ACVRL1 is a type I receptor for the TGF-β family ligands BMP9/GDF2 and BMP10, and is an important regulator of normal angiogenesis. It activates the downstream Smad1/5/8 signaling pathway by binding to BMP9/BMP10, thus promoting angiogenesis while maintaining vascular quiescence and homeostasis. ALK1 has become an important molecular target for tumor anti-angiogenesis.
ACVRL1 expression distribution
ACVRL1 is highly expressed in organs with high blood flow perfusion, such as the liver, lungs, and kidneys, and is mainly expressed in endothelial cells, intestinal cells, endothelial cells, mononuclear phagocytes, trophoblast cells, and fibroblasts.
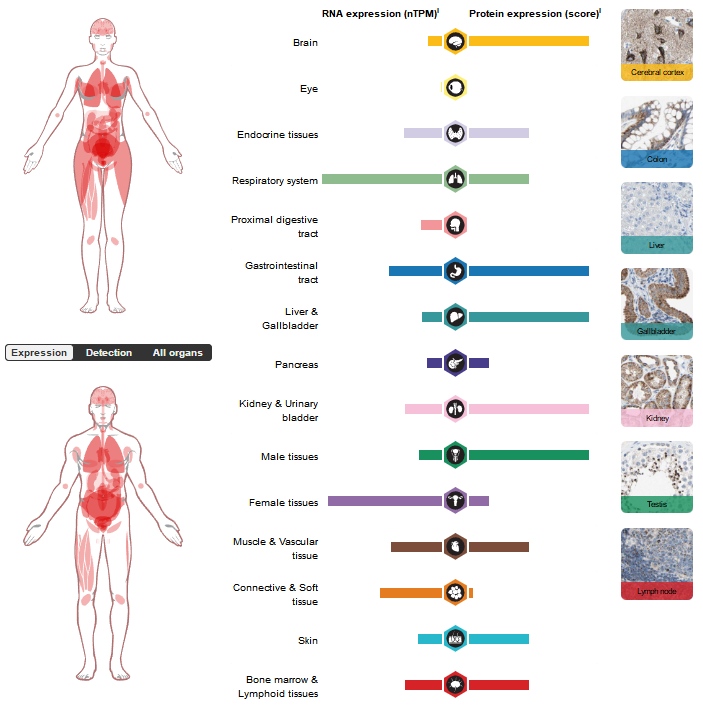
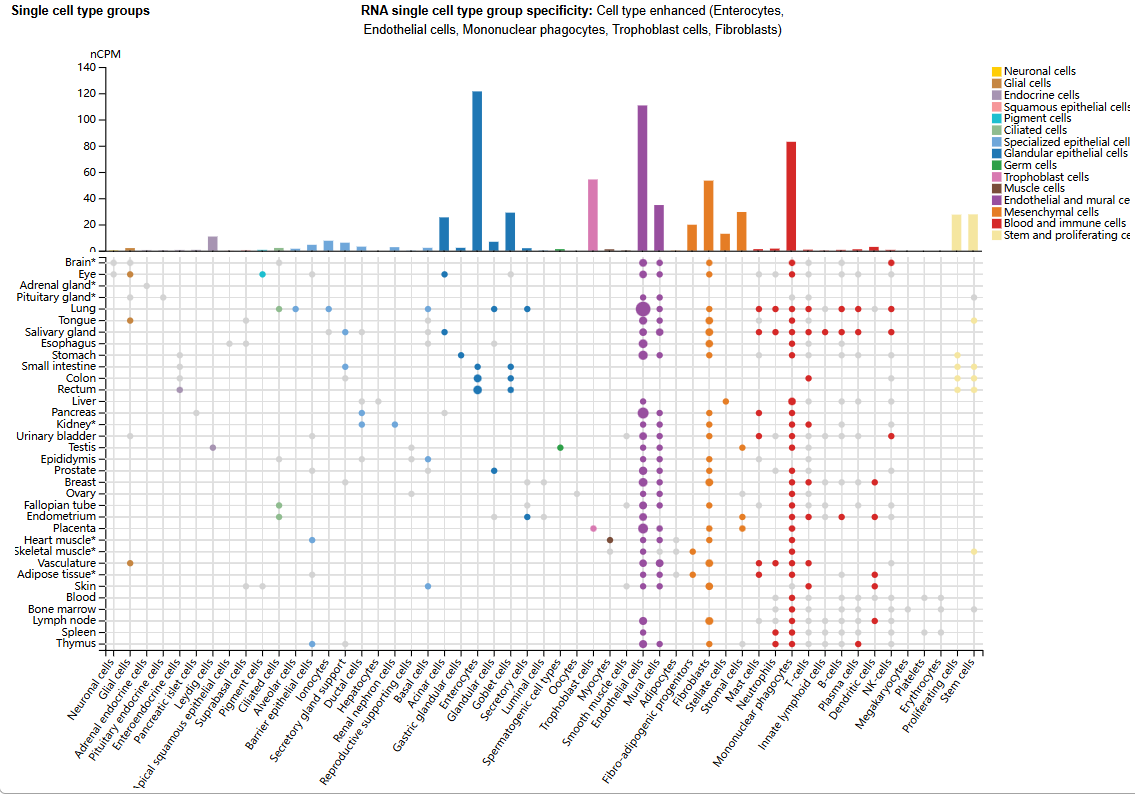
(Data source: uniprot)
Structure of ACVRL1 and its receptor
ACVRL1 is a type I membrane protein composed of 503 amino acids, consisting of an extracellular region, a transmembrane domain, and an intracellular region.
Extracellular region: Contains a ligand-binding domain that specifically recognizes and binds to BMP9 and BMP10.
Transmembrane region: Anchors receptors to the cell membrane.
Intracellular region: GS domain, rich in glycine-serine, is a key site for phosphorylation activation of ACVRL1 by type II receptors (such as BMPR-II).
Kinase domain: Possesses serine/threonine kinase activity. Upon phosphorylation, it can further phosphorylate downstream Smad proteins (mainly Smad1/5/8), initiating signal transduction. ACVRL1 typically forms a complex with type II receptors, and its activity is regulated by co-receptors (such as Endoglin) and intracellular repressor proteins (such as FKBP12).
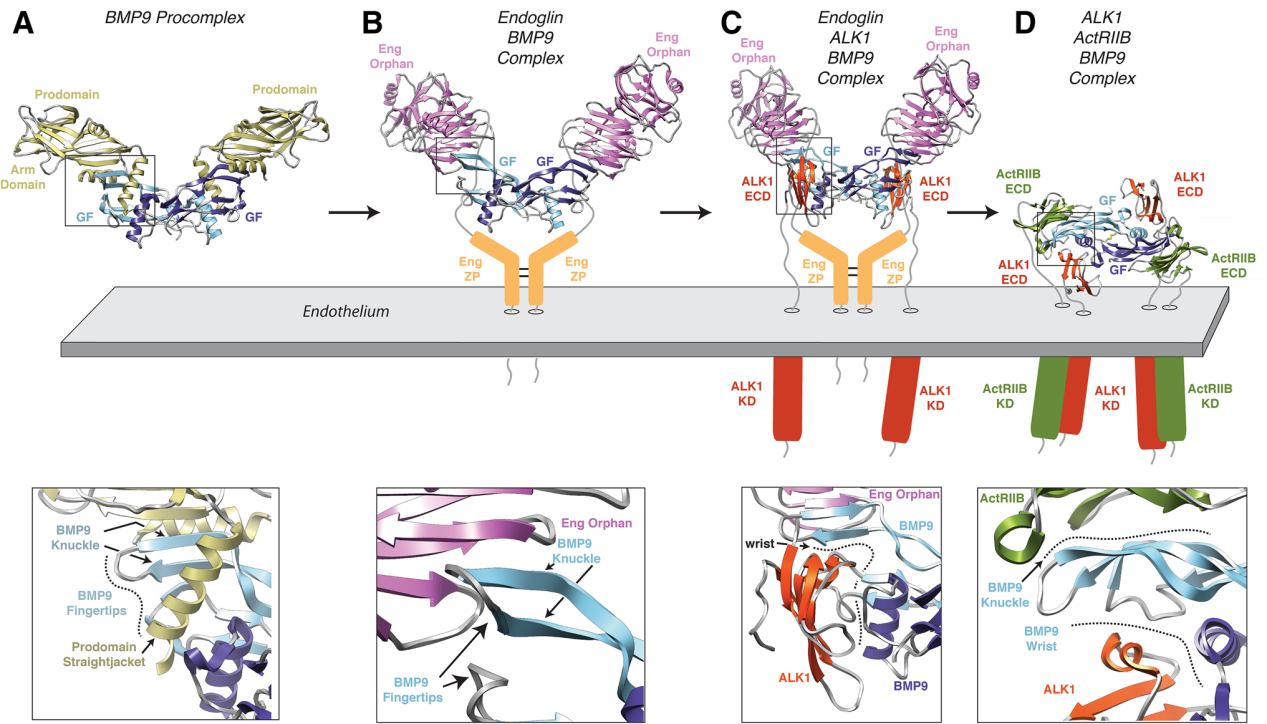
(Data source: Roman BL, et al. Cell Mol Life Sci. 2017)
ACVRL1 signaling pathway and regulation
ALK1 is primarily activated by TGF-β/BMP ligands (especially BMP9 and BMP10), and its signal transduction involves both Smad-dependent and Smad-independent pathways.
BMP9 (a high-affinity ligand) or BMP10 binds to ALK1, forming an ALK1/ BMPRII (type II receptor)/ L- ENG receptor complex. ALK1 is phosphorylated by BMPRII and activates Smad1/5/8. The activated Smad1/5/8 forms a complex with Smad4. This complex enters the cell nucleus, regulates the expression of target genes (such as Id1, Hey1/2, etc.), promotes endothelial cell proliferation and migration, and participates in the angiogenesis activation phase.
After TGF-β1 binds to TβRII, it can only induce Smad1/5/8 phosphorylation in the presence of ALK5. ALK1 and ALK5 are two antagonistic type I receptors in the TGF-β superfamily, jointly regulating the activation and quiescence homeostasis of endothelial cells. ALK5 can activate Smad2/3, promoting angiogenesis.
The co-receptor Endoglin (CD105) is a key regulatory node: its long isoform (L-ENG) enhances ALK1-Smad1/5 signaling, while its short isoform (S-ENG) promotes ALK5-Smad2/3 signaling.
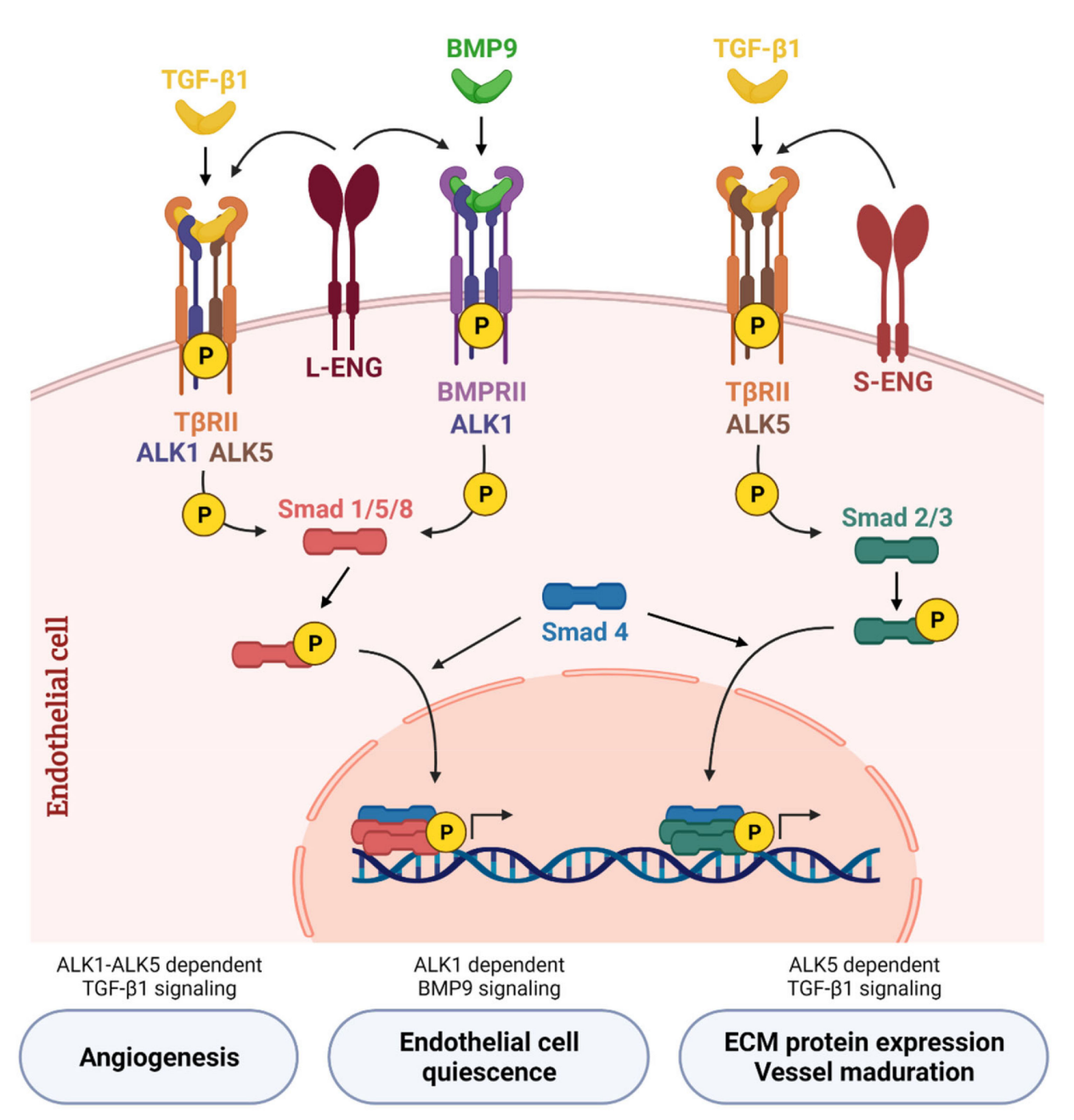
(Data source: Ayuso-Íñigo B, et al. Cancers (Basel). 2021)
The role of ACVRL1 in cancer
The dual role of BMP9-ALK1 in endothelial cell homeostasis can be used for different treatment methods: inhibiting tumor angiogenesis, where ALK1 acts as a pro-angiogenic receptor (BMP9 is activated and promotes angiogenesis through Smad1/5/8 signaling), which can be used to reduce tumor angiogenesis and tumor growth.
The activity of circulating factor BMP9 induces vascular arrest through the ALK1 receptor, and these effects can be enhanced or mimicked to normalize blood vessels for therapeutic purposes. This presents an opportunity to investigate its effects on vascular normalization and to create a favorable tumor microenvironment through the administration of recombinant BMP9 or its analogues.

(Data source: Ayuso-Íñigo B, et al. Cancers (Basel). 2021)
Targeted therapy for ACVRL1
Ascrinvacumab (GT90001) is a monoclonal antibody targeting ACVRL1. Ascrinvacumab inhibits ALK-1/TGF-β signaling and tumor angiogenesis, and is used to treat advanced hepatocellular carcinoma, esophageal squamous cell carcinoma, and adenocarcinoma of the gastroesophageal junction. Ascrinvacumab is a potential first-in-class antibody for which Kintor Pharmaceuticals obtained exclusive global licensing from Pfizer in 2018. This clinical study (NCT03893695) was initiated in Taiwan on May 7, 2019, to evaluate the safety and efficacy of GT90001 in combination with Nivolumab (Opdivo) in patients with advanced HCC who had progressed after first-line treatment with lafenib or lenvatinib. The results presented by GT90001 at ASCO GI 2021 highlight the good safety and efficacy of GT90001 in combination with Nivolumab.
Kintor Pharmaceuticals has developed a bispecific antibody targeting ALK1 and VEGF for the treatment of solid tumors, which is currently in the preclinical research stage.

(Data source: Kaitu Pharmaceutical official website)
DIAG723 is a bispecific antibody targeting ALK1 and BMPR2. DIAG723 restores normal ALK1 signaling by aggregating key cell surface receptors, addressing the root cause of HHT (hereditary hemorrhagic capillary hemorrhage) and the underlying cellular signaling imbalances supporting hereditary and idiopathic polythyroid hypertension (PAH), while providing a differentiated safety profile. Preclinical studies have shown that DIAG723 can prevent and reverse arteriovenous malformations and prevent HHT-related anemia. DIAG723 has received orphan drug designation from the FDA and EMA for the treatment of HHT. Clinical studies have shown that DIAG723 can prevent the formation of hyperbaric oxygen therapy and cardiac remodeling, and improve arterial blood flow. Data indicate that DIAG723 can restore normal signaling in patient-derived cell models of hereditary and idiopathic polythyroid hypertension. DIAG723 is currently in the IND support phase and is expected to enter clinical development in 2026.
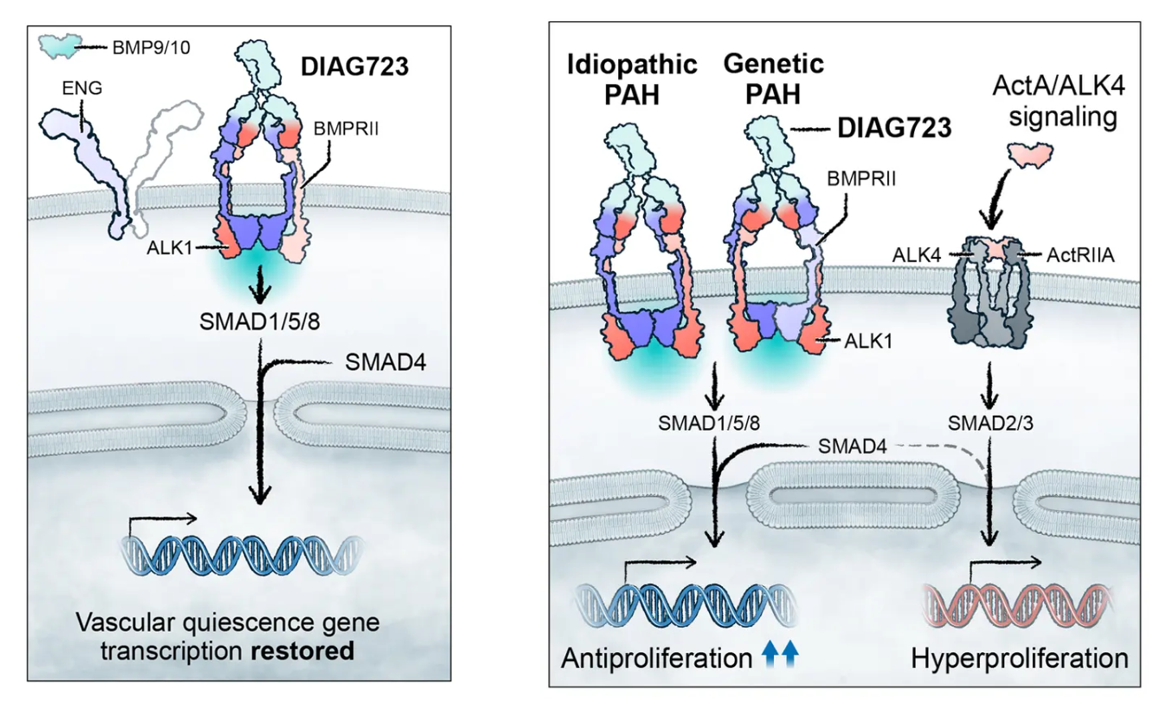
(Data source: Diagonal Therapeutics official website)
