Background
Over the past decade, the number of non-natural monoclonal antibodies has grown exponentially, thanks to the emergence of bispecific and multispecific antibodies. This diversification of major sequences presents challenges to CHO biomanufacturing platforms. Variations in antibody sequences can negatively impact production, typically leading to poor cell growth, low specific productivity, and decreased product quality. Factors contributing to low yields of these difficult-to-express (DTE) antibodies include inefficient folding, assembly mismatch, intracellular accumulation, and chain aggregation. Monoclonal antibody (mAb) production in CHO cells depends on the balanced co-expression of heavy chains (HC) and light chains (LC). Imbalances between HC and LC can reduce titers, impair product quality, and negatively impact cell viability, meaning that the optimal LC/HC expression ratio should be determined as early as possible during cell line development.

On January 9, 2026, researchers from Biomap published a study titled "High-throughput optimization of antibody production in CHO cells by tuning heavy-and light-chain promoter strength" in Front Bioeng Biotechnol. This study presents a high-throughput screening platform that, combined with a Design of Experiments (DoE) strategy, determines the optimal LC/HC expression balance during the transition phase. This platform provides a rapid and scalable method to define antibody-specific LC/HC promoter strength combinations, maximizing productivity without compromising cell health and enabling more informed construction choices before committing to stable clone generation.
Plasmid generation and transfection operations in high-throughput systems
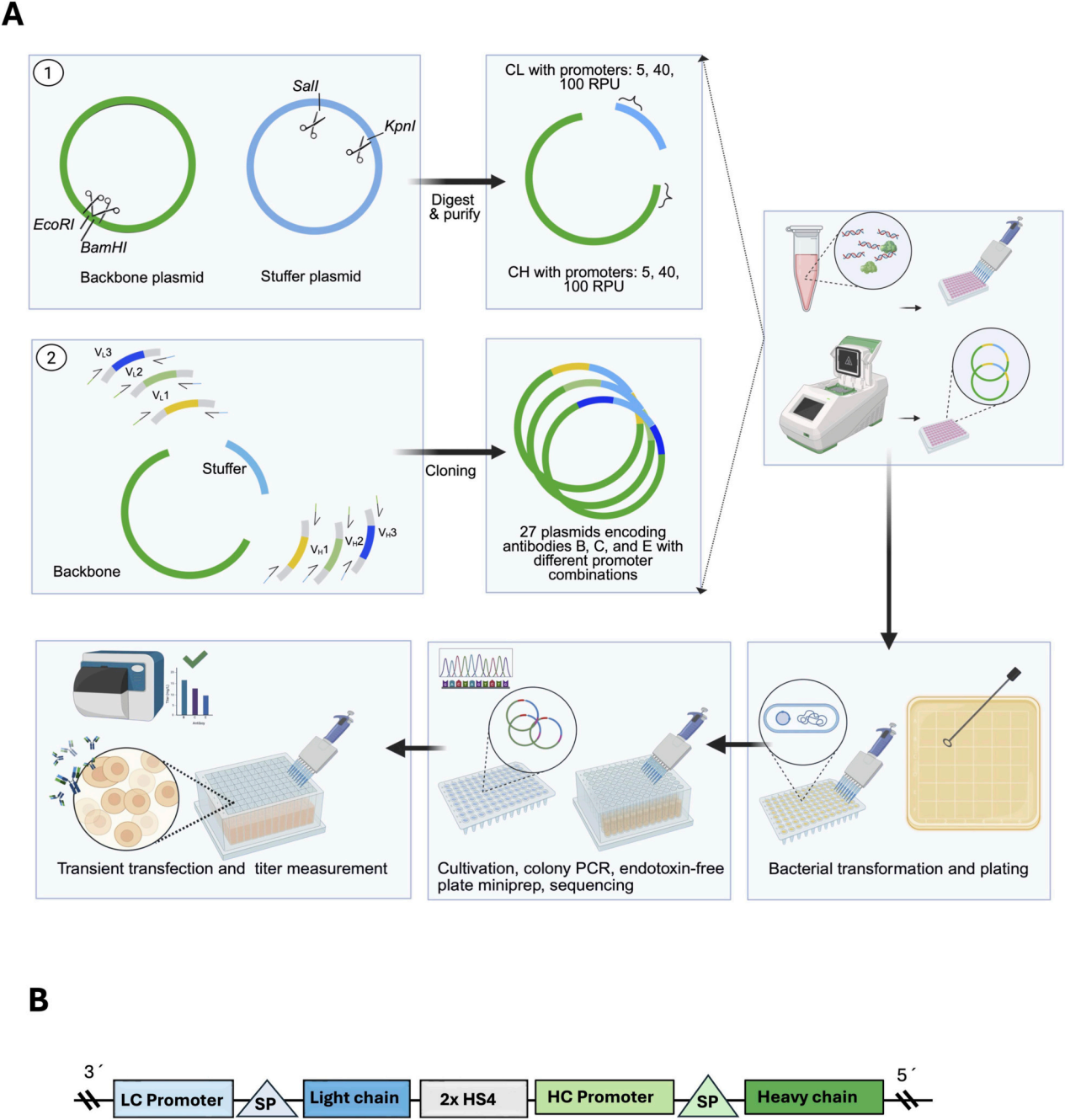
For rapid screening of LC/HC promoter strength ratios in CHO cells, we employed a high-throughput workflow using a single vector in a 96-well format. All steps, from PCR and cloning/colony PCR to miniprep, transfection, and antibody assays, were performed in 96-well plates. Two insulating sequences were inserted between the LC and HC transcription units.
Plasmid assembly consisted of two steps. First, filler plasmids and backbone plasmids carrying the CL and CH regions were constructed, and mAb-specific variable regions (VL and VH) were amplified by PCR. Second, four fragments (CL, CH, VL, VH) were assembled to generate the final LC-HC dual-box structure expressing antibodies B, C, and E. Successful assembly was screened by colony PCR and confirmed by Sanger sequencing across the linkers and variable regions. For each LC/HC promoter condition and antibody, a clone with sequence validation was selected for downstream experiments. Twenty-seven plasmids were rapidly constructed (3 antibody × 9 LC/HC promoter combinations). In the transfection stage, the system achieved an average transfection efficiency of 91% in CHO-S cells, ensuring consistency between wells and data reliability.
Optimal heavy-to-light chain ratio for transient monoclonal antibody production
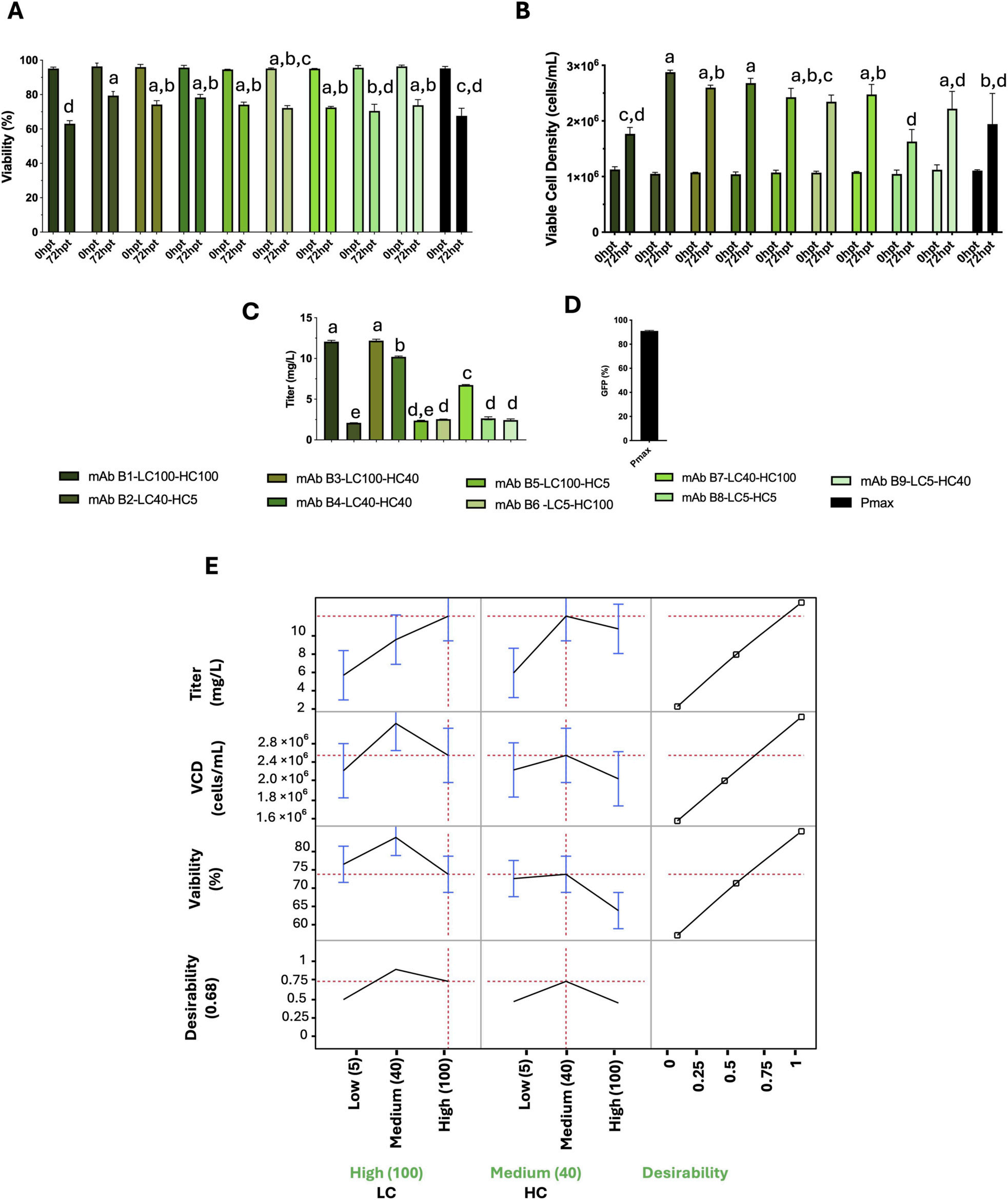
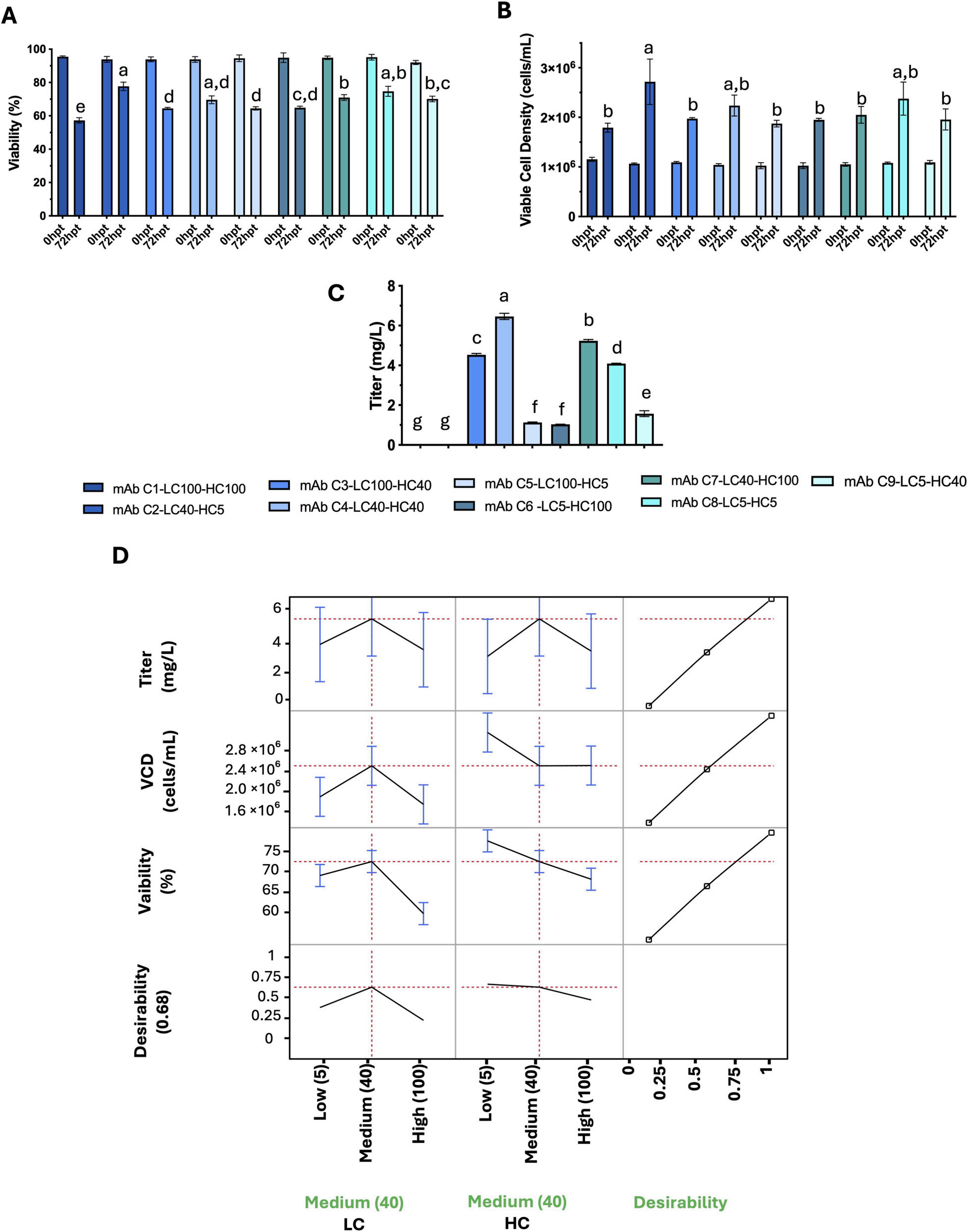
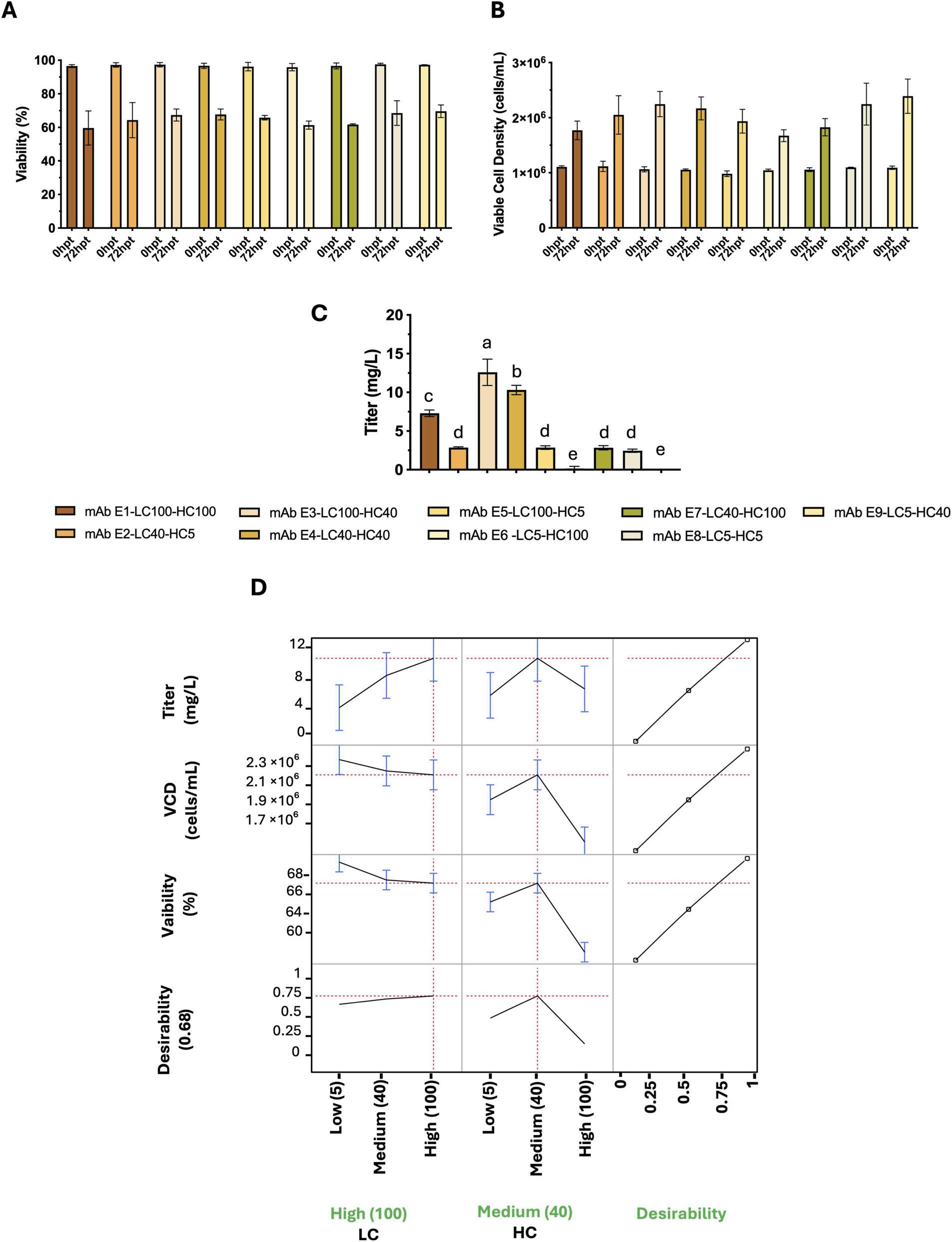
For the D-optimal custom-design experimental design (DoE) approach, three different antibody sequences were analyzed, and the performance of two taxonomic factors (HC and LC) at three expression levels (5, 40, and 100 RPU) was evaluated, with three response variables: viable cell density (cells/mL), viability (%), and titer (mg/L). The optimal promoter combinations for the three antibodies (B, C, and E) differed.
Antibody B: High LC + Medium HC (LC100-HC40)
Antibody C: Medium LC + Medium HC (LC40-HC40)
Antibody E: High LC + Medium HC (LC100-HC40)
This indicates that the optimal light chain/heavy chain promoter ratio is antibody-dependent. For each antibody, the optimal HC/LC ratio must be determined experimentally, as sequence-specific folding kinetics and assembly efficiency shape the ideal expression balance for maximizing yield and maintaining cell viability.
Summarize
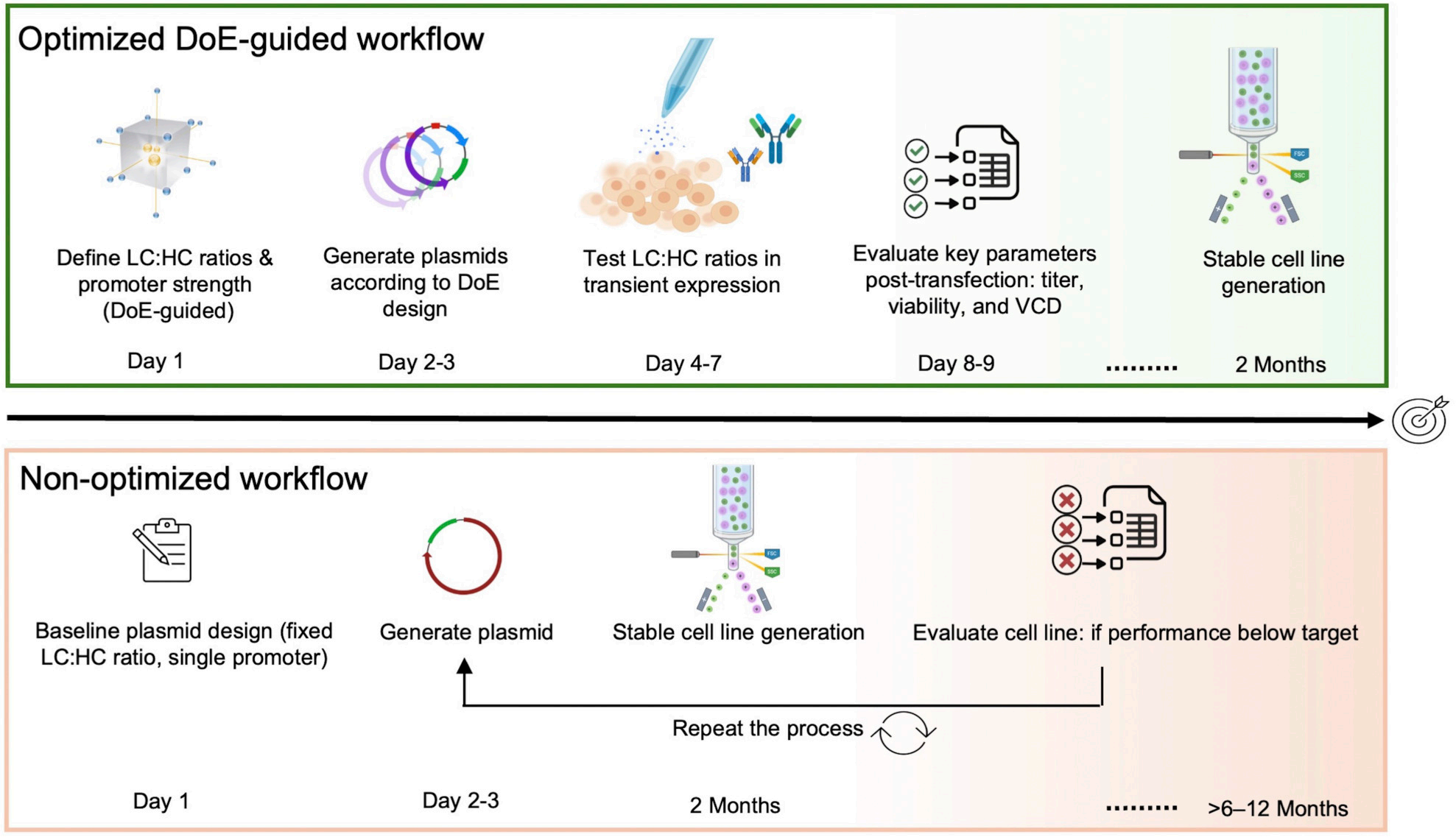
This work demonstrates a microscale, DoE-guided platform that systematically optimizes the expression balance of antibody heavy and light chains by modulating promoters in CHO cells, effectively integrating construct design and bioprocess development into a high-throughput workflow. High-throughput transient screening enables rapid evaluation of the expression performance of multiple construct combinations and early identification of optimal vector designs. This comprehensive approach allows for the generation of stable cell lines within approximately two months. In contrast, conventional, unoptimized workflows rely on baseline vector designs with fixed promoter configurations and expression parameters. Iterative vector redesign and recloning are only required after stable cell line generation when suboptimal performance is identified. This necessitates repeated cell line development cycles, resulting in significantly extended timelines (>6–12 months).