Coagulation factor IX (FIX), also known as F9, is a vitamin K-dependent plasma protein and a serine protease synthesized by hepatocytes. It participates in the intrinsic blood coagulation pathway. FIX exists in proenzyme form, and its activated form, FIXa, plays a crucial catalytic role in the endogenous tenase complex with the assistance of cofactor FVIIIa. Within this complex, FIXa specifically activates coagulation factor X (FX) to convert to FXa. Upon activation, FIXa forms a complex with calcium ions, membrane phospholipids, and coagulation factor VIIIa, thereby activating coagulation factor X. FIX deficiency leads to impaired thrombin production, preventing the efficient conversion of soluble fibrinogen into insoluble fibrin network, thus causing the bleeding tendency characteristic of hemophilia B.

(Data source: Chowdary P, et al. Lancet . 2025)
FIX Structure
F9 is a single-chain glycoprotein with a molecular weight of approximately 55-57 kDa, composed of 415 amino acid residues. It consists of 8 exons and 7 introns. The mature form of FIX exists in plasma and is mainly composed of a signal peptide; a propeptide and γ-carboxyglutamate (Gla) domains; the remainder of the Gla domain and a short hydrophobic, two epidermal growth factor (EGF) domains; a linker region; an activating peptide; and a C-terminal serine protease domain.

(Data source: Bos MHA, et al. Bos MHA. 2024)
The Gla domain: Contains 12 γ-carboxyglutamate (Gla) residues. The first 11 Gla residues are highly conserved with other vitamin K-dependent proteins and are essential for function.
EGF domains: Two tandem EGF-like domains, with the EGF1 domain containing a β-hydroxyaspartic acid (Hya) residue, which is also a vitamin K-dependent modification site. They are primarily responsible for protein-protein interactions. The EGF domains participate in the binding of FIX to coagulation cofactor FVIIIa and may interact with endothelial cell surface receptors, regulating FIX clearance and localization.
Linker region: The linker region (126–145 aa) connects the second EGF domain (EGF-2) to the activating peptide.
Activated peptide: The activated peptide is cleaved and removed from two specific sites during proteolysis: between residues 145-146 and 180-181, by the TF-FVIIa complex and FXIa. Therefore, the activated peptide is not present in the active form of the enzyme FIXa.
The protease domain: The main function is to provide a binding surface for cofactor FVIIIa and substrate FX, and to catalyze the hydrolysis of specific peptide bonds in FX. Structurally, the protease domain consists of two β-tubular structures, each occupying approximately half of the entire structure. It exhibits a typical catalytic triad (His221, Asp269, Ser366).
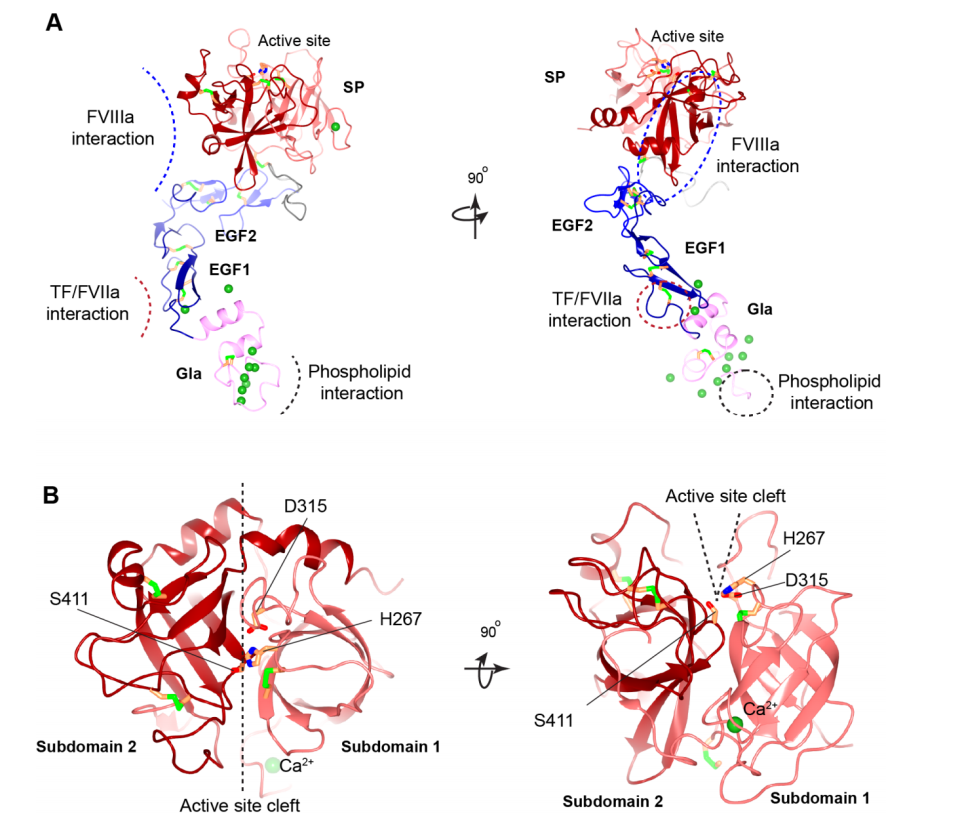
(Data source: Shen G, et al. Int J Mol Sci . 2022)
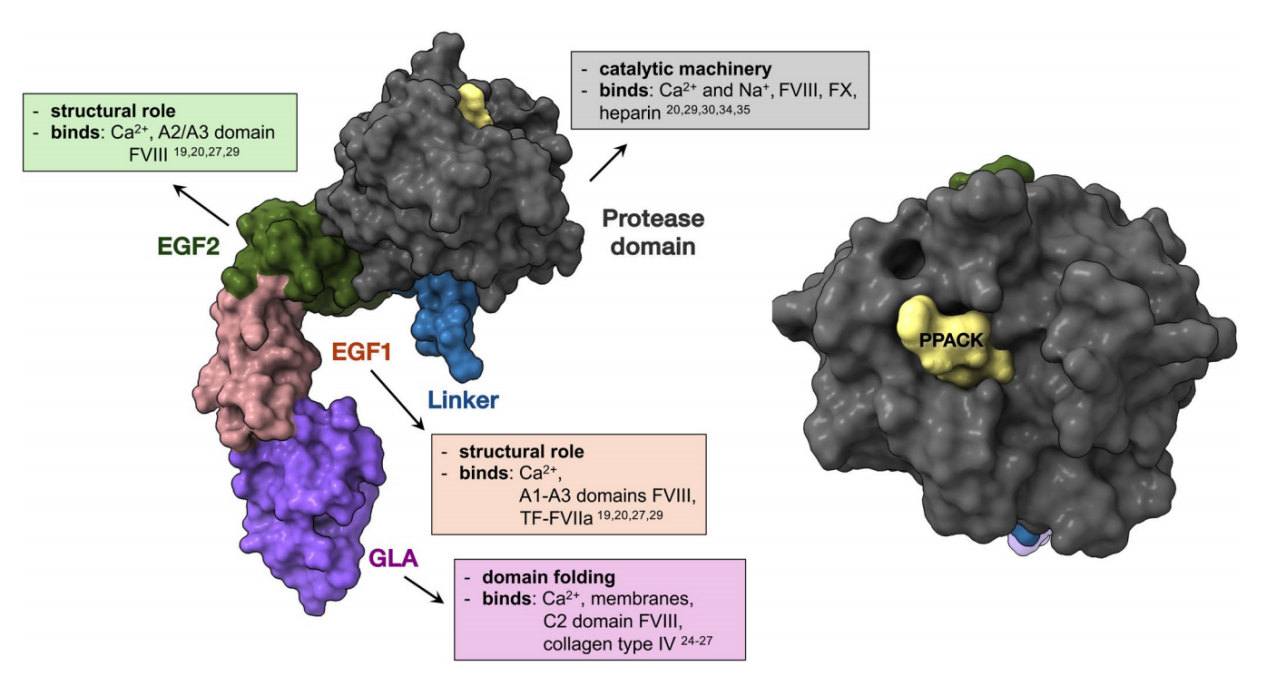
(Data source: Ivanciu L, et al. IUBMB Life. 2025)
Targeted therapy for FIX
Emicizumab is a bispecific antibody developed by Chugai that targets FIX. It binds to the EGF1 domain of FIXa and the EGF2 domain of FXa, thereby co-localizing the two proteins. It has been effectively used for the prophylactic treatment of hemophilia A patients with or without inhibitors.
NXT007 is a next-generation bispecific antibody based on Hemlibra (Emicizumab), a widely used treatment for hemophilia A, and is being developed as a subcutaneous treatment for hemophilia A. The NXTAGE study was a multicenter Phase I/II study evaluating the safety, pharmacokinetics, pharmacodynamics, and efficacy of NXT007. NXT007 was well tolerated during the switch from emecizumab. No thromboembolic events were observed, the frequency of adverse events did not increase in a dose-dependent manner, and there were no adverse events leading to discontinuation or serious adverse events related to NXT007. Roche decided to introduce this investigational drug in August 2022. Currently, Phase I/II clinical trials for hemophilia A are underway, with three Phase III clinical trials planned to begin in 2026.
Mim8 is currently in clinical trials. Mim8 can bind to both FIXa and FXa, but its binding site differs from that of Emicizumab. The FIXa-binding arm of Mim8 binds to FIXa within the protease domain. The FX-binding arm of Mim8 covers a large portion of the FX region, including EGF2 and parts of the protease domain. Mim8 not only co-localizes FIXa and FX but also enhances the catalytic activity of FIXa. The FIXa-binding arm of Mim8 binds to the FIXa peptide (169c-174c) adjacent to the 99c-ring, which is close to the FvIII binding site at 162c-170c. Binding of Mim8 to FIXa at this site may promote a more conformationally active FIXa.

(Data source: Bos MHA, et al. Bos MHA. 2024)
SS327 is a globally pioneering recombinant factor nine product from Gensciences Biotechnology, combining Fc fusion and PEG technologies for long-acting efficacy. It is expected to become the first domestically produced long-acting recombinant factor nine product to meet a dosing frequency of 10-14 days. This product is expected to significantly reduce the number of biopsies and bleeding risk for patients, alleviating their disease and financial burden. The product possesses independent intellectual property rights and has already obtained patents in multiple countries.
