Service Support
Mabnus Biotech provides recombinant protein expression services for human NAMPT.

Background
Calcific aortic valve disease (CAVD) has become a major age-related cardiovascular epidemic, affecting more than 12% of people aged 65 and older, and is characterized by a progressive transition from early fibrotic calcification to severe aortic stenosis. With the aging of the global population, CAVD is the third leading cause of cardiovascular death. Although the pathogenesis of CAVD is gradually being elucidated, the age-related biological changes in the aortic valve remain unclear. Recent advances in aging research have identified common features, including cellular senescence, chronic inflammation, and mitochondrial dysfunction, which drive both aging and the pathogenesis of CAVD.
Nicotinamide adenine dinucleotide (NAD) is a key metabolic regulator of cellular bioenergetics and signal transduction, and its levels gradually decrease during aging. In cardiac tissue, over 85% of NAD production is highly dependent on nicotinamide phosphoribosyltransferase (NAMPT), the rate-limiting enzyme of the salvage pathway. The spatiotemporal dynamics of NAMPT during valvular calcification progression and its precise regulatory mechanisms on valvular cell fate determination remain to be systematically investigated.

On March 17, 2026, a research team led by Dong Nianguo from Wuhan Union Hospital of China, published a study titled "Senescence-associated metabolic alterations aggravate calcific aortic valve disease" in Eur Heart Journal. The study found that the main form of metabolic reconfiguration in aging aortic valves is the inhibition of NAMPT-mediated repair pathways. At the single-cell level, the heterogeneous NAMPT expression patterns between valvular endothelial cells (VECs) and macrophages were depicted, revealing intercellular interactions that coordinate the calcification process. Furthermore, the intrinsic NAMPT heterogeneity within macrophages was found to have a dual function in driving the progression of coronary vascular disease (CAVD), promoting inflammatory activation and extracellular matrix (ECM) remodeling. These findings suggest that NAMPT deficiency in endothelial cells is a major initiating factor for age-related CAVD, in which recruited macrophages amplify the disease through inflammatory cascades and collagen degradation.
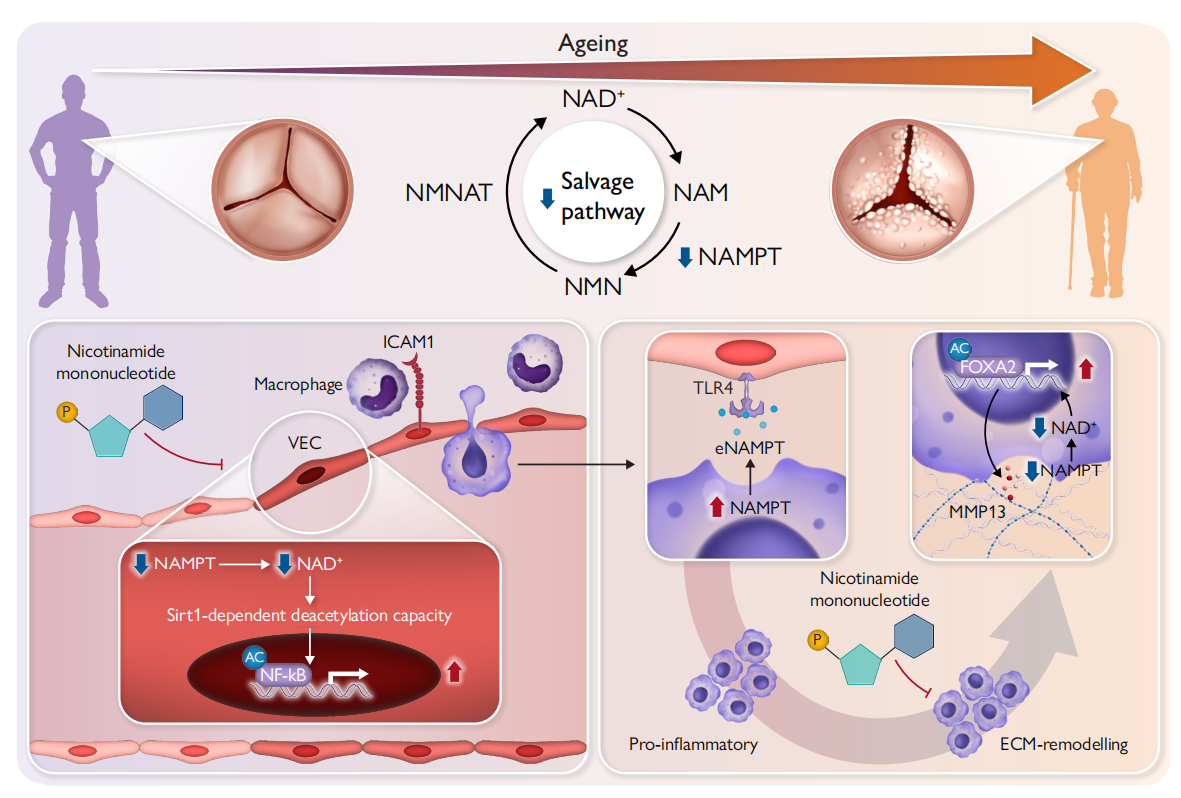
The NAMPT-mediated NAD+rescue pathway is significantly downregulated in the aortic valve of aging individuals
Transcriptomic analysis revealed the most significant decrease in the expression of NAMPT, a key enzyme in NAD+ synthesis, in the aortic valves of aging individuals, and this decrease was strongly correlated with age rather than disease severity. Even in aging valves without significant calcification, reduced NAD+ levels and activation of early osteogenic signaling were observed, indicating that these are markers of early age-related remodeling.

NAMPT deficiency exacerbates aortic valve calcification and heart failure in mice.
Heterozygous NAMPT-deficient mice developed aortic stenosis, leaflet thickening, and calcification earlier than wild-type mice on a high-fat diet, accompanied by decreased ejection fraction and myocardial fibrosis, indicating that haplo-insufficient NAMPT exacerbates age-driven intrinsic valvular remodeling. Treatment to eliminate senescent cells was effective in wild-type mice but had limited effect in NAMPT- deficient mice, suggesting that NAMPT deficiency exacerbates the refractory nature of the fibrosis-calcification trajectory during aging.

Single-cell sequencing reveals that downregulation of NAMPT in endothelial cells drives macrophage recruitment
Single-cell RNA sequencing revealed that NAMPT expression was most significantly decreased in valvular endothelial cells (VECs) of aging valves, while NAMPT expression was upregulated in infiltrating macrophages. In elderly human aortic valves, the interaction between vascular endothelial cells (VECs) and mononuclear phagocytes was significantly enhanced. These findings suggest that the downregulation of endothelial NAMPT expression during aging is closely related to enhanced CXCL-mediated endothelial-macrophage interactions and the recruitment of inflammatory macrophages.

NAMPT downregulation drives inflammation of valvular endothelial cells through increased NF-κB acetylation
Both drug inhibition of NAMPT (FK866) and gene knockout experiments significantly upregulated the expression of ICAM1 and VCAM1 in vascular endothelial cells (VECs) and significantly inhibited the deacetylation activity of SIRT1. Crucially, the loss of SIRT1 is similar to this inflammatory activation phenomenon, and its mechanism involves enhanced NF-κB p65 acetylation, a post-translational modification that promotes nuclear translocation and enhances pro-inflammatory transcription. In summary, these findings suggest that NAMPT downregulation drives vascular endothelial cell inflammation by impairing SIRT1-mediated deacetylation, leading to increased NF-κB activation levels.

Early NMN supplementation can reduce valvular inflammation and delay the progression of calcification in mice
Histopathological analysis established the therapeutic window: early intervention in haplo-underdose mice and conditional knockout mice reduced valve thickness and inhibited calcification, while late intervention did not produce significant benefit. These results establish NMN as an effective early intervention strategy to slow CAVD progression. However, its efficacy in the established calcified environment appears limited, with evidence mainly confined to molecular-level attenuation rather than significant reversal, thus highlighting the need for characterization of dynamic changes in effector macrophages.

Adaptive upregulation and heterogeneity of NAMPT in macrophages
At both the transcriptomic and protein levels, high NAMPT expression is significantly associated with pro-inflammatory macrophage polarization, and NAMPT upregulation, as an adaptive response, supports the energy requirements during pro-inflammatory polarization.

Extracellular NAMPT derived from macrophages can amplify endothelial inflammation
NAMPT exerts its effects through different intracellular forms (iNAMPT) and extracellular forms (eNAMPT), with eNAMPT acting as a potent amplifier of the inflammatory cascade. Mechanistically, pharmacological inhibition (TAK242) or endothelial TLR4 gene knockout significantly attenuates rhNAMPT-induced NF-κB signaling. eNAMPT, as a damage-associated molecular model (DAMP), further amplifies endothelial inflammation by binding to TLR4 receptors on the endothelial cell surface, creating a vicious cycle. Macrophage-derived eNAMPT directly drives inflammatory activation in VE Cs.

Senescent macrophages drive MMP13-mediated valve collagen remodeling via FOXA2 acetylation
In aging BMDMs, the clearance of NAD+ promotes FOXA2 acetylation, thereby enhancing MMP13 transcription. This cascade ultimately leads to ECM degradation, which in turn results in valve calcification. Notably, NMN supplementation also represents a promising therapeutic strategy to combat this pathological progression.

Summary
This study reveals that decreased NAMPT levels in aging VECs impair the conversion of NAM to NMN, weaken SIRT1-mediated p65 deacetylation, and activate NF-κB, thereby increasing ICAM1 expression to recruit macrophages. Macrophage-derived eNAMPT activates endothelial cell TLR4, while age-associated remodeled macrophages induce FOXA2-MMP13-mediated matrix destruction, accelerating calcification; NMN supplementation may disrupt this cycle. Calcific aortic valve disease is caused by endothelial NAD+ deficiency and is exacerbated by metabolically diverse macrophages. This compartmentalized NAD+ cycle links inflammatory senescence to matrix catastrophe. Early NAD+ supplementation via nicotinamide mononucleotide and NAMPT-targeted interventions warrant clinical evaluation as potential therapies for calcific aortic valve disease.
