Von von Willebrand factor (VWF) is the largest known polysaccharide protein in the blood, controlling platelet adhesion and aggregation. Its normal function requires the assembly of VWF into large, disulfide-linked polymers. VWF plays a crucial role in maintaining hemostasis by forming molecular bridges between the subendothelial collagen matrix and the platelet surface receptor complex GPIb-IX-V, promoting platelet adhesion to the site of vascular injury. It also acts as a molecular chaperone for coagulation factor VIII, transporting it to the injury site, stabilizing its heterodimeric structure, and preventing premature clearance from the plasma. Von von Willebrand disease (VWD) is the most common inherited bleeding disorder worldwide.
VWF expression distribution
VWF is mainly expressed in the endometrium, testes, bone marrow, cerebral cortex, and colon tissue, and is also expressed in endothelial cells, macrophages, and platelets.


(Data source: uniprot)
The structure and receptors of VWF
VWF is a secreted protein composed of 2813 amino acids. Pre-VWF contains a 22-amino acid signal peptide, a 741-amino acid propeptide (domains D1 and D2), and a 2050-amino acid mature subunit comprising multiple domains. Each VWF has four modules in its D domain (VWD, C8, TIL, and E8). Pre-VWF monomers form tail-to-tail homodimers via interchain disulfide bonds between the cystine knot (CK) domains in the ER. After being transported to the Golgi apparatus, these pre-VWF dimers assemble into head-to-head multimers via interchain disulfide bonds between the D3 domains of two pre-VWF dimers. These multimers can aggregate into tubules called Weibel-Palade bodies within storage particles, playing a crucial role in hemostasis by delivering VWF multimers into circulation.
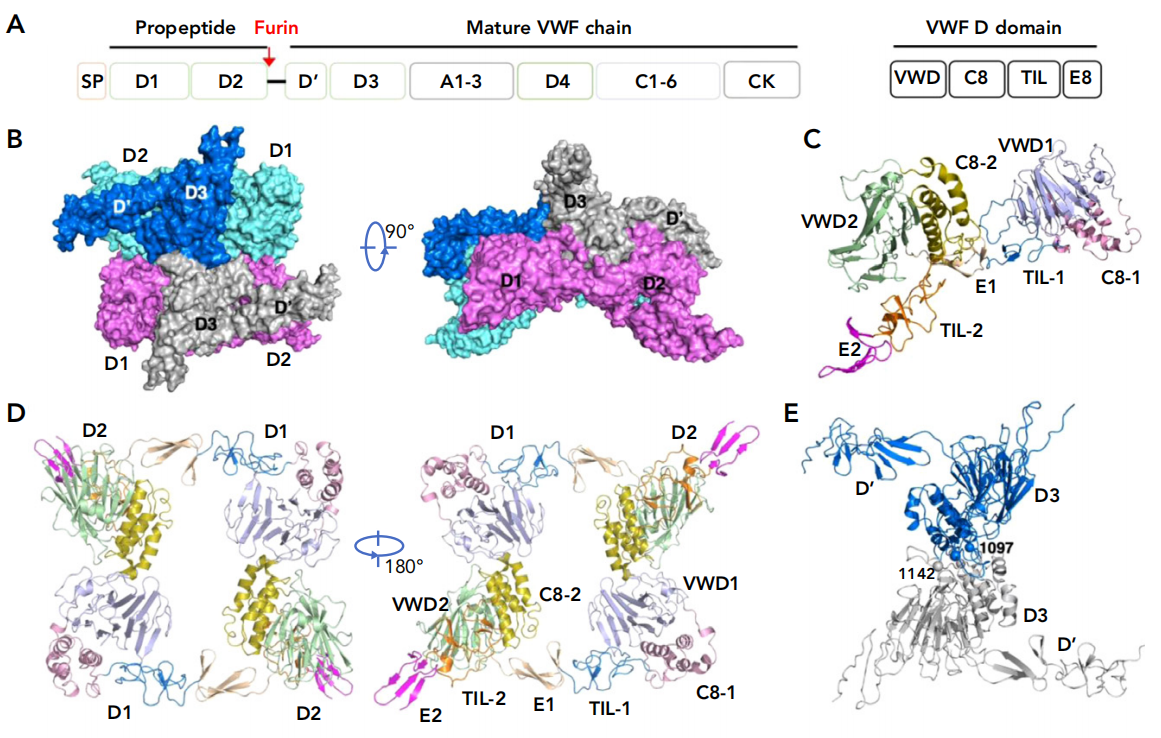
(Data source: Zeng J, et al. Blood. 2022)
VWF can interact with a variety of structurally different binding partners; some of these ligands have binding sites located in specific domains.

(Data source: Atiq F, et al. Blood. 2024)
Biological functions of VWF
In addition to hemostasis, VWF plays an important role in inflammation, angiogenesis and wound healing. Its various biological effects are mediated by its action on endothelial cells, neutrophils, leukocytes, macrophages, dendritic cells, tumor cells, smooth muscle cells and osteoclasts.
Effects of VWF on inflammation: The interaction of VWF with specific macrophage receptors (particularly LDL receptor-associated protein 1) initiates significant downstream inflammatory signaling (including MAPK kinase protein kinase and nuclear factor kappa). Activation of κB ultimately triggers macrophages to adopt the M1 pro-inflammatory phenotype. VWF can also affect the inflammatory response in other ways; VWF has also been reported to bind to dendritic cells. This increases the possibility that VWF influences the adaptive immune response.
Effects of VWF on angiogenesis: VWF primarily exerts an inhibitory effect on the regulation of angiogenesis through a variety of different mechanisms. These include the putative roles of the chaperone proteins Angpt-2 and Gal-3, which are normally transported to vascular smooth muscle cells (WPBs) through their interaction with VWF.
Effects of VWF on Wound Healing: Dermal wound healing is significantly delayed in VWF-deficient mice. In wounded tissues, the heparin-binding domain (tyrosine 1328-alanine 1350) of the VWF A1 domain has been shown to bind to important growth factors, including platelet-derived growth factor, VEGF, FGF, and members of the transforming growth factor β family. Localized angiogenesis impairment is observed at the wound site in VWF-deficient mice, which may be one reason for the observed poor wound healing. Other ischemia-related animal models have also reported von Willebrand factor (VWF) promoting and inhibiting angiogenesis, suggesting that these effects may vary depending on specific conditions and tissues.

In addition to its role in bleeding and thrombotic disorders (including von Willebrand disease, thromboembolic thrombocytopenic purpura, deep vein thrombosis, pulmonary embolism, myocardial infarction, and ischemic stroke), VWF has other pathogenic effects in a range of other non-hemostatic diseases, such as sickle cell disease (SCD), liver disease, and tumor-related diseases (breast cancer, thyroid cancer, and gastric cancer). VWF plays a role in regulating specific aspects of tumor cell biology, particularly in metastasis, apoptosis, and tumor angiogenesis.
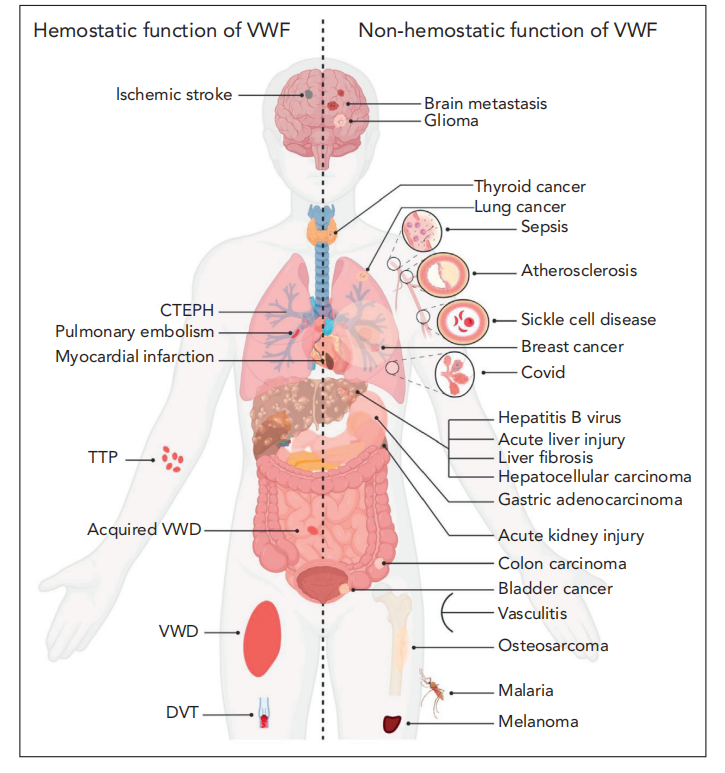
(Data source: Atiq F, et al. Blood. 2024)
VWF-targeted therapy
Cablivi (caplacizumab) is a bivalent anti-von Willebrand factor (vWF) nanobody, VHH, used in combination with plasma exchange and immunosuppressive therapy to treat patients experiencing episodes of acquired thrombotic thrombocytopenic purpura (aTTP), also known as immune-mediated thrombotic thrombocytopenic purpura (iTTP). Cablivi is the first and only treatment designed to prevent the formation of microthrombi, small blood clots that form in the microvascular system and help prevent organ damage. Cablivi is currently available in nearly 30 countries, including the United States, the European Union, the United Kingdom, Switzerland, Brazil, Colombia, Japan, and five Gulf Coast countries. Cablivi received priority review due to its approval in China and has been granted priority review designation by the FDA for pending label expansion to include treatment in adolescents aged 12 years and older.
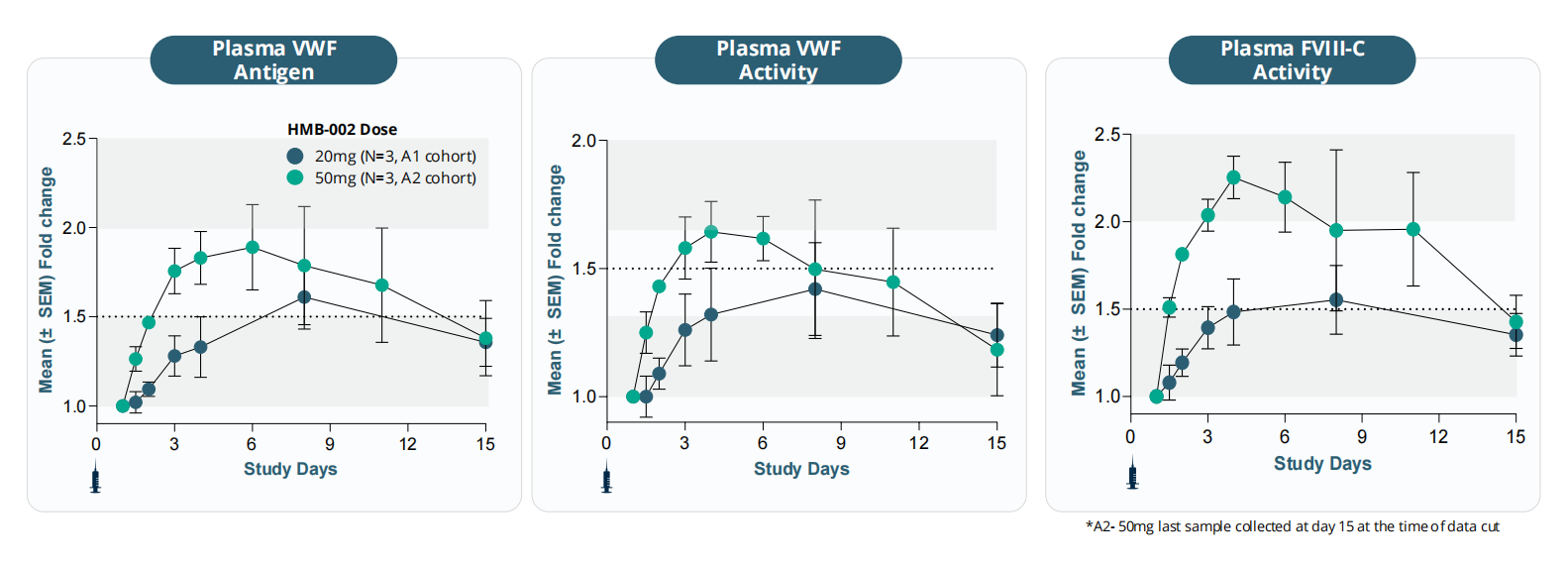
HMB-002 is a monoclonal antibody targeting VWF, developed by Hemab ApS. VWF accumulates in VWD and restores hemostasis, binding to FcRn receptors and circulating via the FcRn pathway, thus delaying VWF clearance. Elevated VWF levels promote platelet aggregation, increase FVIII accumulation, and support thrombin generation and clot formation. HMB-002 aims to accumulate endogenous von Willebrand factor as a prophylactic treatment option to prevent and reduce the frequency of bleeding events in patients with von Willebrand disease (VWD). Numerous clinical studies are currently underway for HMB-002. The VELORA Pioneer trial (NCT06754852) is a phase 1/2 clinical study of HMB-002 for the treatment of von Willebrand disease. HMB-002 is being developed as a prophylactic therapy aimed at reducing the number and severity of bleeding events in VWD patients through a convenient subcutaneous administration regimen.
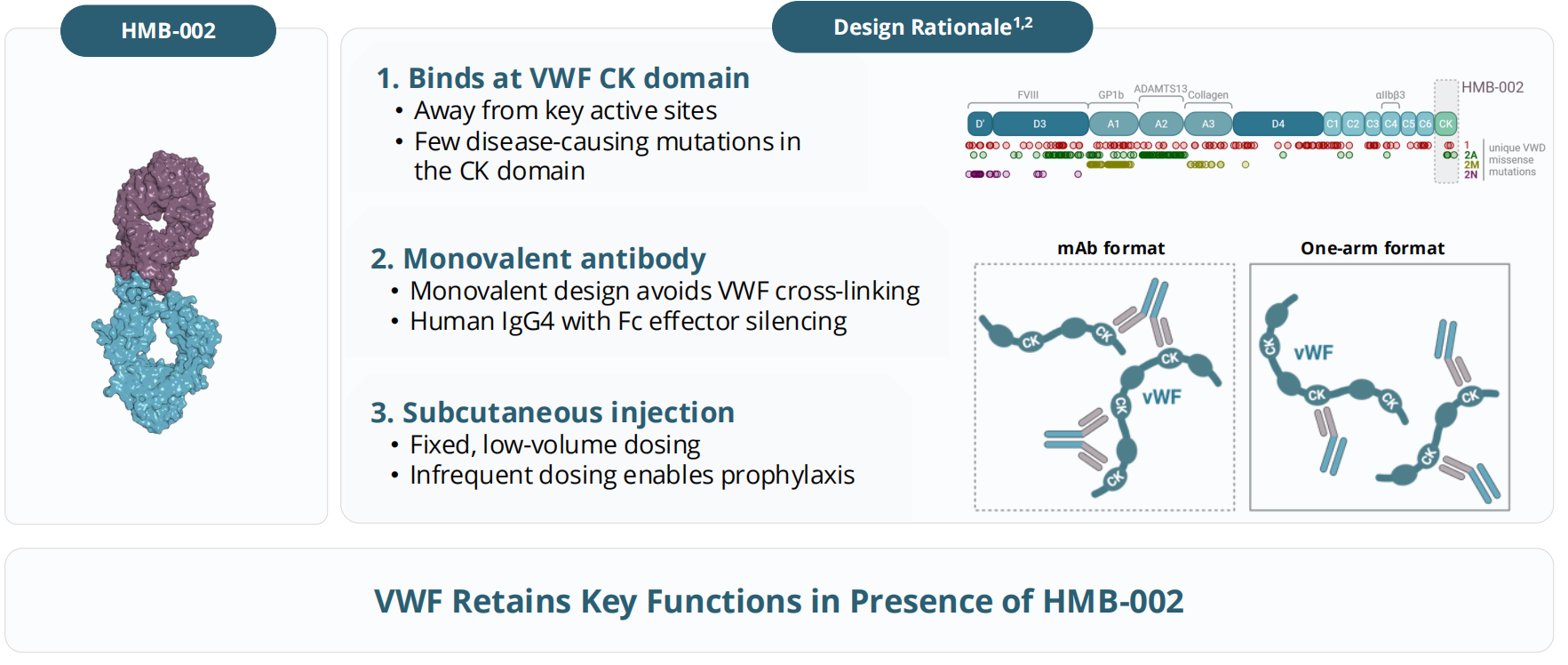
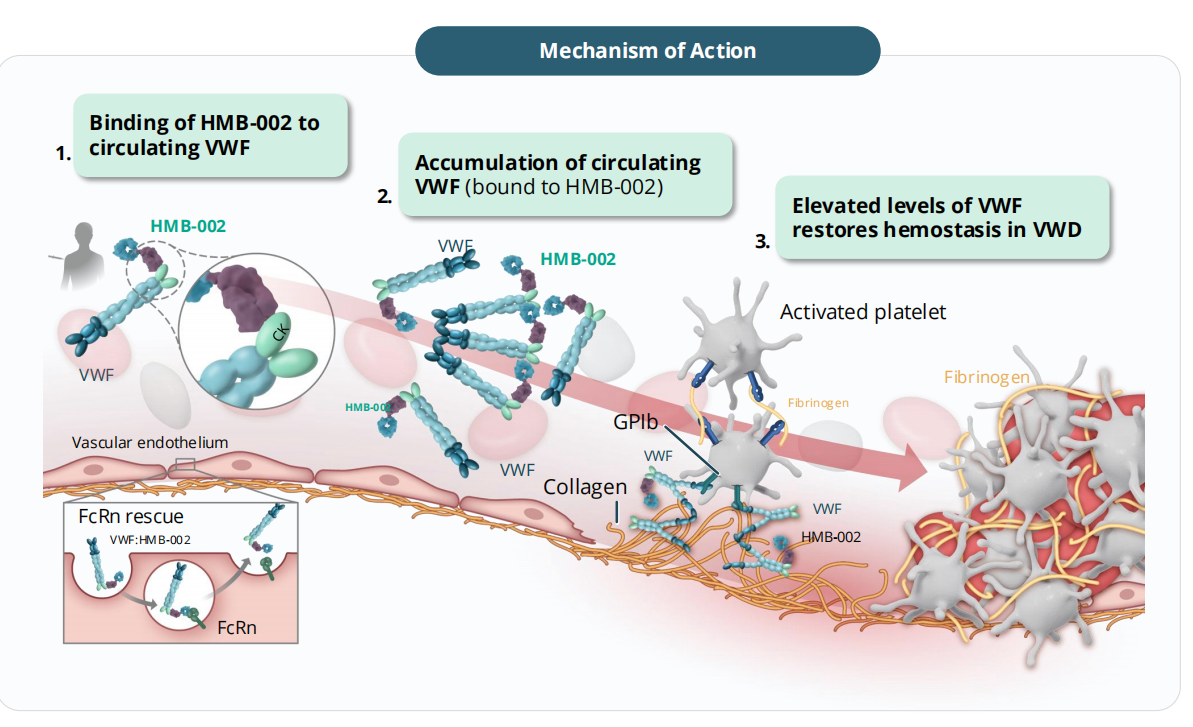
The dose-dependent accumulation of von Willebrand factor antigen, von Willebrand factor activity, and coagulation factor VIII activity was increased and the accumulation time was prolonged. In the A2 cohort (50 mg dose), the accumulation of all three parameters (PD parameters) exceeded 1.5 times the initial value, and this accumulation state lasted for at least 8 to 10 days.
(Data source: Hemab ApS official website)
