The complement system is central to the first line of defense against invading pathogens. However, excessive activation of complement and/or loss of complement regulation contribute to the development of autoimmune diseases, systemic inflammation, and thrombosis. The alternative complement pathway, one of the three pathways of the complement system, plays a crucial role in amplifying complement activation and signal transduction. Complement factor D (CFD) is a serine protease in this pathway and is the rate-limiting enzyme required for the formation of C3 convertase. Activated CFD cleaves factor B (CFB) upon binding to complement C3b, activating the C3 convertase in the alternative pathway and initiating the amplification loop of the complement cascade. CFD is an adipokine secreted by adipose tissue that participates in metabolic regulation and plays a complex and critical role in disease.
(Data source: Barratt J, et al. Front Immunol. 2021)
CFD structure
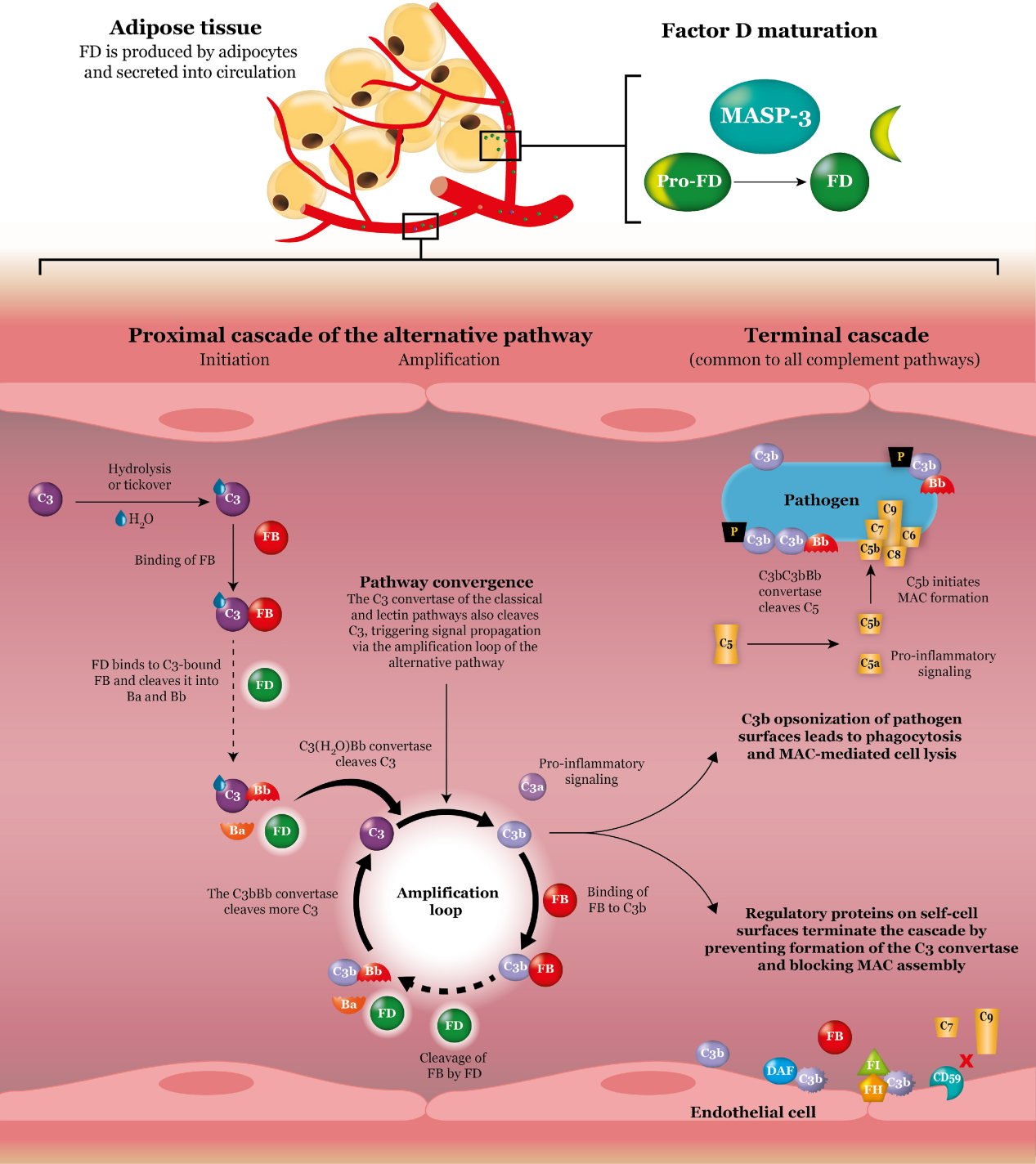
CFD is a secreted protein with a molecular weight of 24.4 kDa. The immature CFD has two variants: variant 1 consists of 251 amino acids, including an 18-amino acid leader sequence; variant 2 consists of 260 amino acids, including a 25-amino acid leader sequence. Except for the propeptide, these two variants have the same polypeptide chain and form the same mature pre-CFD. As an inactive zymogen, pro-CFD consists of 235 amino acids and is activated by cleavage by serine protease-3 (MASP-3), a lectin pathway-associated protein, to form the mature CFD. The activated CFD, with 228 amino acids, consists of a single serine protease domain (SPD) containing a catalytic triad (His41, Asp89, and Ser183).

(Data source: Sekine H, et al. Immunol Rev. 2023)

(Data source: Alphafold)
The role of CFD in the complement system
First, CFD is responsible for the cleavage of C3-bound CFB to generate C3(H2O)Bb convertase, which breaks down C3 into C3b and the pro-inflammatory signaling allergen C3a. Second, CFD is responsible for the cleavage of C3b-bound CFB in the amplification loop to generate the major C3 convertase C3bBb, which amplifies the signal and produces more C3b and C3a molecules. The classical complement pathway and the lectin complement pathway converge at the amplification loop of the alternative pathway, thus their signals are also amplified by CFD. In addition to being integrated into the C3 convertase complex as part of signal amplification, C3b also plays a role in coating cells for phagocytosis and initiating MAC formation and cell lysis through subsequent reactions. The inhibitory effects of these activities on the cell surface are controlled by a series of regulatory proteins.

(Data source: Kong Y, et al. Front Immunol. 2024)
The role of CFD in disease
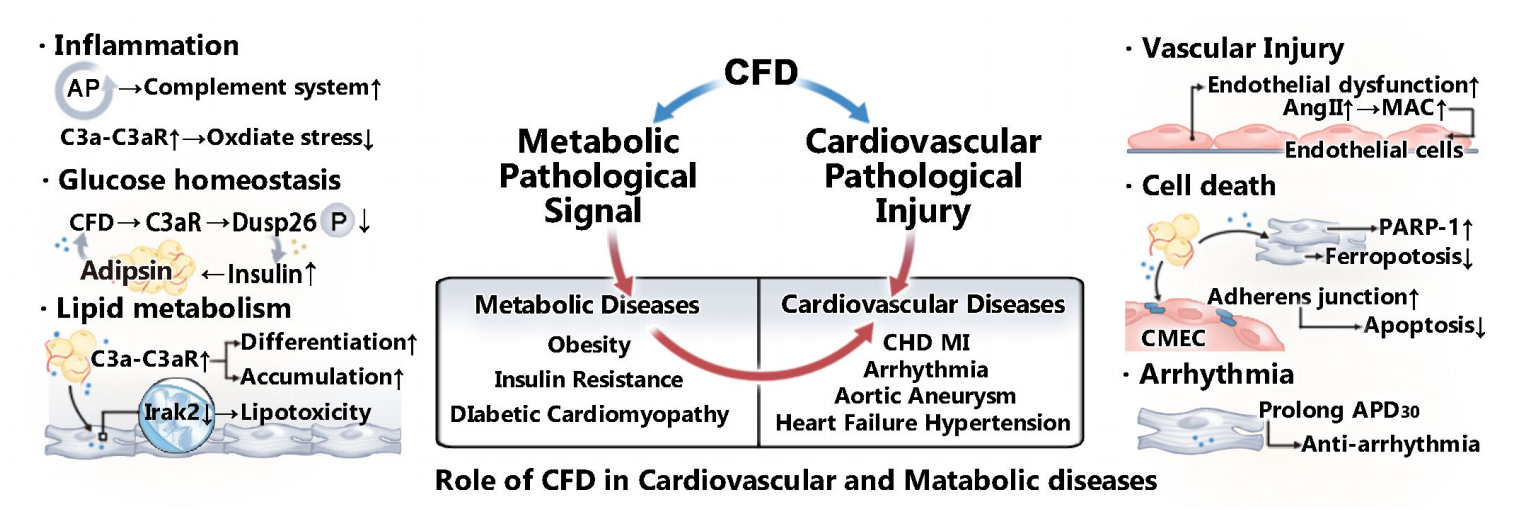
CFD is both a key activating enzyme of the complement substitution pathway (synergistically with MASP-1, C3, and C5) and an important adipokines regulating lipid metabolism and glucose homeostasis. It participates extensively in the progression of various diseases by activating the complement cascade and regulating metabolic pathological signals.
(Data source: Kong Y, et al. Front Immunol. 2024)
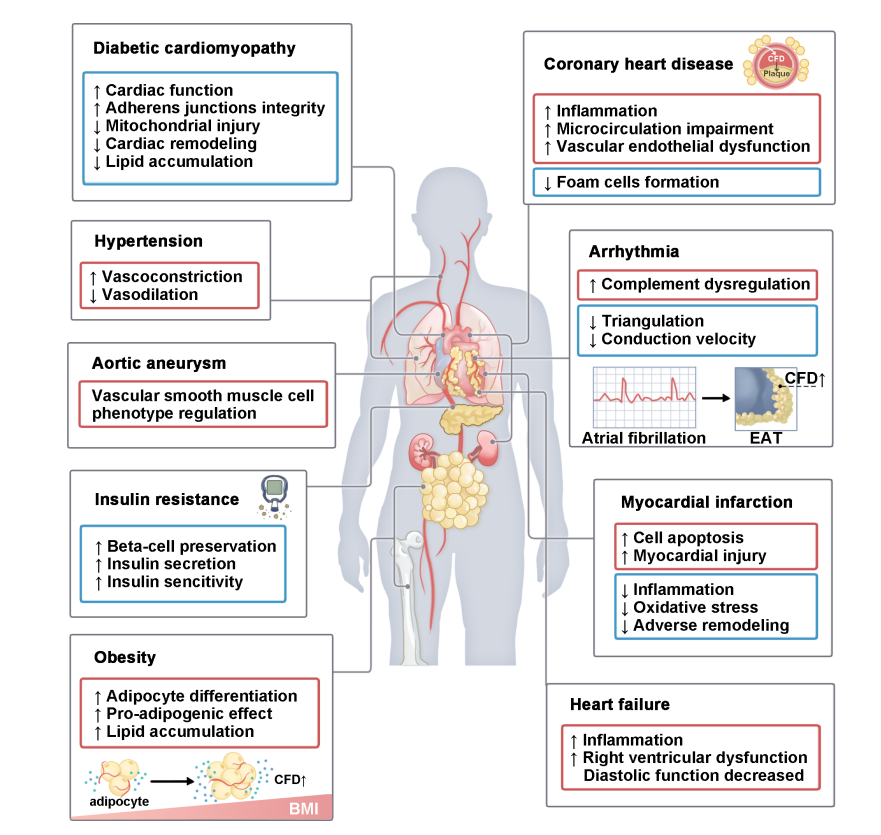
CFD, along with MASP-1, C3, and C5, participates in the hemostatic response. While playing a protective role in preventing bleeding, it can also exacerbate thrombotic and inflammatory diseases. Regarding lipid metabolism, although CFD promotes foam cell formation and exacerbates vascular lesions, it can inhibit lipotoxicity and protect cardiomyocytes from ferroptosis. CFD has a dual role in regulating cell fate: it can maintain tight junctions in microvascular endothelium by inhibiting oxidative stress, but it may also induce cardiomyocyte apoptosis through excessive activation of PARP-1. CFD plays an important role in various cardiovascular diseases, including diabetic cardiomyopathy, hypertension, aortic aneurysm, insulin resistance, obesity, and coronary heart disease. In some diseases, CFD has both pathogenic and protective effects.

(Data source: Kong Y, et al. Front Immunol. 2024)
CFD-targeted therapy
Many CFD inhibitors have been designed, and a significant portion of them have entered clinical trials. Due to their unique mechanisms, CFD inhibitors may achieve even more profound clinical benefits in the future.
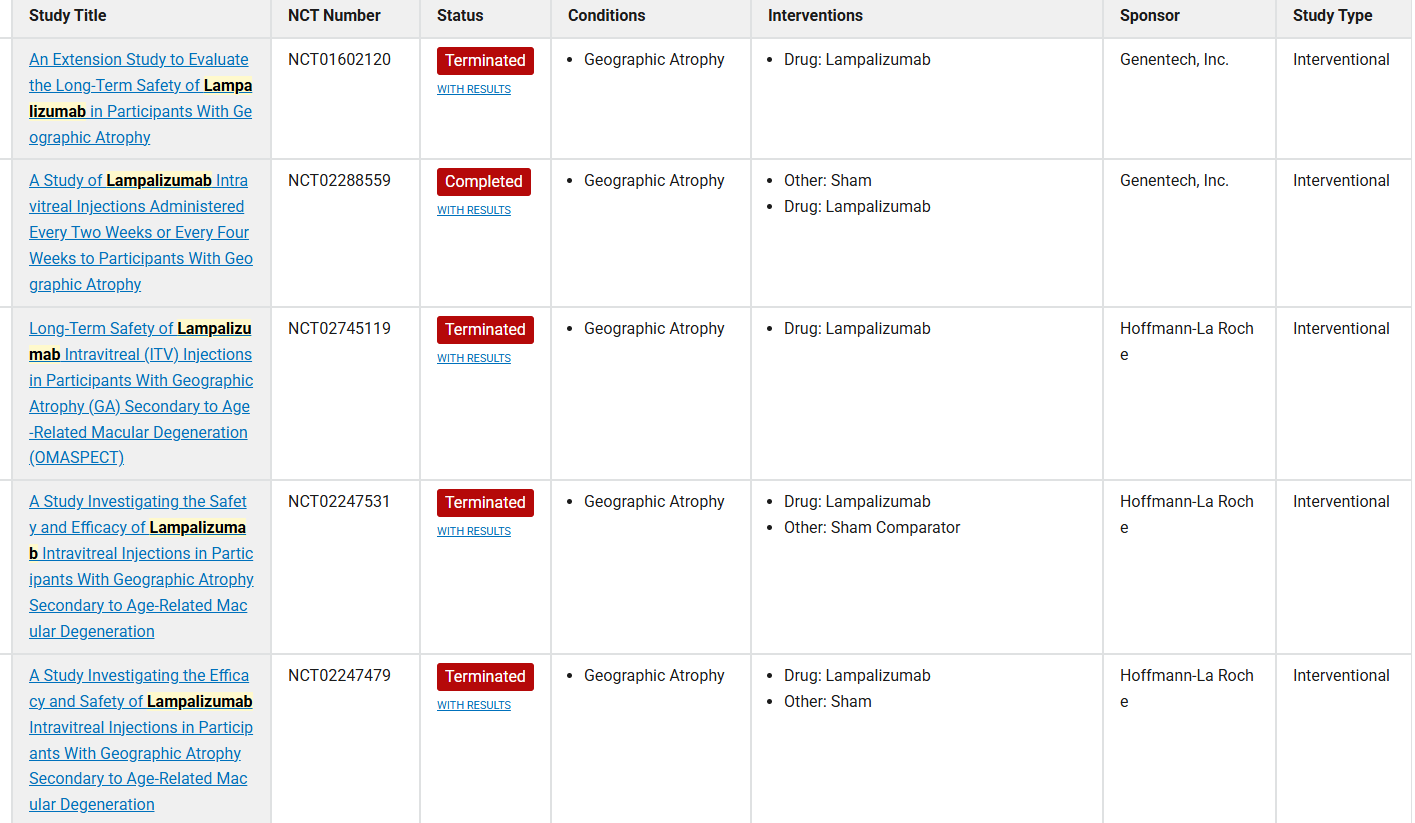
Danicopan (ALXN2040) is an oral complement factor D inhibitor primarily used as adjunctive therapy in patients with polymyxin B (PNH) (NCT04469465). Danicopan increased hemoglobin concentrations, and no serious adverse events were reported in the studies. Furthermore, Danicopan is currently undergoing a Phase II clinical trial in patients with general glomerulonephritis (GA) (NCT05019521). Additionally, BCX9930 is another oral complement factor D inhibitor used to treat PNH (NCT04702568) and C3 glomerulonephritis (NCT05162066).
Currently, only one anti-CFD antibody is being used in clinical trials to treat diseases mediated by alternative pathways. Lampalizumab, a humanized monoclonal antibody targeting a CFD antigen-binding fragment, showed promising results in a phase II clinical trial administered intravitreally to patients with geographic atrophy (GA) secondary to age-related macular degeneration. However, it failed to reduce GA enlargement in two phase III randomized clinical trials, leading Roche to officially terminate further development plans for Lampalizumab for geographic atrophy. Future research into new antibody treatment strategies is necessary.
(Data source: clinicaltrials)
