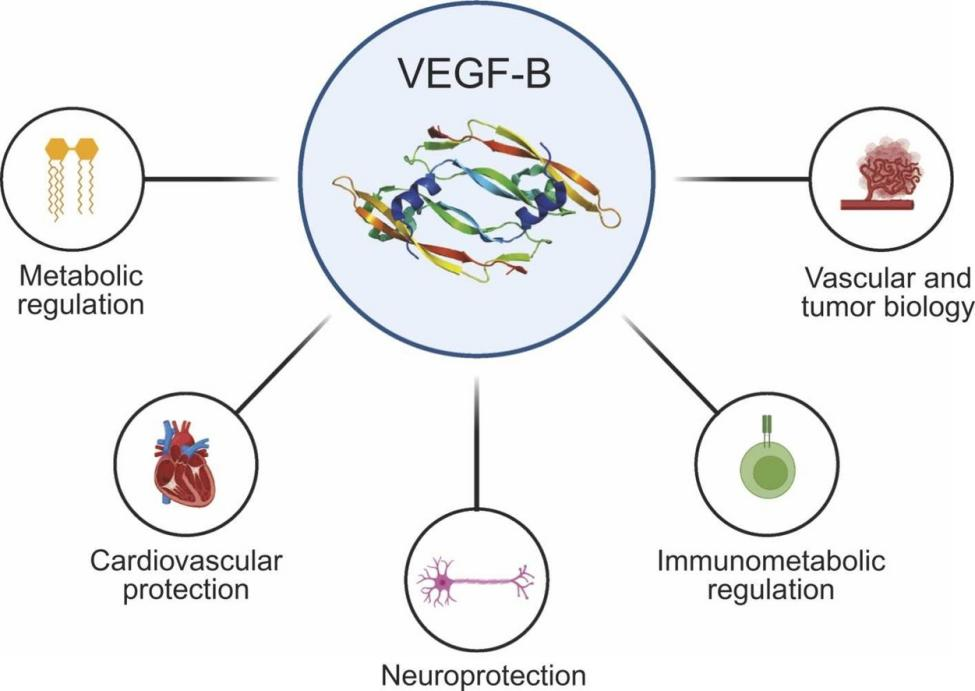
Vascular endothelial growth factor beta-beta (VEGFB) is an endothelial cell growth factor belonging to the VEGF family. The VEGF family comprises five core members: VEGF-A, VEGF-B, VEGF-C, VEGF-D, and placental growth factor (PlGF), which coordinate the development, maintenance, and remodeling of blood vessels and lymphatic vessels. VEGF-B is a key regulator of the cross-talk between angiogenesis, metabolism, and immunity. Unlike VEGF-A, which strongly induces angiogenesis and vascular permeability, VEGF-B's role is more subtle and tissue-specific, such as regulating lipid transport and mediating cellular stress responses without causing vascular leakage. This emerging functional profile highlights the importance of VEGF-B as a molecular mediator in systemic homeostasis and as a promising therapeutic target in diseases such as metabolic disorders, neurodegenerative diseases, cardiovascular diseases, and cancer.
(Data source: Lee C , et al. Pharmacol Res. 2026)
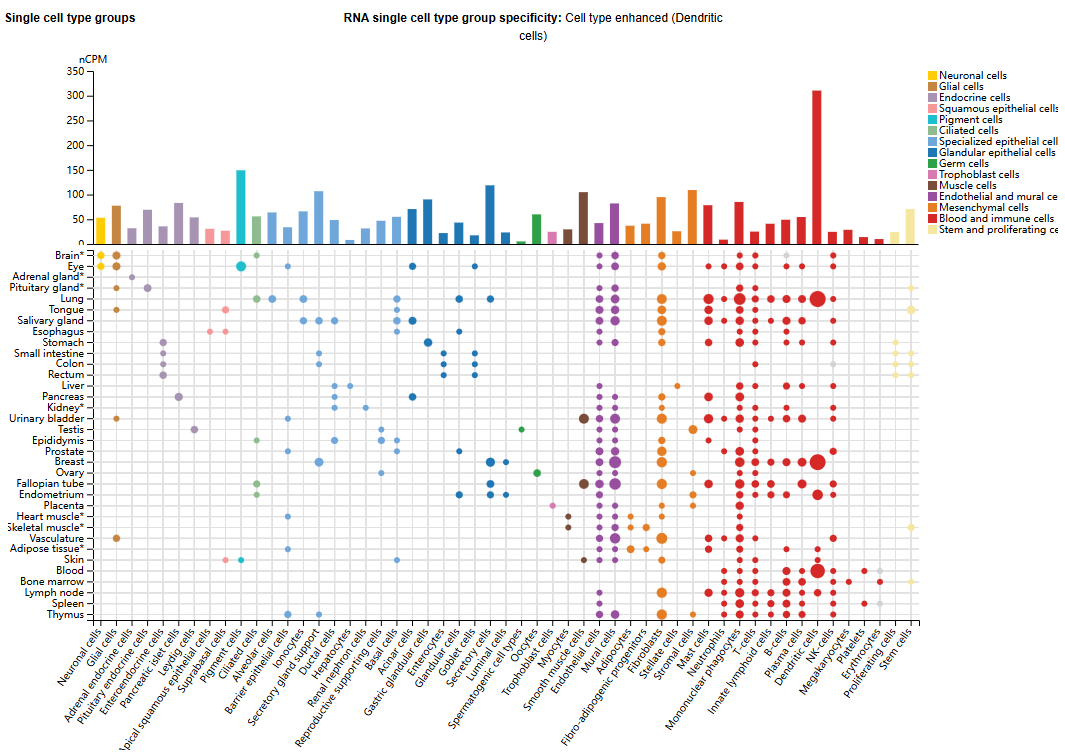
VEGFB expression distribution
VEGF-B is most abundant in metabolically active tissues, including the heart, skeletal muscle, brain, and kidneys, with VEGF-B167 accounting for over 80% of the transcripts. In mouse and human tissues, the transcript levels of VEGF-B167 and VEGF-B186 are comparable, although VEGF-B167 is generally dominant in the heart, skeletal muscle, and brain. VEGF-B186 expression varies across different tissues and tends to increase under pathological or stressful conditions, including tumors and ischemic myocardium.
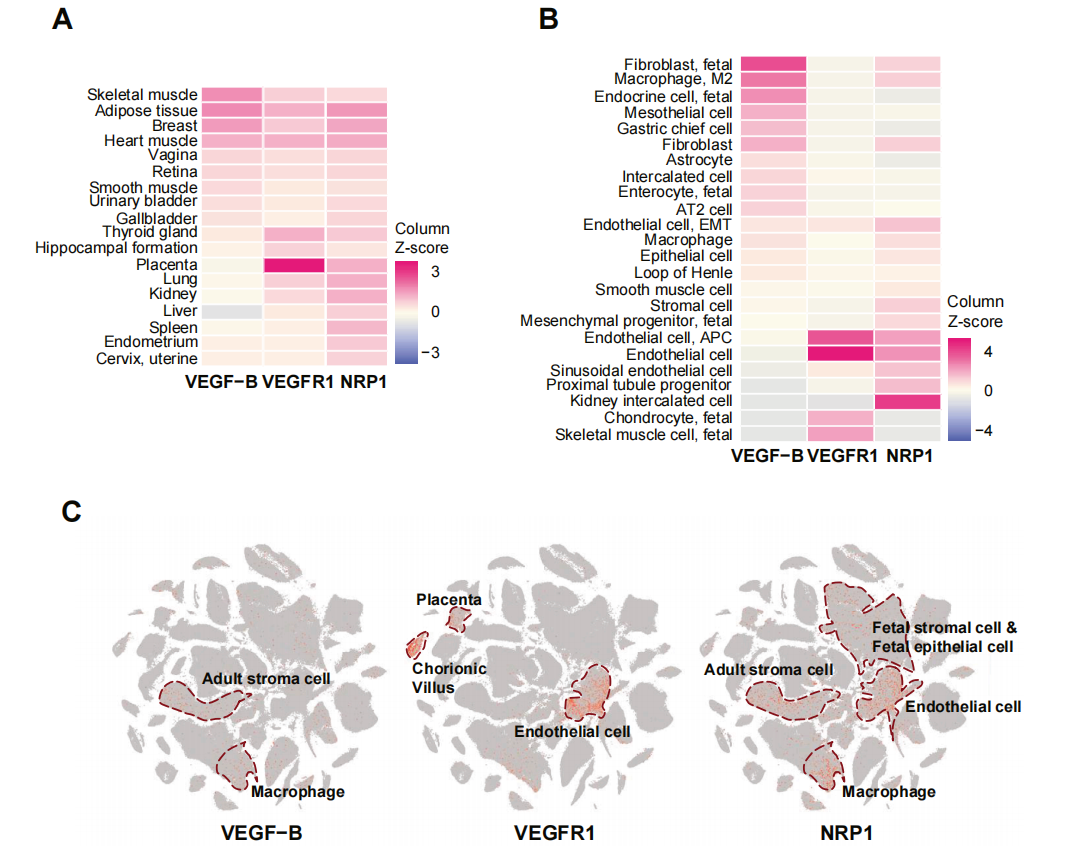
VEGF-B is widely expressed in various cell types, such as astrocytes and intercalation cells, with high expression observed. VEGF-B expression has also been detected in fibroblasts, macrophages, and some fetal cells (such as fetal endocrine cells, fetal fibroblasts, and fetal intestinal epithelial cells).
(Data source: uniprot)
(Data source: Lee C , et al. Pharmacol Res. 2026)
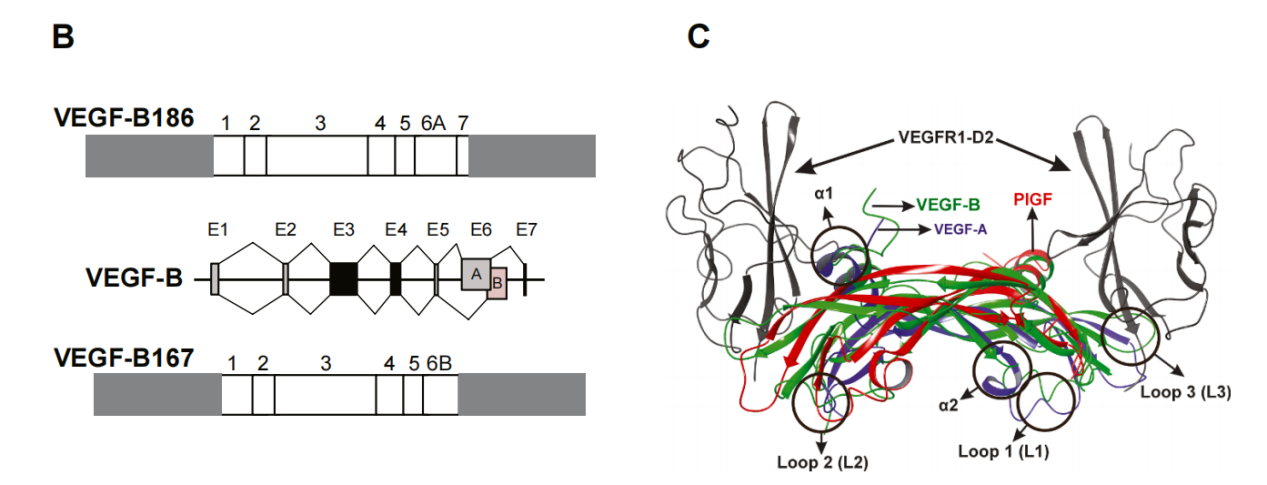
Structure and receptor of VEGFB
VEGF-B is encoded by a seven-exon gene with an exon-intron structure similar to VEGF-A. VEGF-B forms disulfide-linked homodimers (44–54 kDa) and can heterodimerize with VEGF-A . Alternate splicing of exons 6 and 7 generates two major isoforms, VEGF-B167 and VEGF-B186, which have an N-terminal domain (exons 1–5) containing eight conserved cysteine residues for disulfide bond formation and dimerization. This domain represents the VEGF homology domain (VHD), a specific subset of the PDGF homology domain. VEGF-B167 binds to heparin and neurotrophic factor-1, while the binding of VEGF-B186 to neurotrophic factor-1 is regulated by proteolysis.
VEGF-B167 (188 aa) includes C-terminal exon 6B and exon 7, producing a highly basic HBD that is anchored to the ECM and cell surface via heparin sulfate proteoglycans (HSPG). VEGF-B186 (207 aa) instead includes exon 6A, introducing a frameshift to generate a hydrophobic, O-glycosylated C-terminal domain lacking the HBD motif, making it highly diffusive.
(Data source: Lee C , et al. Pharmacol Res. 2026)
VEGF-B primarily interacts with two receptors, VEGFR1 (Flt1 in mice) and neurotrophic factor 1 (NRP1). The D1-D2 construct of VEGFR1 exhibits a higher affinity for VEGF-B than for PlGF, while the D1-D3 construct shows comparable affinity for both ligands. Structural analysis reveals significant differences in their interfaces, despite shared key determinants of VEGFR1 binding between VEGF-A and VEGF-B.
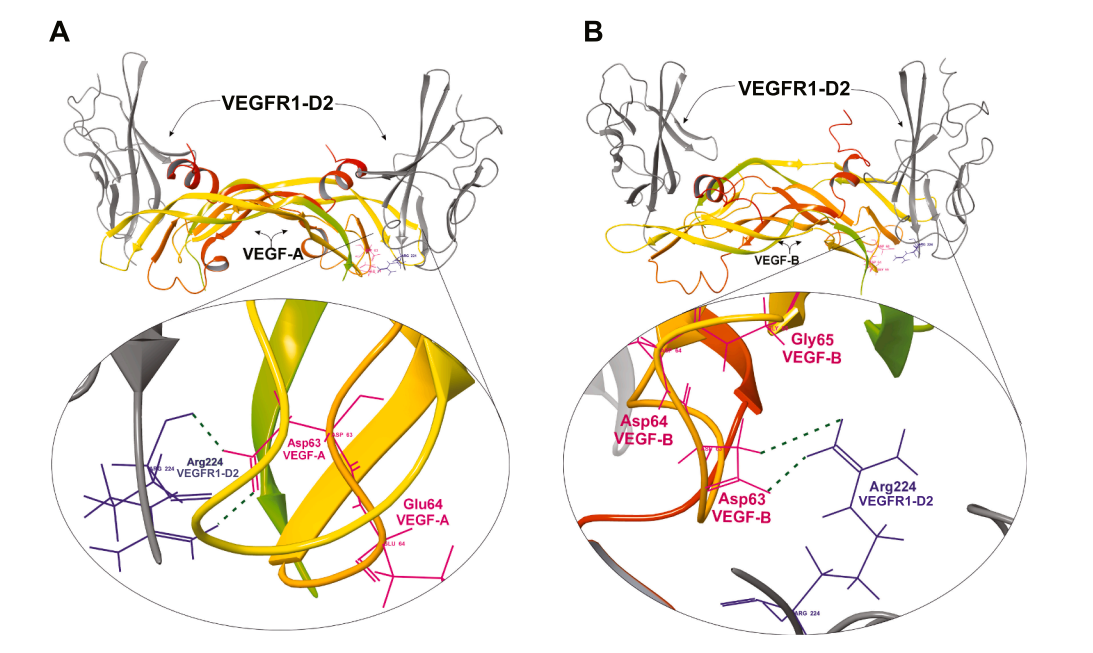
(Data source: Lee C , et al. Pharmacol Res. 2026)
The role of VEGFB
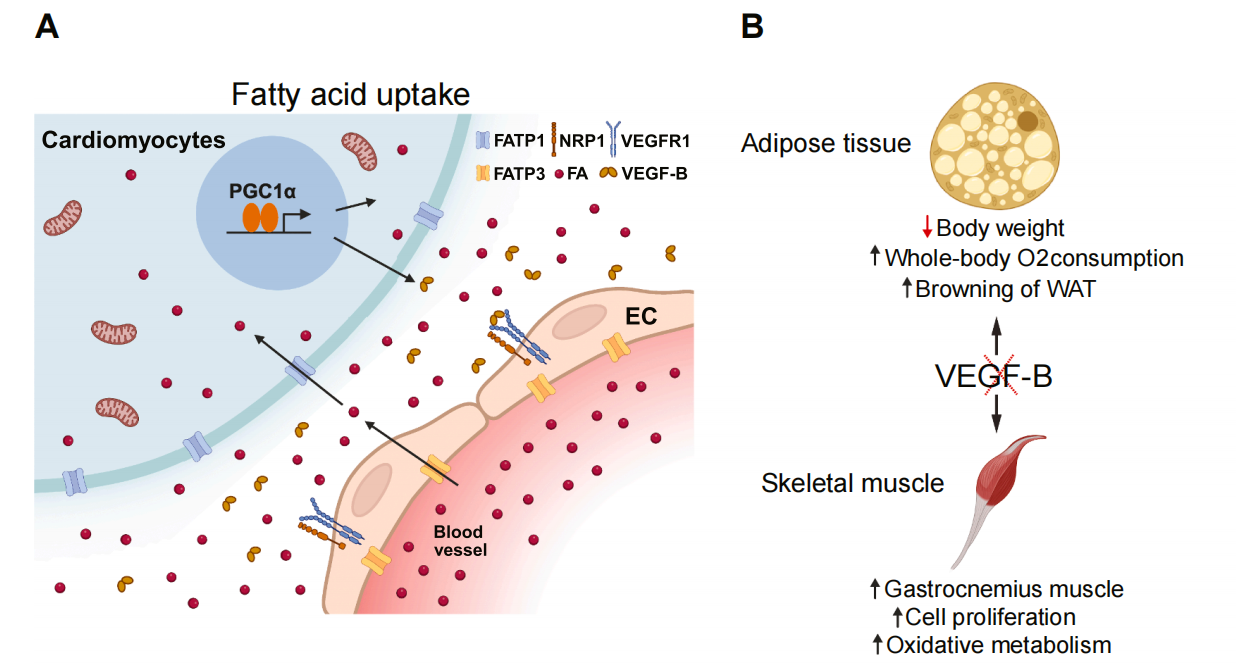
The role of VEGFB in lipid metabolism: VEGFB mediates endothelial cell-mediated fatty acid uptake via VEGFR1/NRP1 signaling. It induces FATP3 on the endothelial membrane, promoting the transfer of fatty acids to adjacent cardiomyocytes. Inhibition of VEGFB is associated with improved skeletal muscle metabolism, white adipose tissue browning, and weight loss. VEGFB/VEGFR1 signaling regulates fatty acid oxidation (CaMKKβ-AMPK-ACC-CPT) and gluconeogenesis (IRS1-PI3K/AKT-FOXO1/GSK3β-PEPCK/G6Pase).
The role of VEGF-B in cardiac function: VEGF-B186 (diffuse form) promotes angiogenesis and metabolic recovery; VEGF-B167 (matrix-bound form) maintains local nutrient signal transduction. VEGF-B186 promotes the proliferation of subendothelial capillaries in the heart of aged mice and improves functional recovery while reducing fibrosis. VEGF-B167 maintains heart quality but can induce vascular leakage under chronic load and adverse hypertrophy.
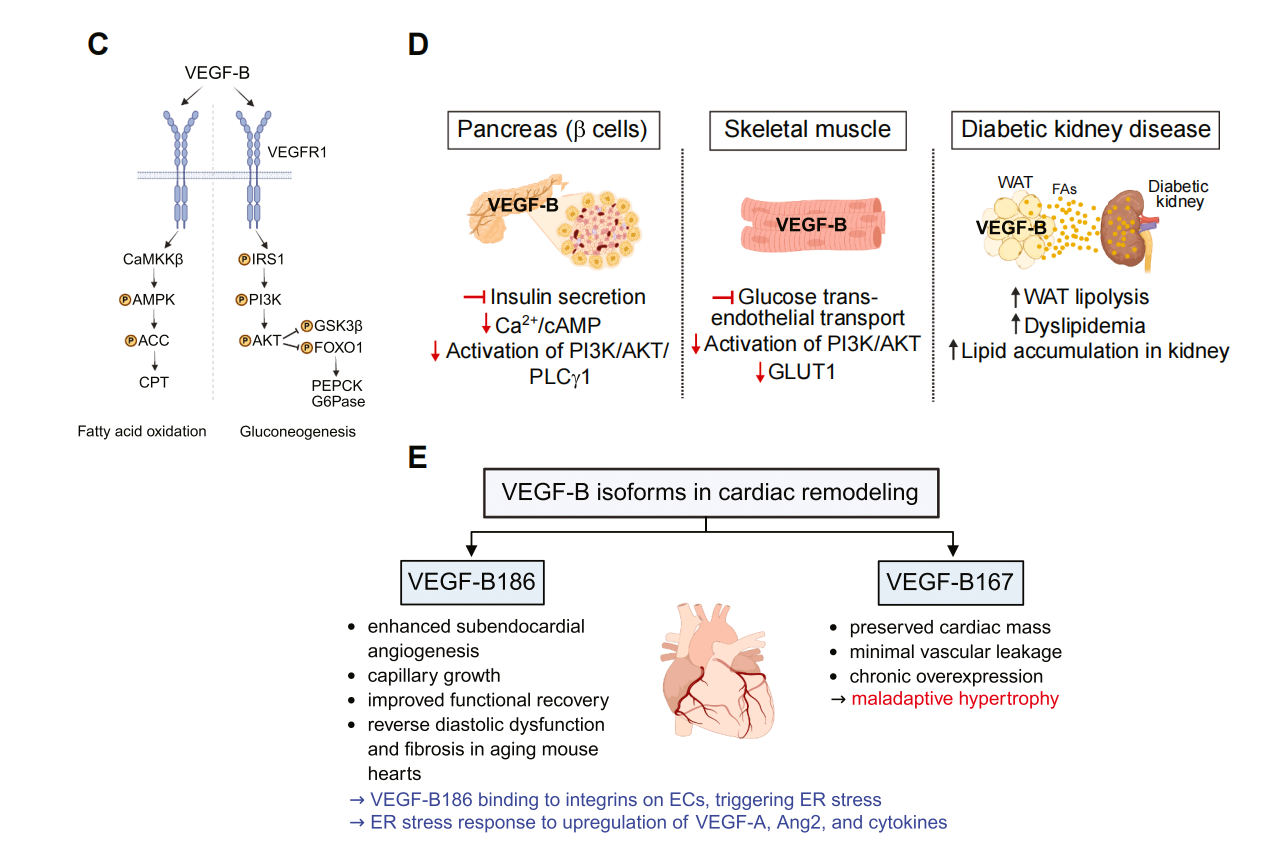
(Data source: Lee C , et al. Pharmacol Res. 2026)
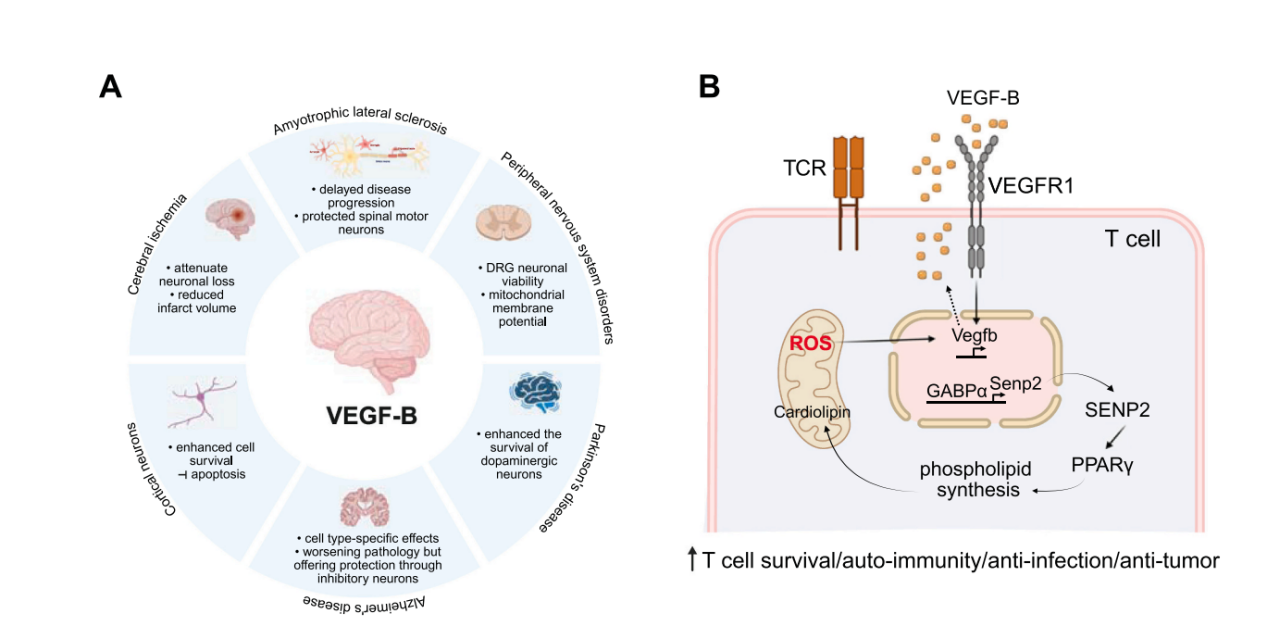
The Role of VEGF-B in Neuroprotection: VEGF-B is a key neurotrophic factor with broad significance for neuronal survival in neurodegenerative diseases, making it a therapeutic target for neuroprotective therapies. Mediated by VEGFR1 and NRP1, these receptors activate the survival-promoting PI3K/AKT and ERK1/2 signaling pathways. These cascades inhibit apoptosis and promote neuronal survival under stress. In cultured cortical neurons, VEGF-B enhances survival in a dose-dependent manner under hypoxic conditions. In primary spinal motor neuron cultures, the VEGF-B186 isoform protects against degeneration through VEGFR1-dependent kinase activity without angiogenesis. This low permeability provides an advantage for central nervous system applications.
The role of VEGF-B in T cells: VEGF-B binds to VEGFR1, affects the GABPα and SENP2 signaling pathways, regulates phospholipid synthesis through PPARγ, and enhances T cell survival, autoimmunity, anti-infection and anti-tumor responses by regulating reactive oxygen species (ROS) and cardiolipin metabolism.
The role of VEGF-B in diabetic retinopathy: VEGF-B exacerbates vascular leakage in the retina, involving the CDC42, ZO1, and VE-cadherin signaling pathways, thereby exacerbating the pathological process of the disease.
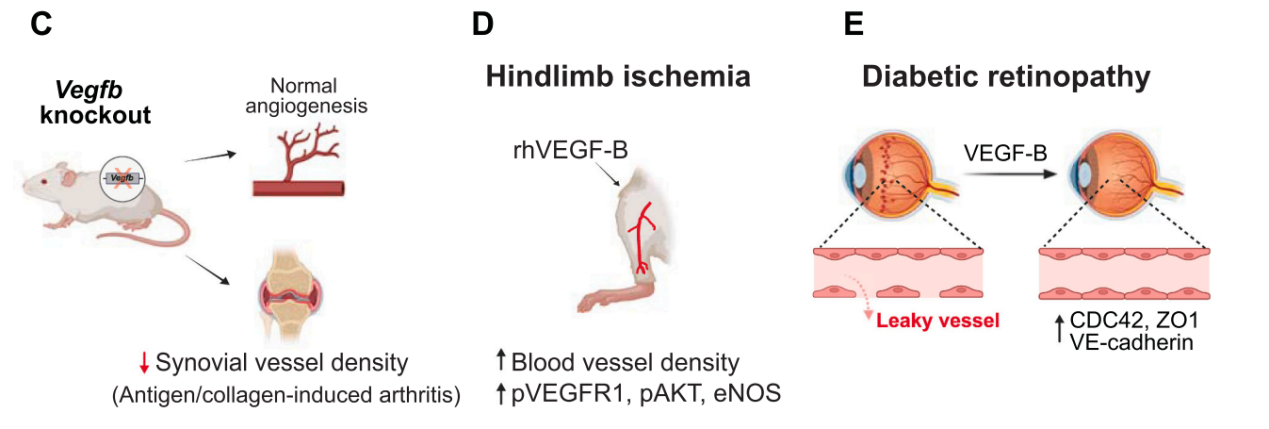
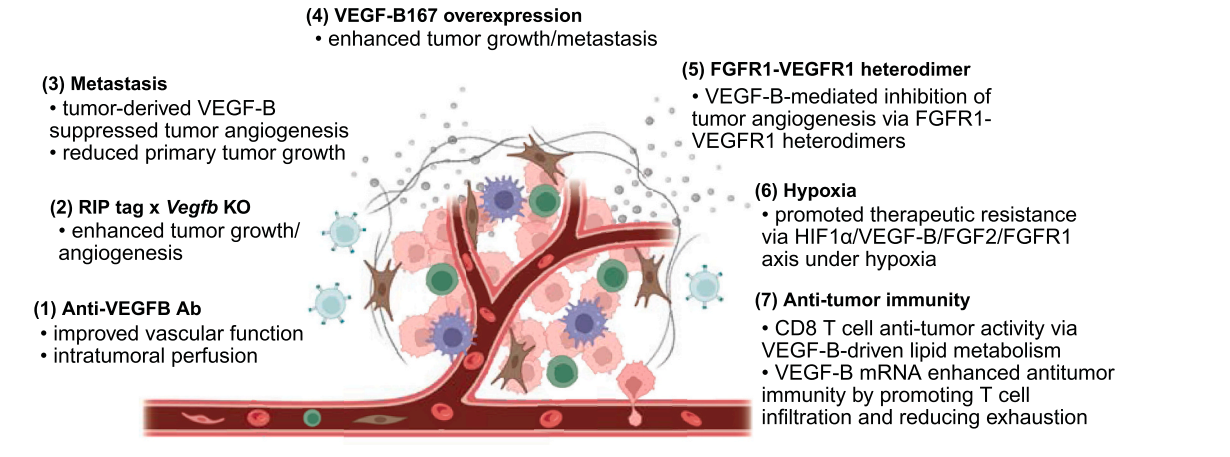
The role of VEGF-B in tumors: Tumor-derived VEGF-B inhibits angiogenesis and limits tumor expansion. Overexpression of VEGF-B167 promotes metastasis. Furthermore, FGFR1-VEGFR1 heterodimerization inhibits angiogenesis synergistically with VEGF-B, FGF2, and the hypoxia signaling pathway. VEGF-B-driven lipid metabolism in hypoxic tumors contributes to treatment resistance. Finally, VEGF-B mRNA has been reported to enhance CD8 + T cell-mediated antitumor immune responses by preventing T cell exhaustion.
(Data source: Lee C , et al. Pharmacol Res. 2026)
Targeted therapy for VEGFB
Reflocibart (CSL-346) is a monoclonal antibody targeting VEGFB for the treatment of endocrine and metabolic diseases, as well as urogenital diseases. The highest stage of its development is Phase 2 clinical trials, and there are currently no new research updates.
EB-105 is the world's first trispecific antibody to enter clinical trials for the treatment of retinal diseases. It contains three functional units that inhibit VEGF-A, VEGF-B, PlGF, angiopoietin-2 (Ang-2), and IL-6R, which can lead to vascular leakage, angiogenesis, and retinal inflammation. EB-105's first indication is diabetic macular edema (DME). EB-105 is wholly developed and owned by Diagnostics.
