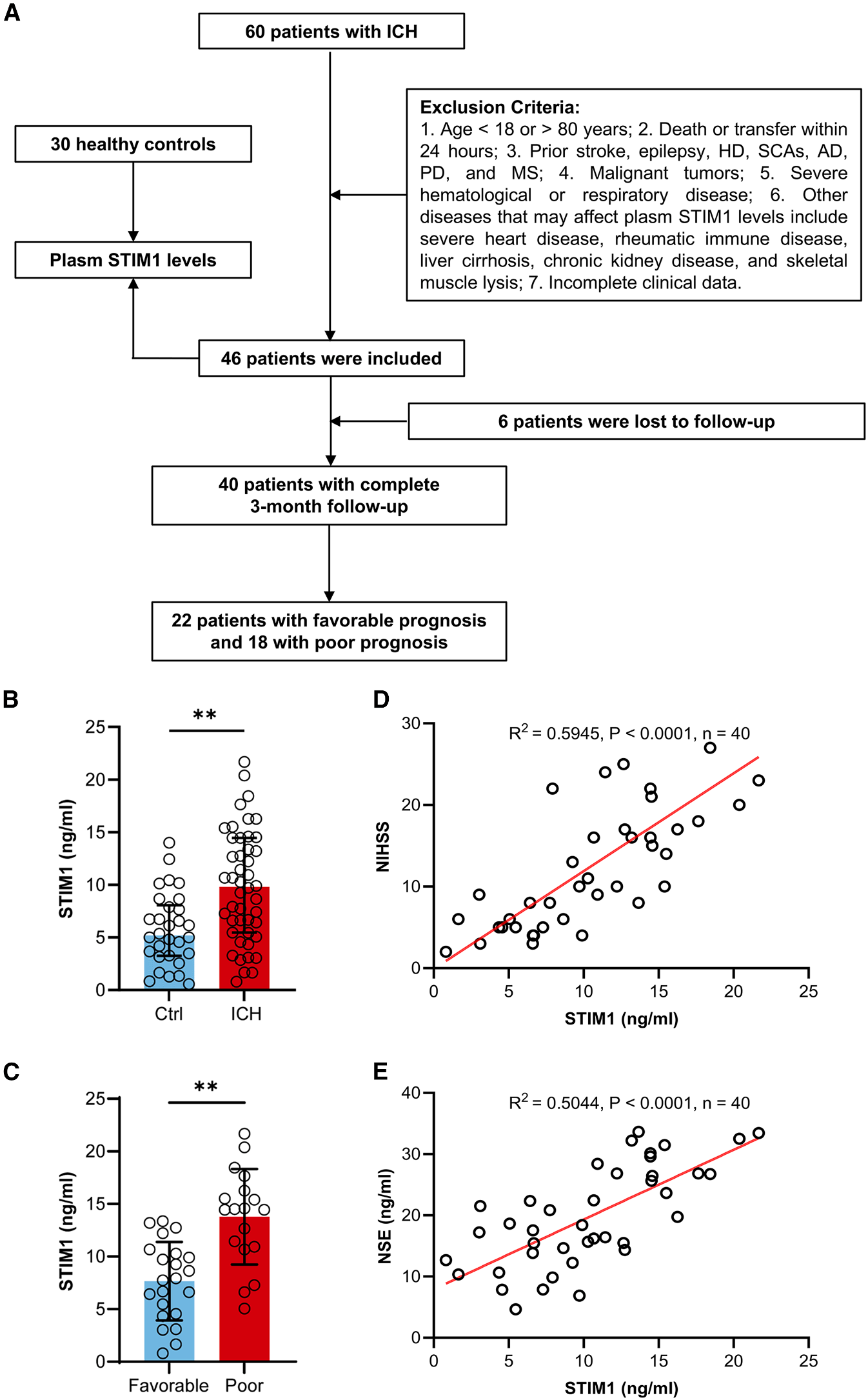
Upregulation of STIM1 expression following ICH is associated with poor prognosis
Intracerebral hemorrhage (ICH) induces increased neuronal STIM1 expression, and plasma STIM1 levels are higher in ICH patients with poor prognosis than in those with good prognosis. Plasma STIM1 levels are positively correlated with neuron-specific enolase (NSE) levels, indicating that STIM1 is involved in the neuronal damage process after ICH. These results suggest that plasma STIM1 levels are significantly elevated in ICH patients, and they have a poorer prognosis after discharge.

Neuronal STIM1 gene knockout improves brain injury after ICH
STIM1 knockout mice, generated using the Cre-LoxP recombination method, exhibited milder cerebral edema, more surviving neurons, and lower plasma NSE levels. Behavioral experiments showed that STIM1cko consistently improved modified neurological severity scores (mNSS) and sensory, motor, and cognitive deficits. STIM1 neuronal deletion reduced brain tissue and neurological damage after ICH.

STIM1 gene knockdown inhibits neuronal ferroptosis after ICH
STIM1 knockdown significantly reduced the degree of lipid peroxidation. STIM1 knockdown also significantly reduced the levels of malondialdehyde (MDA), 4-hydroxybada (4-HNE), and reactive oxygen species (ROS). Furthermore, STIM1 silencing increased the expression and activity of the key ferroptosis resistance factor GPX4 and decreased the reduced glutathione/oxidized glutathione (GSH/GSSG) ratio. In vitro and in vivo results showed that STIM1 silencing effectively reduced iron accumulation, and STIM1 knockdown effectively inhibited neuronal ferroptosis after ICH.

STIM1 regulates neuronal homeostasis after ICH by interacting with TFR1
Purified STIM1-CD and TFR1-CD proteins were constructed using a eukaryotic expression system, and their direct interaction was verified by coIP, immunofluorescence colocalization, and surface plasmon resonance (SPR) experiments. STIM1 overexpression after ICH did not affect iron accumulation in TFR1-knockdown HT22 cells. These results indicate that STIM1 interacts with its downstream target TFR1 after ICH to regulate neuronal iron homeostasis.
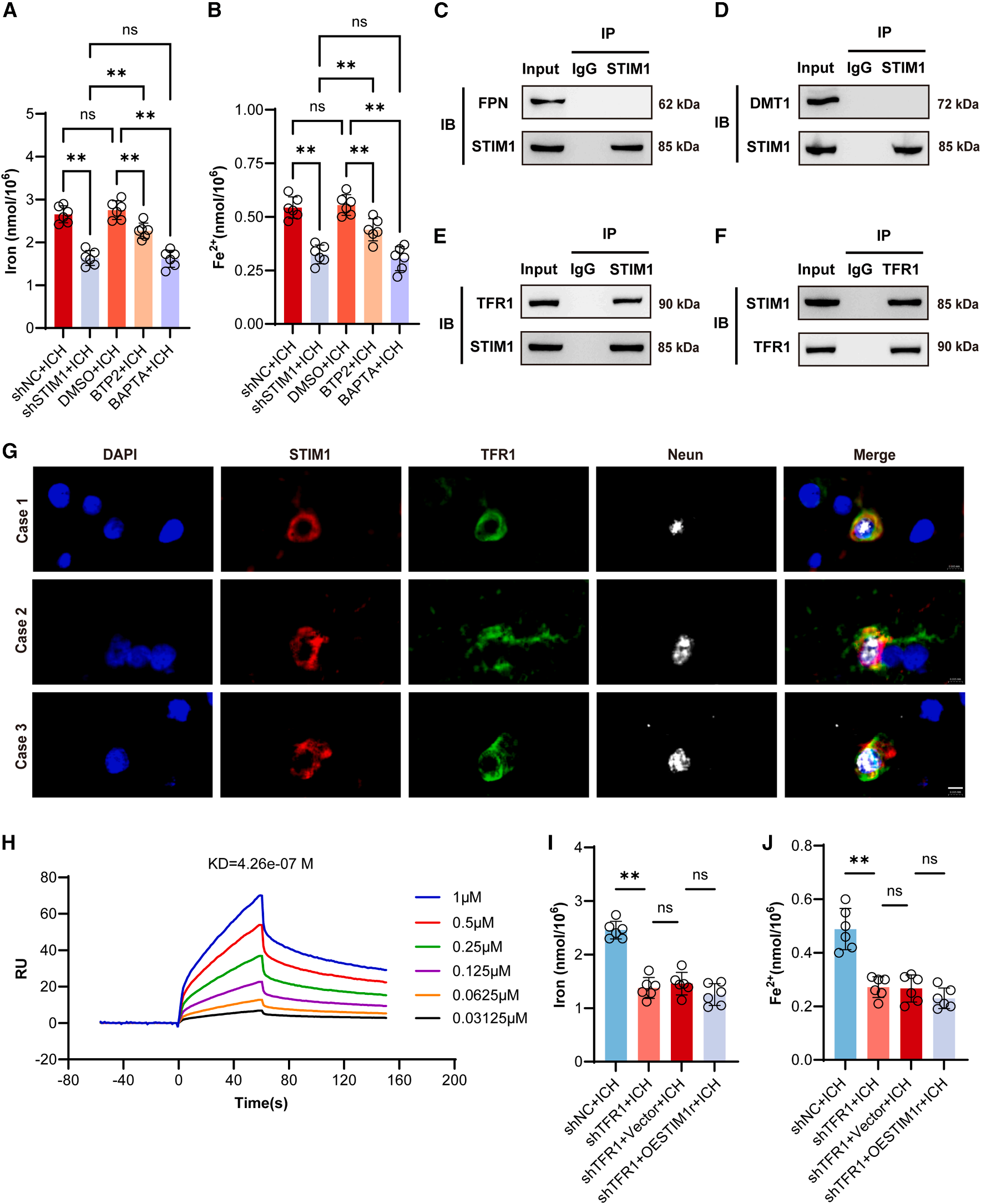
STIM1 promotes iron influx by enhancing TFR1 activity
Molecular docking and kinetic simulations revealed that the Lys385 site on STIM1 chain A contributed the highest ΔG binding and maintained hydrogen bond interactions with TFR1, Glu44, and Glu45. Mutation of STIM1 Lys385 significantly reduced the interaction between STIM1 MUT and TFR1 in STIM1 knockdown cells. STIM1 MUT reduced FL-TF uptake, and the STIM1 Lys385 mutation also attenuated STIM1-ORAI1 interaction and SOCE. However, SOCE inhibition did not affect STIM1-TFR1 binding, TFR1 expression, or TFR1 activity, indicating that STIM1-TFR1 interaction-mediated iron uptake is a biological process independent of STIM1/ORAI1-mediated SOCE. These results suggest that STIM1 can regulate neuronal iron metabolism after ICH through SOCE-independent pathways.
STIM1 MUT significantly reduced total iron content and improved neuronal survival. STIM1 MUT also significantly alleviated lipid peroxidation, reduced MDA and ROS levels, and increased GPX4 activity and the GSH/GSSG ratio. Furthermore, STIM1 MUT rescued mitochondrial morphological changes associated with ferroptosis. Taken together, these results suggest that the interaction between STIM1 and TFR1 regulates neuronal iron uptake and ferroptosis after ICH by influencing TFR1 activity.

STIM1 inhibitor S-IN-1 can inhibit neuronal ferroptosis after cerebral hemorrhage and improve brain injury
Based on virtual screening using the crystal structure of the STIM1-TFR1 complex, the candidate compound SIN1S-IN-1 was identified. By inhibiting the interaction between STIM1 and TFR1, it alleviated neuronal siderophore death after ICH. In ICH mice treated with S-IN-1, cerebral edema was reduced, the number of surviving neurons increased, and plasma NSE levels decreased. This indicates that S-IN-1 can improve brain tissue and neurological damage in ICH mice and has potential for clinical application.

Summarize
This study highlights the crucial role of STIM1 in promoting neuronal iron apoptosis and exacerbating brain injury after ICH. Mechanistically, STIM1 possesses a unique molecular function in directly regulating iron homeostasis, which promotes iron accumulation through its interaction with TFR1. S-IN-1, a specific inhibitor of the STIM1-TFR1 interaction, offers a promising therapeutic strategy for inhibiting neuronal iron stripping and improving brain injury. These findings contribute to our understanding of the pathophysiological mechanisms of neuronal iron stripping after ICH and provide promising targets for the treatment of brain injury.
