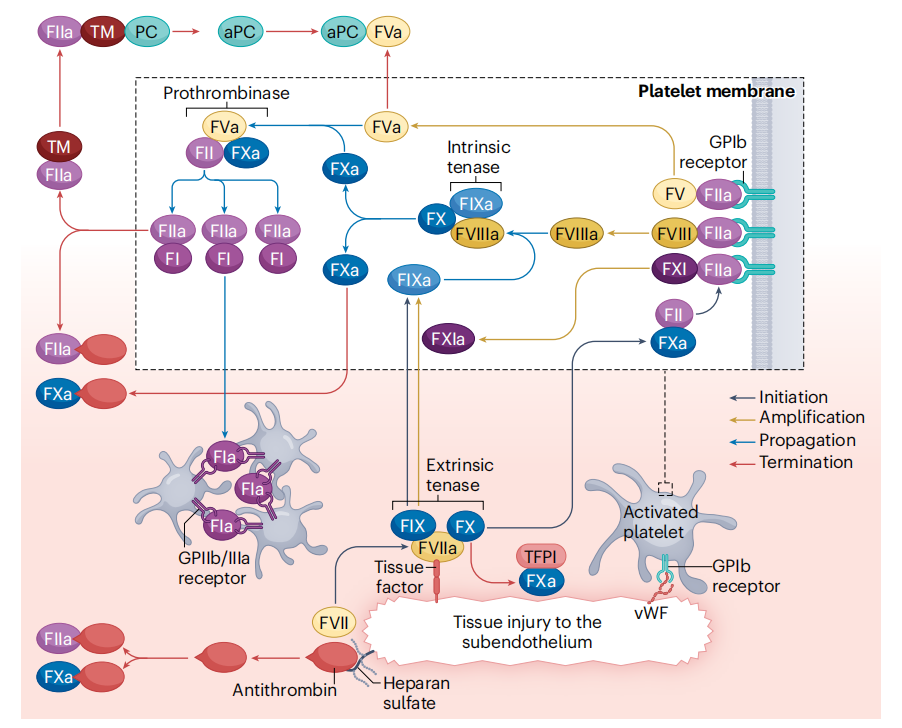
Coagulation factor XI (F11), also known as plasma prothrombin (PTA), or FXI, is a serine protease primarily synthesized in the liver, with lower rates of production in the pancreas and renal tubules. FXI can be activated by thrombin or activated FXII to FXIa. Polyanions, including chromatin from activated neutrophils (neutrophil extracellular trap) or inorganic phosphate polymers released from activated platelets (polyphosphates), can promote thrombin activation of FXII in the thrombotic microenvironment and enhance FXI activation. In cases where blood is exposed to artificial surfaces leading to thrombosis, the primary mechanism of FXI activation is FXIIa.
(Data source: Capodanno D, et al. Nat Rev Cardiol. 2025)
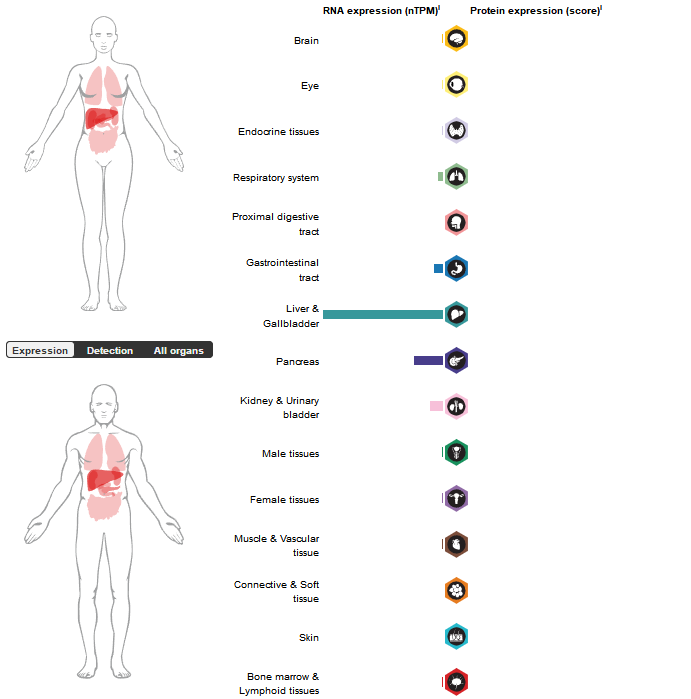
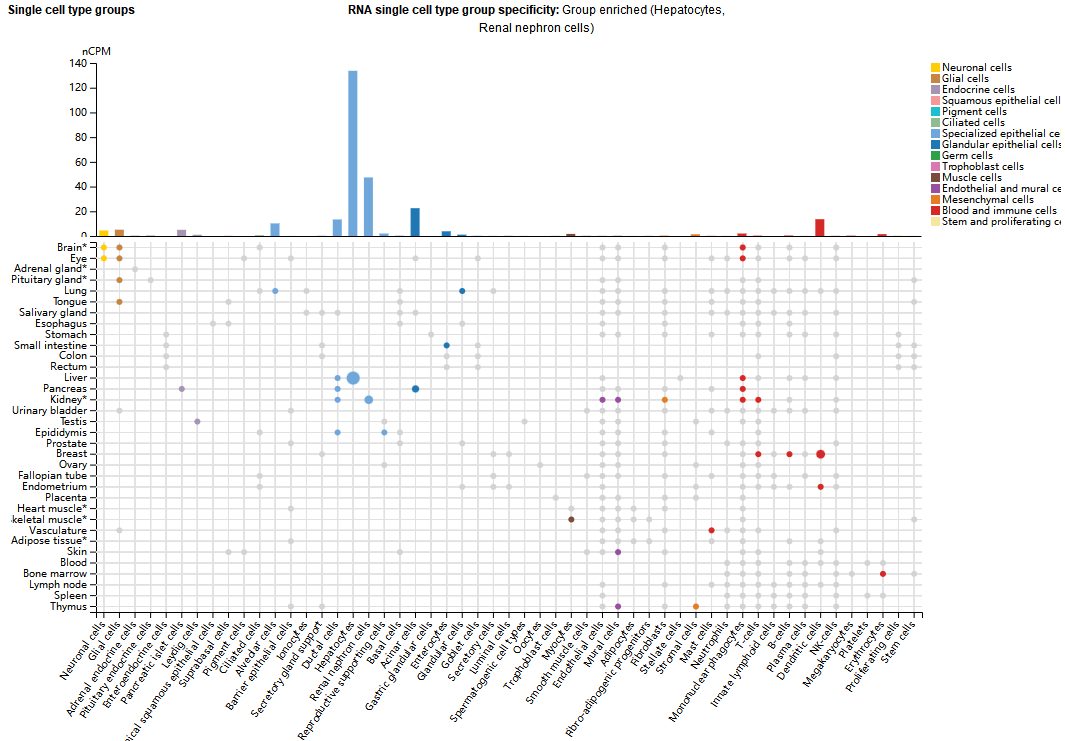
F11 expression distribution
F11 is expression-specific, mainly synthesized in liver tissue, and is also specifically expressed in platelets, kidneys, and pancreatic islet cells, participating in special functions such as local hemostasis.
(Data source: uniprot)
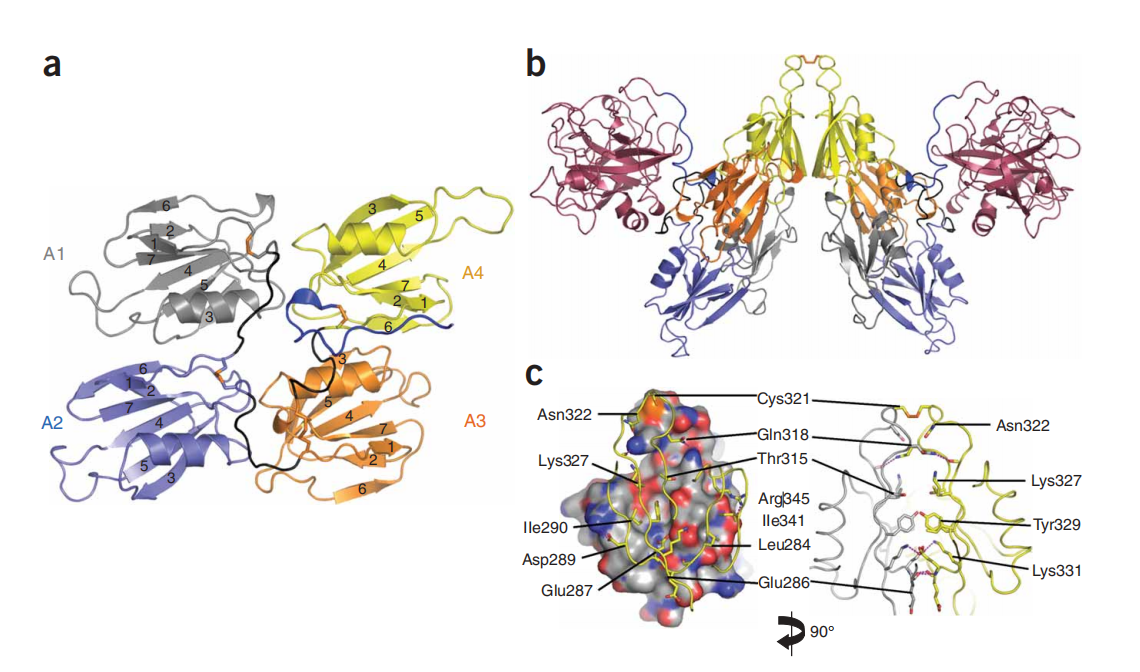
The structure of F11 and its receptor
F11 is a secreted protein, an 80 kDa glycosylated protein composed of 625 amino acids. FXI is a homodimer composed of two identical monomers, a conformation unique among plasma serine proteases. Activation of FXI by thrombin or α-FXIIa produces FXIa, a heavy chain consisting of a four-macro domain (A1-A4) and a light chain of a catalytic serine protease domain covalently linked by disulfide bonds. The macro domains A1 and A2 contain a thrombin binding site and an HK binding site, respectively. A3 contains a FIX, heparin, and GPIb-IX binding site, and A4 contains a cysteine residue that forms the disulfide bond required for FXI dimer formation.
(Data source: Papagrigoriou E, et al. Nat Struct Mol Biol. 2006)
The role of F11 in hemostasis and thrombosis
During hemostasis, thrombi formed via the tissue factor pathway are largely unaffected by FXI activation. This pathway begins with the binding of FVII or its activated form (FVIIa) to extravascular tissue factor at the site of injury. The abundant presence of tissue factor in the perivascular hemostatic environment triggers strong thrombin generation via the FVIIa-tissue factor complex, thereby activating FXI to FXa, which in turn forms thrombin and fibrin. This pathway makes the feedback activation of FXI by thrombin less important for thrombus formation. Furthermore, the FVIIa-tissue factor complex also activates FXI. FXa, along with its cofactor FVIIIa, sustains thrombin generation for hours to days by continuously activating FXI, thus maintaining the coagulation process. This pathway is prominent in severe bleeding disorders such as hemophilia A and hemophilia B, which are associated with inherited deficiencies in factor VIII or factor IX, respectively. Although FXI plays a relatively minor role in physiological hemostasis, its effects are more pronounced in areas with high fibrinolytic activity, such as the oropharynx, nasopharynx, and urinary system. Activated FXIa plays a secondary role in hemostasis (i.e., coagulation) but a key role in thrombosis (i.e., thrombosis propagation).
In the process of arterial and venous thrombosis, the activation of FXI is primarily triggered by thrombin generated via the tissue factor pathway. This thrombin is exposed on the surface of damaged endothelial cells or expressed on various cellular components. Individuals generally lacking FXI have a lower risk of venous thromboembolism and ischemic stroke, while individuals with high plasma FXI levels have a higher risk of venous thromboembolism. FXI is a key player in the pathological thrombosis process, playing a particularly important role in promoting the rapid rupture and subsequent amplification phase of intravascular thrombus growth.

(Data source: Capodanno D, et al. Nat Rev Cardiol. 2025)
Classification and Mechanism of Action of F11 Inhibitors
Several classes of drugs that have been developed or are under investigation targeting FXI and/or FXIa include antisense oligonucleotides (ASOs), monoclonal antibodies, small molecule compounds, natural inhibitors, aptamers, and small interfering RNA (siRNA). Antisense oligonucleotides and siRNA exert their effects by regulating the expression and degradation of target mRNAs in the hepatocyte nucleus; aptamers, small molecules, monoclonal antibodies, and natural peptides act directly on FXI and/or FXIa in the extracellular space; molecules such as FELIAP (inhibitory aptamer of coagulation factor XI), FIIa, FXIa, Ir-CPI (contact inhibitor of castor beetle and tick), and RISC (RNA-induced silencing complex) also have related mechanisms of action.
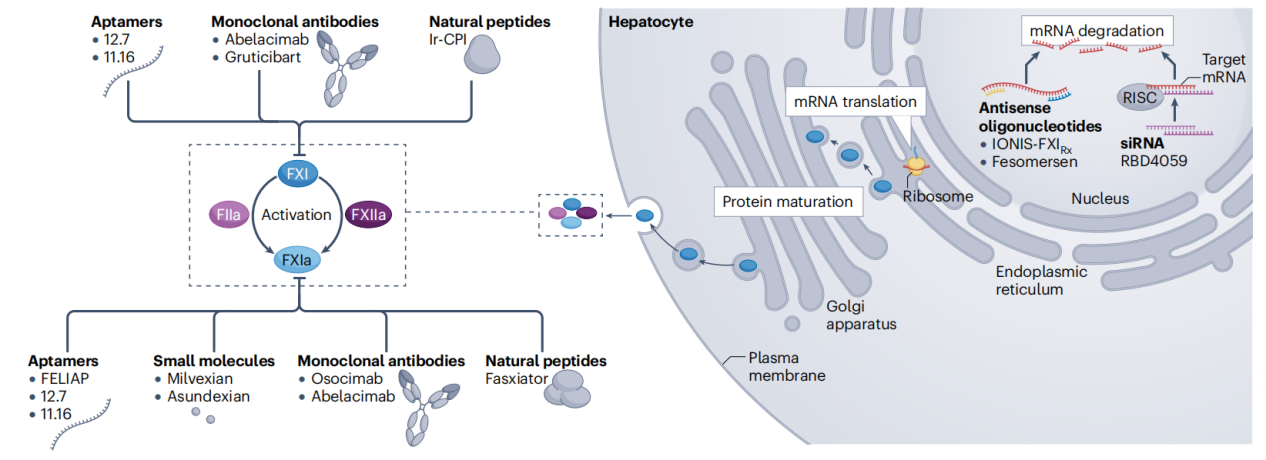
(Data source: Capodanno D, et al. Nat Rev Cardiol. 2025)
Abelacimab is a monoclonal antibody that targets FXI, originally developed by Anthos Therapeutics. In February 2025, Novartis announced its acquisition of Anthos Therapeutics. Abelacimab is a novel, highly selective, fully human monoclonal antibody designed to induce effective hemostasis and anticoagulation by inhibiting factor XI. The AZALEA-TIMI 71 Phase 2 study was an event-driven, randomized, active-controlled, blinded, parallel-group study whose primary endpoint was to evaluate the efficacy of two blinded doses of abelacimab versus open-label rivaroxaban in patients with atrial fibrillation (AF) at intermediate to high risk of stroke. Phase 2 data showed that patients taking abelacimab had a significant reduction in bleeding events compared to AF patients taking standard-of-care direct oral anticoagulants (AZALEA). Compared to rivaroxaban, abelacimab 150 mg showed an approximately 50% improvement in net clinical outcomes. Three Phase 3 clinical trials are underway in patients at risk of arterial and venous thrombosis.

(Data source: Anthos Therapeutics official website)

(Data source: Capodanno D, et al. Nat Rev Cardiol. 2025)
