Zinc transporter ZIP6, also known as estrogen regulatory protein LIV-1 or SLC39A6, is a transmembrane zinc transporter responsible for maintaining intracellular zinc homeostasis and playing a crucial role in cell proliferation, differentiation, and metabolism. Zinc transporters function in zinc homeostasis and the induction of epithelial-mesenchymal transition (EMT). When bound to SLC39A10, the heterodimer formed by SLC39A10 and SLC39A6 mediates zinc uptake by cells, triggering EMT.
SLC39A6 expression distribution
While SLC39A6 has limited expression in normal tissues, LIV-1 is highly expressed in breast cancer (93%), melanoma (82%), prostate cancer (72%), ovarian cancer (48%), and uterine cancer (30%). LIV-1 is considered one of the attractive cell surface targets for developing ADC therapy.
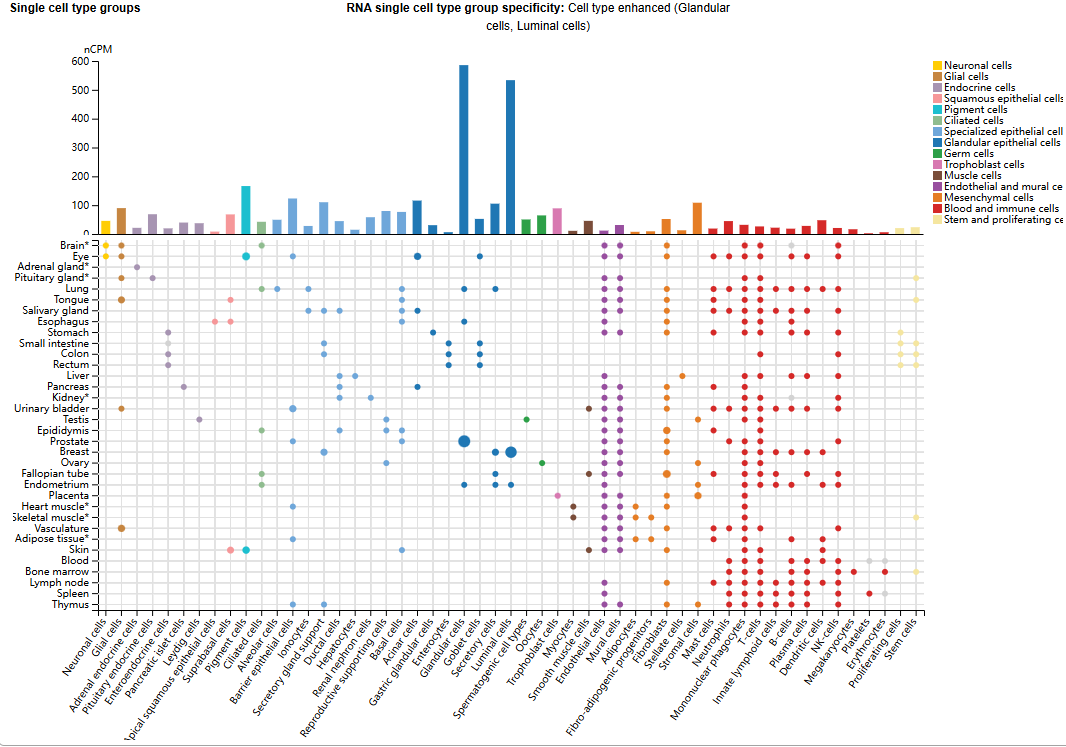
(Data source: uniprot)
SLC39A6 Structure
SLC39A6 is a six-transmembrane protein belonging to the LIV-1 subfamily. It exists in two isomers with lengths of 755 and 433 amino acids. Both its amino and carboxyl terms are located extracellularly or within vesicles. A histidine-rich domain exists between the third and fourth transmembrane domains; this region binds zinc ions and exhibits metalloproteinase activity.
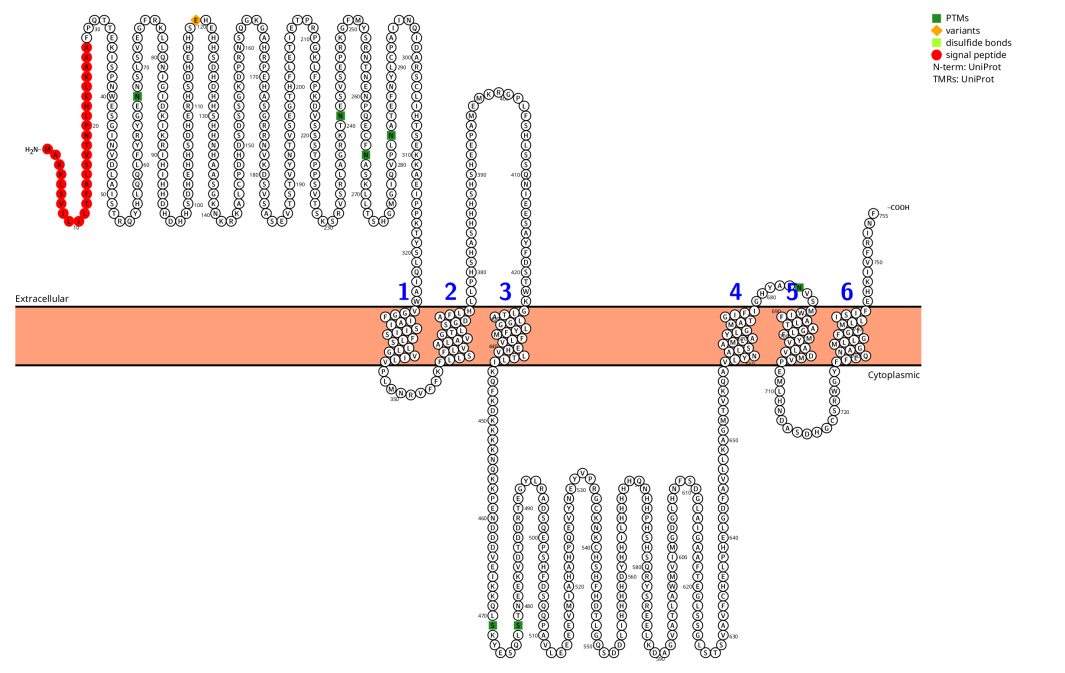
(Data source: protecter)
SLC39A6 in cancer progression
SLC39A6 drives epithelial-mesenchymal transition (EMT) through the STAT3 signaling pathway and inhibits E-cadherin expression, thereby significantly promoting cancer cell migration, invasion, and anti-anorepopulation ability. This protein is often highly expressed in various malignant tumors (such as triple-negative breast cancer, esophageal cancer, and liver cancer), and its elevated levels are usually closely related to advanced stage, lymph node metastasis, and shortened patient survival. SLC39A6 also acts as a key regulator of the immune microenvironment, mediating zinc ion influx to activate CD4+ T cells, and its expression level is positively correlated with the number of tumor-infiltrating lymphocytes (TILs). Although it may exhibit tumor-suppressive characteristics in some estrogen receptor-positive breast cancers, in aggressive subtypes such as triple-negative breast cancer, it mainly plays a carcinogenic role that promotes tumor progression.
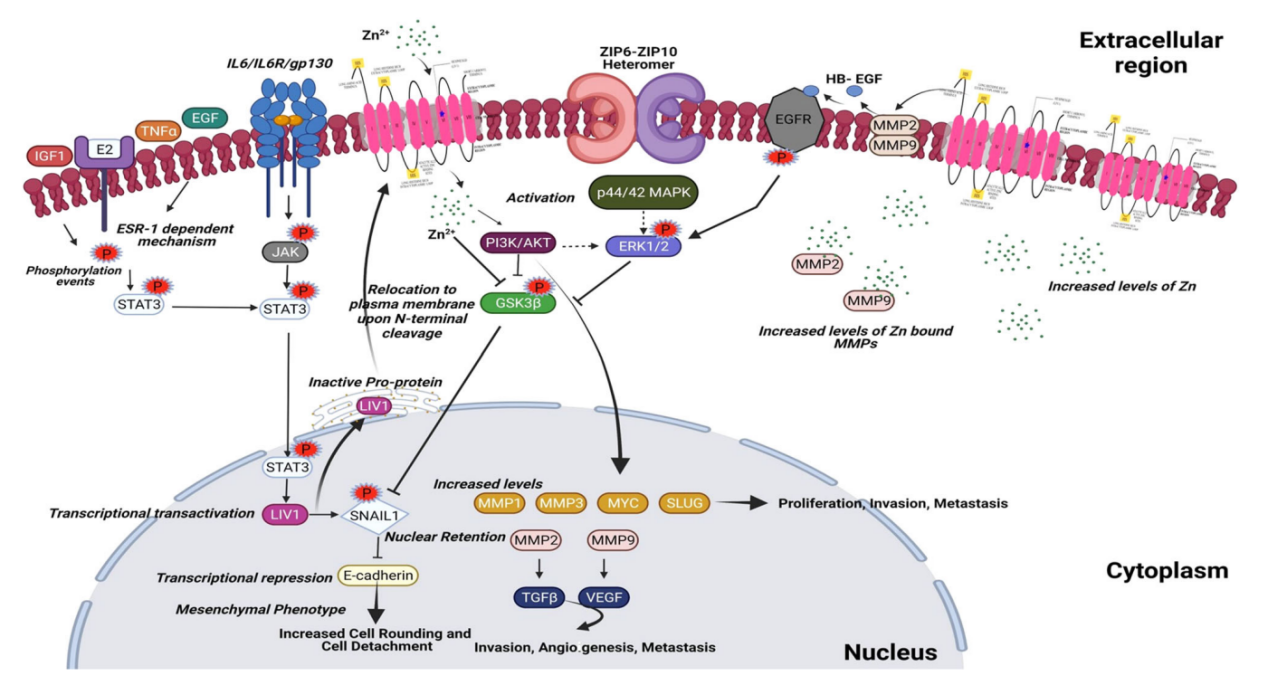

(Data source: Saravanan R, et al. J Cell Physiol. 2022)
Targeted therapy for SLC39A6
Given its high specific expression on the tumor surface, SLC39A6 has become an ideal target for antibody-drug conjugates (such as SGN-LIV1A), which can effectively treat cancer by inducing mitotic arrest and immunogenic cell death (ICD).
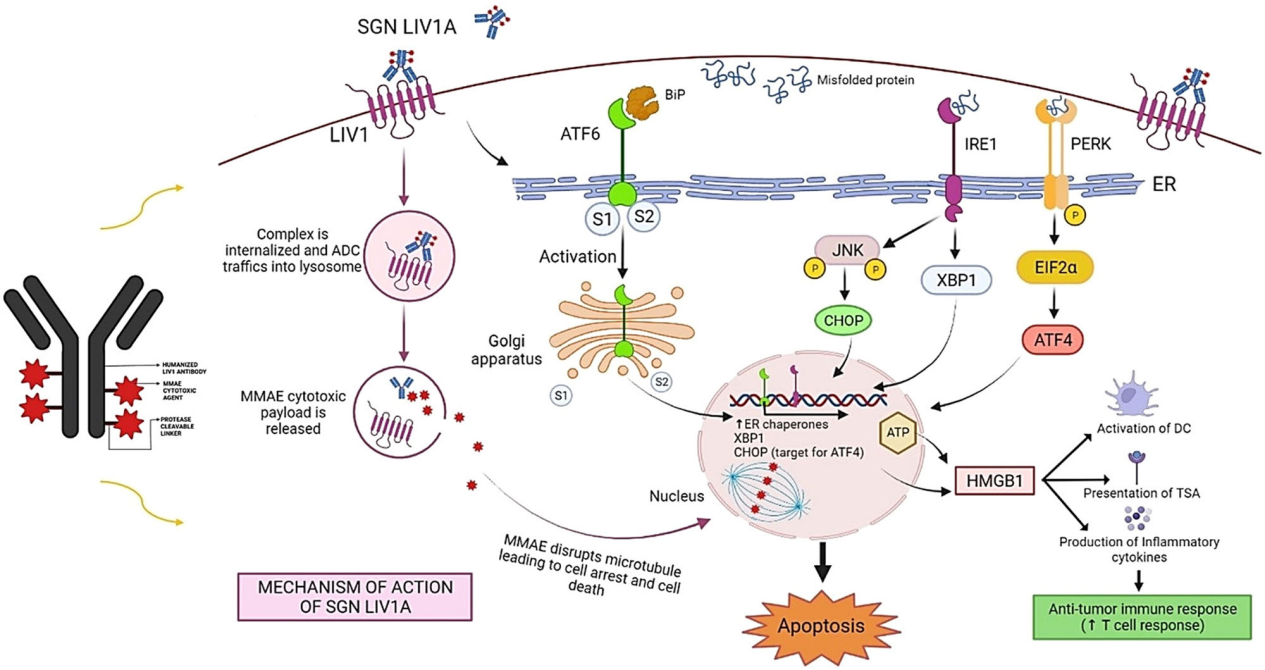
(Data source: Saravanan R, et al. J Cell Physiol. 2022)
BRY812 is an antibody-drug conjugate targeting SLC39A6 developed by BioRay Biotech, carrying the microtubule inhibitor monomethylolpropamine E (MMAE) as a cytotoxic payload. BRY812 is used to treat solid tumors such as gynecological cancer and prostate cancer. It has demonstrated a favorable safety and tolerability profile in patients with advanced solid tumors and shown promising clinical efficacy. A Phase Ib study is currently underway to optimize the dosage and further evaluate its efficacy in triple-negative breast cancer, HR+HER2 low-expressing or negative breast cancer, and other solid tumors.
Ladiratuzumab Vedotin (SGN-LIV1A) is currently the most advanced ADC drug targeting SLC39A6. Developed by Seagen (now acquired by Merck), it consists of a humanized anti-SLC39A6 monoclonal antibody conjugated to the microtubule inhibitor MMAE via a cleavable linker. The antibody specifically binds to SLC39A6 on the surface of tumor cells, enters the cell via endocytosis, releases MMAE, disrupts the microtubule network, and induces apoptosis. Clinical trials have been conducted in solid tumors such as triple-negative breast cancer (TNBC) and non-small cell lung cancer (NSCLC), showing encouraging objective response rates (ORR), especially in previously treated advanced patients.
TST013 is a next-generation ADC targeting the clinically validated tumor antigen LIV-1, which is highly expressed in breast cancer and other solid tumors. This ADC molecule binds to a site-specific linker of a TOPO-I inhibitor, along with an internally humanized antibody with distinct epitopes and prolonged pharmacokinetics.
Transcenta's main LIV-1 ADCs (ADC-1 and ADC-2) are engineered using the company's proprietary antibodies, specifically binding to a topoisomerase I (Topo I) inhibitor payload. These ADCs demonstrated significantly higher tumor regression activity than MMAE-based ADCs in a TNBC tumor model. The significantly enhanced antitumor activity of ADC-1 and ADC-2 is likely attributed to the high binding affinity and high internalization efficiency of Transcenta's proprietary antibodies to LIV-1, and the high cytotoxicity of the topoisomerase I inhibitor to cancer cells. These data warrant further investigation as potential next-generation therapeutics for LIV-1-targeted ADCs (ADC-1 and ADC-2) in LIV-1-expressing breast cancer and other solid tumors.

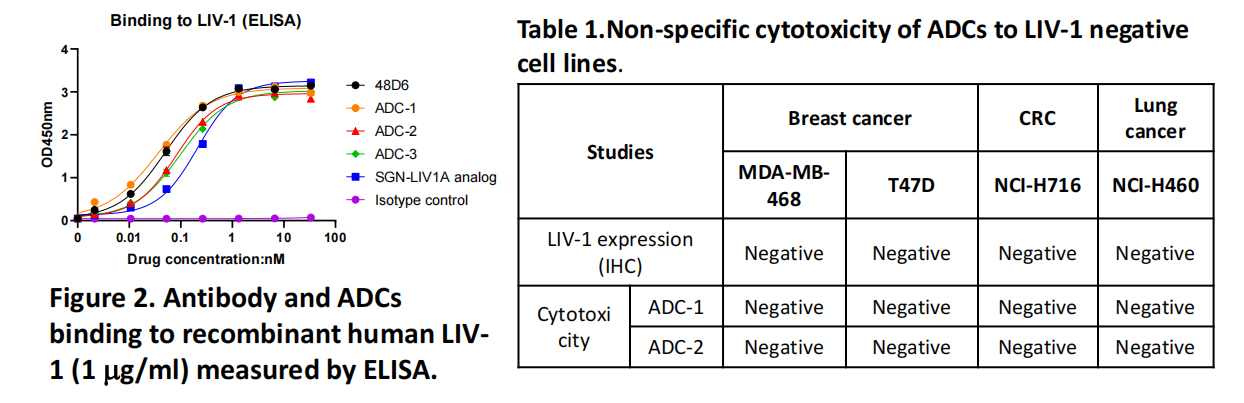
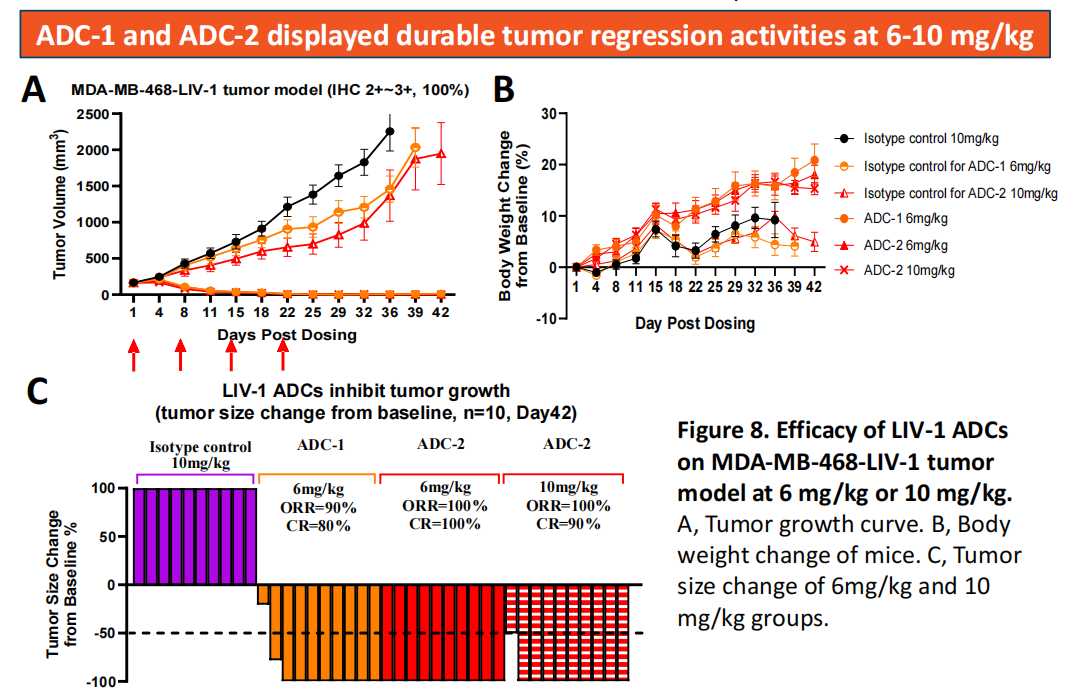
(Data source: Transcenta Group official website)
