Serum amyloid protein P component (APCS), also known as SAP or PTX2, belongs to the pentamer protein family. It can interact with DNA and histones and may clear nuclear material released from damaged circulating cells. It can also act as a calcium-dependent lectin. SAP plays a useful and sensitive role as a biomarker for the diagnosis and treatment of immune-related diseases such as amyloidosis, fibrosis, cardiovascular diseases, respiratory diseases, and neuropsychiatric disorders. As an immunomodulatory factor, SAP binds to a variety of molecules, including DNA and apoptotic debris, bacterial polysaccharides, amyloid deposits, and viral proteins.
APCS expression distribution
APCS is mainly expressed in liver tissue, synthesized by hepatocytes, and specifically enriched under pathological conditions (especially at sites of amyloid deposition).
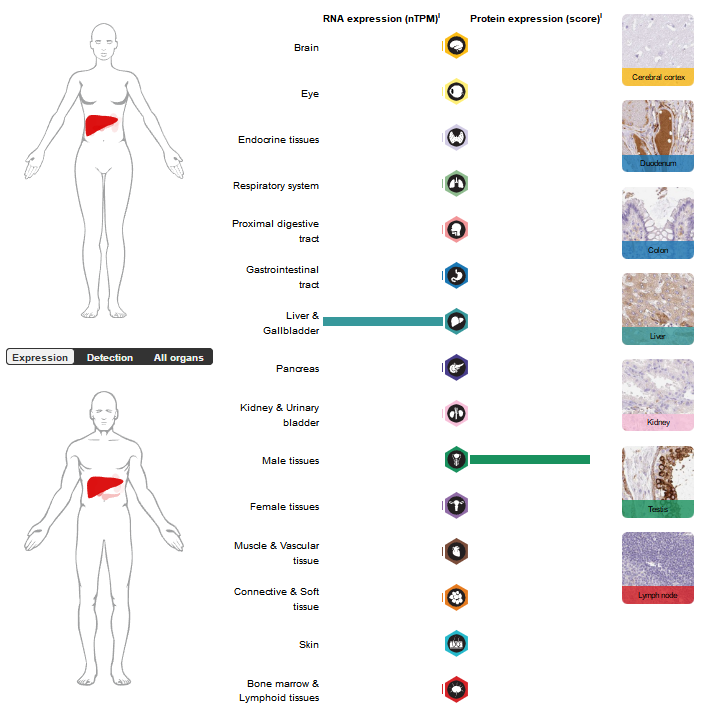
(Data source: uniprot)
Structure and receptors of APCS
APCS is a secreted protein whose monomer consists of 223 amino acids. It is a cyclic pentamer formed by five identical subunits of approximately 25 kDa non-covalent bonds, exhibiting high structural stability. Each subunit contains a conserved pentraxin domain, forming a universal platform for ligand binding. It possesses calcium-dependent ligand binding capability and a unique flattened β-micelle structure similar to legume lectins. On each side of the disk, each polymer has a short α-helix, while on the opposite surface are five bicalcium sites responsible for ligand binding. The C-terminal domain binds to FcγR on the surface of immune cells, initiating downstream effects.
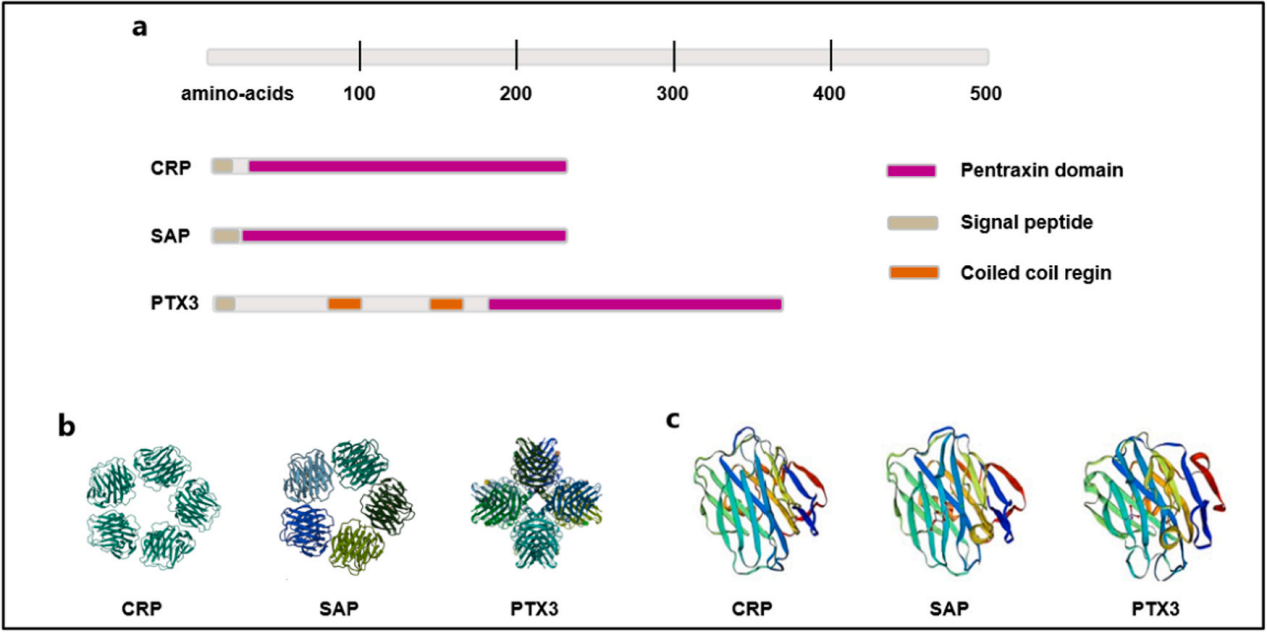
(Data source: Wang H, et al. Mol Immunol. 2024)
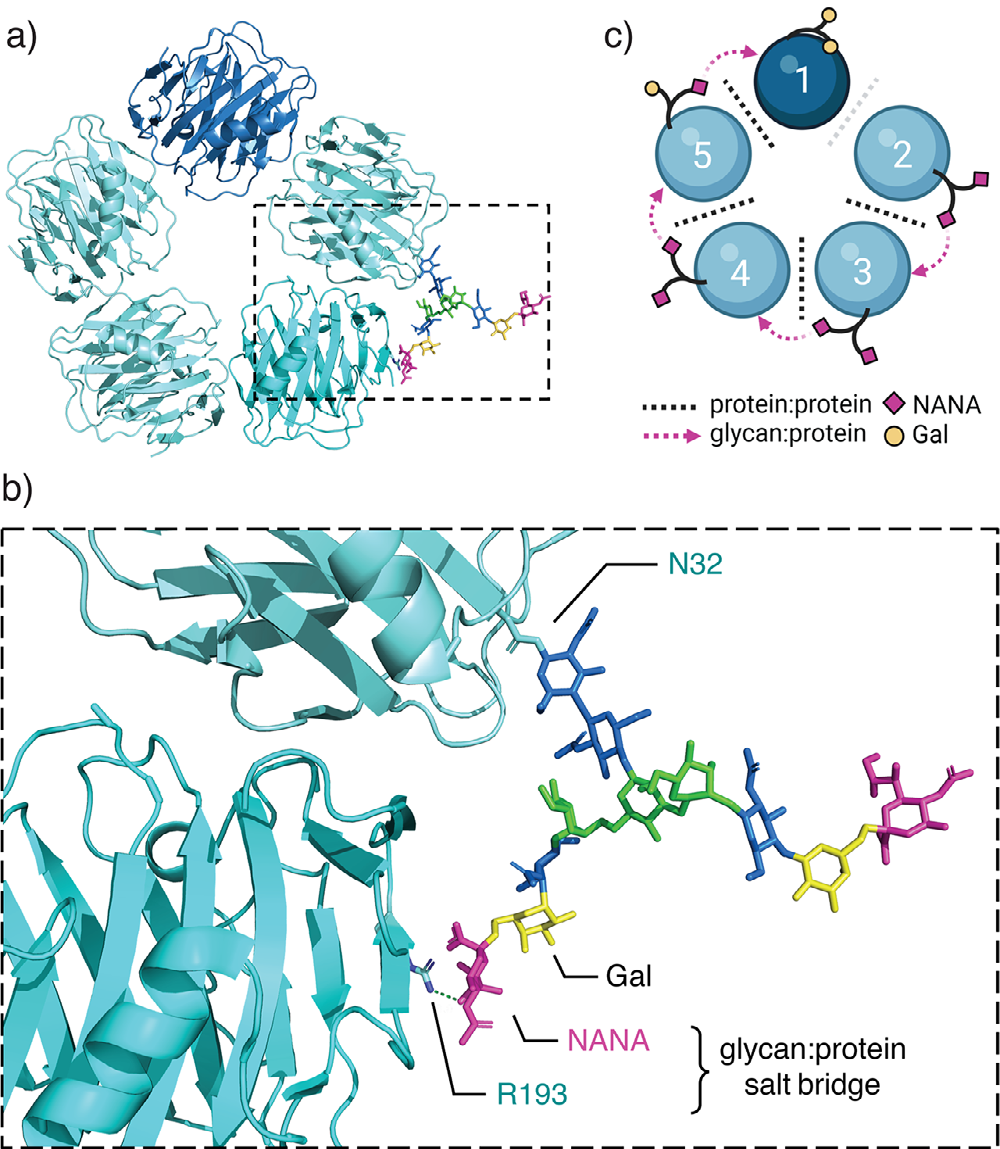
(Data source: Bittner P, et al. Angew Chem Int Ed Engl. 2025)
APCS Functions
SAP binds to various ligands, such as DNA, chromosomes, and apoptotic debris, in a calcium-dependent manner. These ligands are exposed upon cell death or tissue damage for further phagocytosis and clearance. SAP also binds to high-density lipoprotein (HDL) and very low-density lipoprotein (VLDL), indicating its involvement in lipoprotein metabolism. SAP significantly influences the treatment of various immune-related diseases through its binding to the complement system and FcγR (typically binding to FcγR1 or III). SAP reduces neutrophil adhesion, macrophage activation and polarization, enhances the phagocytosis of cellular debris, and inhibits fibroblast differentiation. APCS plays an important role in diseases such as fibrosis, amyloidosis, cardiovascular diseases, neuropsychiatric disorders, respiratory diseases, and microbial infections.

(Data source: Wang H, et al. Mol Immunol. 2024)
Targeted therapy for APCS
Dezamizumab (GSK2398852) is a monoclonal antibody targeting amyloid-dependent inflammatory craniomyocytes (APCS) for the treatment of inflammatory diseases. Developed by GlaxoSmithKline, it is currently in Phase 1 clinical trials. Dezamizumab specifically binds to the SAP protein, blocking the interaction between SAP and amyloid fibrils, promoting the clearance of amyloid fibrils, thereby reducing amyloid deposition in organs and improving organ function. In the Phase 1 clinical trial, Dezamizumab showed good safety and preliminary efficacy, effectively clearing amyloid deposits in organs such as the liver, spleen, and kidneys. NCT03044353 is a multiple-treatment study evaluating GSK2398852 administration followed by repeated administration in combination with GSK2315698. The primary objective of this study was to assess the reduction in cardiac amyloid burden after repeated anti-SAP therapy in all study groups via CMR, and to evaluate the safety and tolerability of repeated anti-SAP therapy, including compatibility with chemotherapy in patients with AL systemic amyloidosis. However, due to the cessation of research and development work on amyloidosis, GSK terminated this Phase II trial on October 16, 2019.
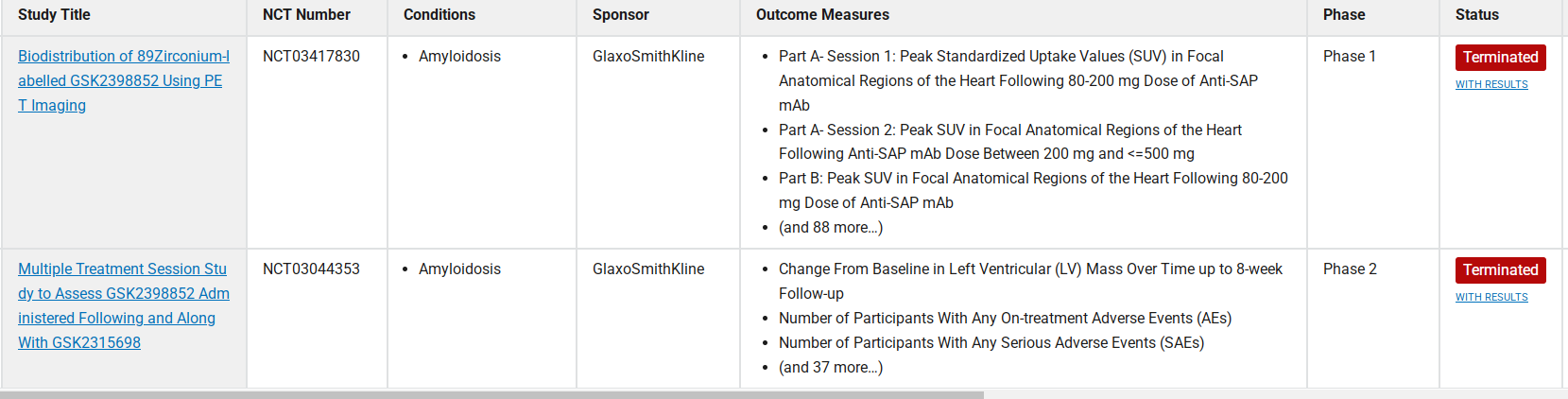
(Data source: clinicaltrials)
