C3 is a core effector molecule of the complement system, mediating various functions through different binding sites and their corresponding receptors. Complement C3 is the central hub of three complement activation pathways (classical pathway, alternative pathway, and lectin pathway), and plays an important role in immune regulation, inflammation control, vascular diseases, and Alzheimer's disease.

(Data source: Batista AF, et al. Int J Mol Sci. 2024)
C3 expression distribution
Complement C3 is primarily synthesized by hepatocytes in the liver and secreted into the bloodstream in an inactive precursor form under physiological conditions. C3 can also be produced locally by various extrahepatic tissues and cells under inflammatory or pathological conditions. Vascular endothelial cells, epithelial cells of barrier organs (such as the intestine, lung, and kidney), fibroblasts, and immune cells including monocytes, macrophages, dendritic cells, and T lymphocytes have all been found to express C3 in response to pro-inflammatory stimuli such as interleukin-1β (IL-1β), tumor necrosis factor-α (TNF-α), and Toll-like receptor ligands.
(Data source: uniprot)
The structure and composition of C3
C3 is a secreted protein, mainly composed of 1663 amino acids. Complement C3 is a modular protein that functions as a key node in the complement cascade. Its structure consists of a β chain forming a stable macroglobulin (MG) core and an α chain containing regulatory domains such as desensitizing toxin fragments, CUB domains, thioester-containing domains (TED), and C345c domains.
The C3 precursor (Pro-C3) is oxidized during folding, forming 13 disulfide bonds, and is subsequently translated into a complex N-linked sugar at three distinct sites (Asn-85, Asn-939, and Asn-1617). Before being secreted from the cell, pro-C3 is cleaved by furin proteases to produce two distinct polypeptides, commonly referred to as the β chain (residues 23-667; 75 kDa) and the α chain (residues 672-1663; 110 kDa), which remain covalently bound to native C3 (185 kDa) via intermolecular disulfide bonds. Although the mature C3 protein consists of 13 distinct domains, a series of proteolytic reactions result in the formation of various fragments of C3 that differ not only in composition and three-dimensional structure but also in functional properties. C3 can be broken down into C3a, C3b, iC3b, C3c, C3f, etc.

(Data source: Geisbrecht BV, et al. Semin Immunol. 2022)
Activation of complement C3
Complement C3 integrates signals from three classic pathways: the alternative pathway (triggered by pathogens or self-alteration), the lectin pathway (recognizing microbial patterns via lectins), and the classical pathway (via antibody-antigen immune complexes). Complement C3 is cleaved into two major fragments, C3a and C3b , by either the classical/lectin pathway's C3 convertase (C4b2a) or the alternative pathway's C3 convertase (C3bBb). Locally produced C3 outside of hepatocytes further enhances complement activity.
C3a promotes inflammation through G protein-coupled receptors (C3aR) , acting as a potent pro-inflammatory mediator, thereby regulating immune cell activation, chemotaxis, and vascular permeability.
Larger C3b fragments undergo conformational rearrangement, exposing reactive thioester groups within the TED domain. This facilitates covalent binding of C3b to nearby pathogen surfaces, apoptotic cells, or damaged tissue. Surface-bound C3b plays a crucial role in enhancing the complement cascade, promoting C5 convertase assembly, and initiating the formation of the membrane attack complex (MAC). C3b is further processed into iC3b, C3dg, and C3d, which regulate immune responses via complement receptors (CR1-CR4). These receptors retain important roles in phagocytosis, immune complex clearance, B cell co-stimulation, and the promotion of adaptive immune memory through complement receptor binding.


(Data source: Li Y, et al. Clin Sci (Lond). 2025)
The role of C3 in disease
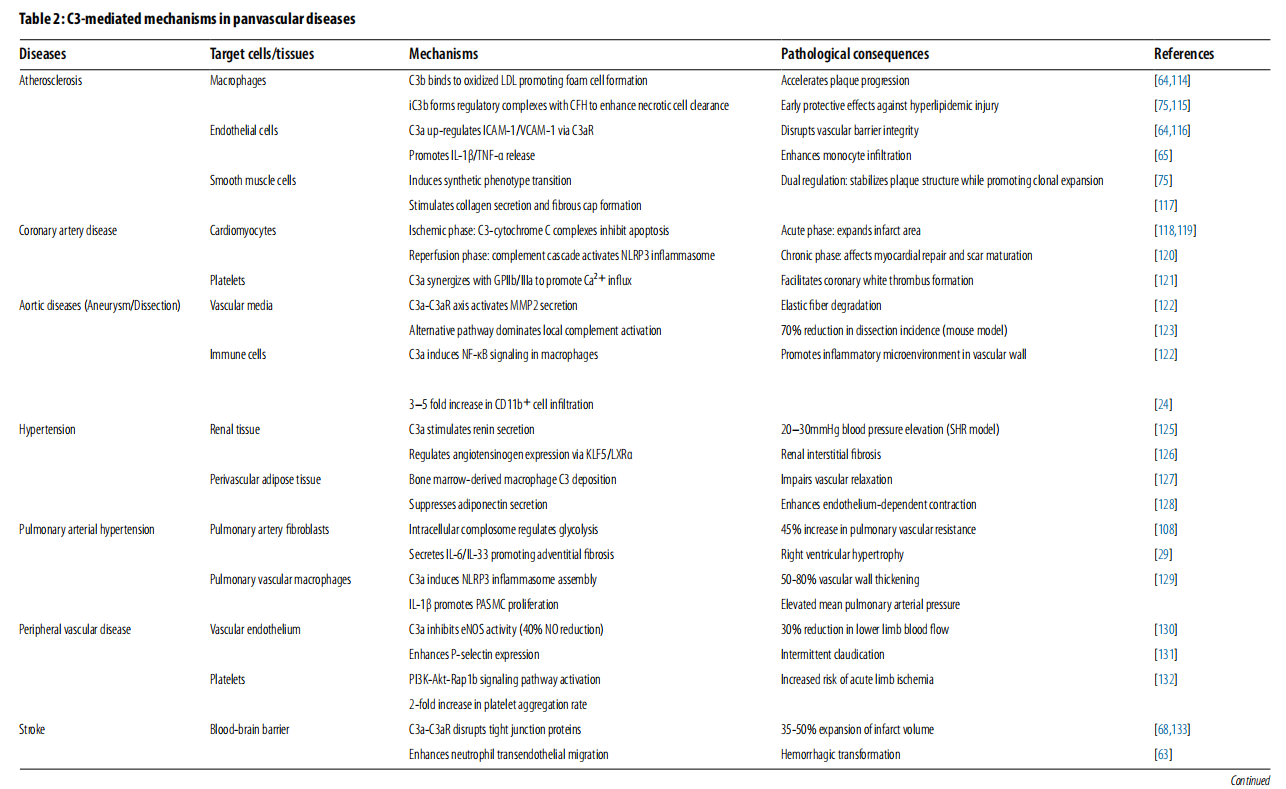
The role of C3 in inflammation and cardiovascular disease
Upon activation, C3 generates effector fragments, amplifies inflammation, and initiates downstream cascade reactions, transforming early immune responses into structural tissue damage. In addition to its classic role in immune defense, C3 has diverse effects on various cell types involved in vascular remodeling, forming a cell-specific regulatory network that controls vascular permeability, immune cell recruitment, and tissue remodeling.
Complement C3 regulates immune-vascular interactions among various cell types. In endothelial cells, C3a/C3aR signaling promotes the production of pro-inflammatory cytokines, leukocyte adhesion, cytoskeleton disruption, and reduced nitric oxide synthesis. In vascular smooth muscle cells (VSMCs), C3a and iC3b drive phenotypic transition, proliferation, and fibrous cap formation, while C5a induces matrix degradation via matrix metalloproteinases (MMPs). In innate immune cells, C3 fragments enhance the recruitment of neutrophils and monocytes, promote CR3-mediated phagocytosis, and polarize M1 macrophages. In adaptive immune cells, intracellular C3 signaling regulates T cell activation and metabolic reprogramming, while C3d enhances B cell co-stimulatory effects. Platelet-bound C3a and C5a promote activation and thrombosis. In fibroblasts, C3a induces myofibroblast transformation and fibrosis, while iC3b supports dendritic cell activation.

(Data source: Li Y, et al. Clin Sci (Lond). 2025)
Complement C3 exhibits a dual effect depending on the stage of the disease. Early activation promotes the production of anti-inflammatory cytokines, polarizes M2 macrophages, and limits downstream C5 activation. In the progressive stage, continued activation drives plaque development, vascular remodeling, lipid metabolism dysfunction, VSMC migration, and inflammation.

(Data source: Li Y, et al. Clin Sci (Lond). 2025)

(Data source: Li Y, et al. Clin Sci (Lond). 2025)
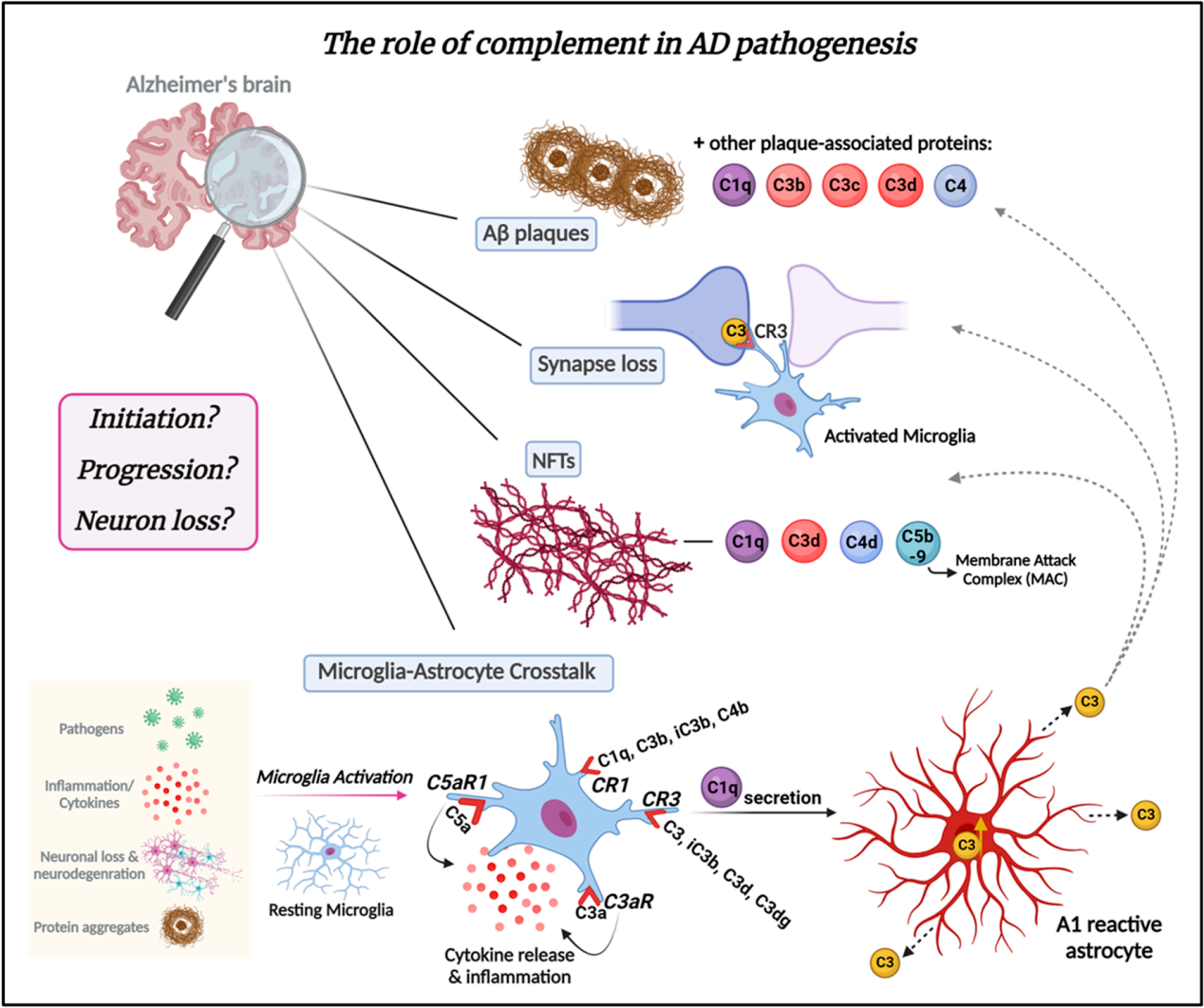
The role of complement C3 in Alzheimer's disease (AD)
The complement system plays a crucial role in normal brain development, aging, and the progression of Alzheimer's disease (AD) by regulating synaptic pruning and elimination, as well as neuronal migration. Complement proteins C1q, C3b, C3c, and C3d are associated with amyloid plaques and degenerative neurites within them throughout the pathogenesis of AD, and synaptic loss is another key feature of AD.

Complement C3/C3b plays a key role in AD by marking weak synapses and sending "eat me" signals, emerging during synaptic elimination. Microglia carry CR3 receptors, recognize marked synapses, and eliminate them through phagocytosis. Complement proteins, such as C1q, C3d, C4d, and C5b-9, are also co-localized with neurofibrillary tangles (NFTs) in AD. Crosstalk between microglia and astrocytes is an important component of AD pathogenesis and progression, and may be mediated by complement. C1q secreted by activated microglia induces A1-responsive astrocytes. A1 astrocytes are characterized by increased C3 expression and secretion. C3 is released from A1-responsive astrocytes and deposits on amyloid plaques, weakened synapses, and NFTs.
C3 has also been identified as a key player during brain development and aging. C3 levels have been shown to increase in the brain and cerebrospinal fluid (CSF) of aging patients and those with Alzheimer's disease (AD), and excessive activation of C3 is associated with neurological damage. During neuroinflammation, C3 may play a crucial role in promoting synaptic injury. C3 is activated after C1q triggers the classical complement cascade, thereby clearing pathogens. Therefore, knocking out or inhibiting C3 (which would suppress all downstream components of complement activation) could be a promising therapeutic target for a variety of diseases, including Alzheimer's disease.
(Data source: Batista AF, et al. Int J Mol Sci. 2024)
C3 targeted therapy
LP-005 is a bifunctional antibody fusion protein developed by Longbio that targets C3b and C5, exhibiting enhanced bioactivity in complement inhibition (CP), antigen inhibition (AP), and complement inhibition (LP) and C3b deposition detection. Simultaneously, LP-005 is engineered to optimize surface charge (PI) and FcRn binding, making it the world's first Q4W bifunctional complement inhibitor. LP-005 is primarily intended for the treatment of paroxysmal nocturnal hemoglobinuria (PNH), complement-mediated kidney disease, and other complement-related disorders.

(Data source: Longbio official website)
CG001 is an antibody fusion protein targeting C3, developed by Shanghai ComGen Biopharmaceutical Co., Ltd. It exerts a cell-targeted complement inhibitory effect at the core C3 level of complement activation. It not only effectively inhibits the activation of both the classical and lectin pathways of complement but also more effectively inhibits the activation of the alternative complement pathway, thereby effectively preventing the production or function of all active complement products, including C3a/C5a, C3b/iC3b, and C5b-9n (MAC). It has shown exceptionally good efficacy in various disease models, including myasthenia gravis, against complement hyperactivation disorders. CG001 has obtained clinical trial approvals from the China NMPA and the US FDA to conduct clinical research.
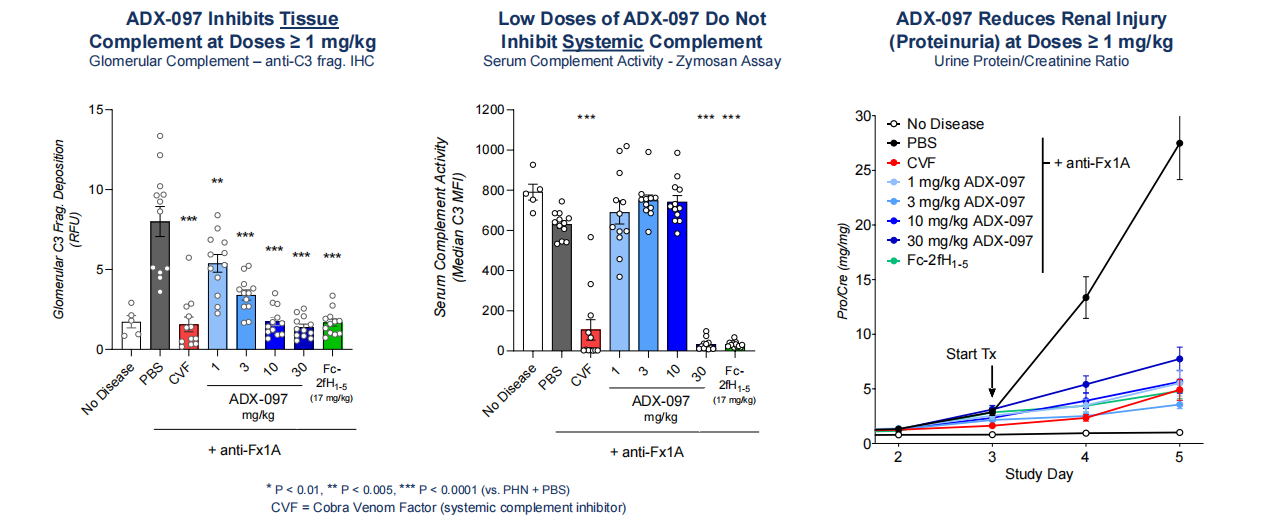
ADX-097, developed by Q32 Bio, is a C3d-targeting fusion protein currently in Phase 1 clinical trials for the treatment of autoimmune diseases and diseases related to anti-neutrophil cytoplasmic antibodies. ADX-097 offers a unique targeted therapy that inhibits the activation of the alternative pathway (AP) in diseased tissue while minimizing systemic complement blockade. ADX-097 is a bifunctional fusion protein containing two portions of the first five shared sequences (fH1-5) of human factor H linked to a humanized anti-C3d antibody. ADX-097 is designed to target diseased tissue by binding to C3d deposited at complement activation sites, providing local blockade. ADX-097 reduces proteinuria and inhibits complement activity in the glomeruli, with dosage not affecting systemic complement activity.

(Data source: Q32 Bio official website)
