Fibroblast growth factor 4 (FGF4), also known as Kaposi's sarcoma FGF, plays an important role in the regulation of embryonic development, cell proliferation, and cell differentiation and is required for normal limb and heart valve development during embryogenesis.
(Data source: AlphaFold)
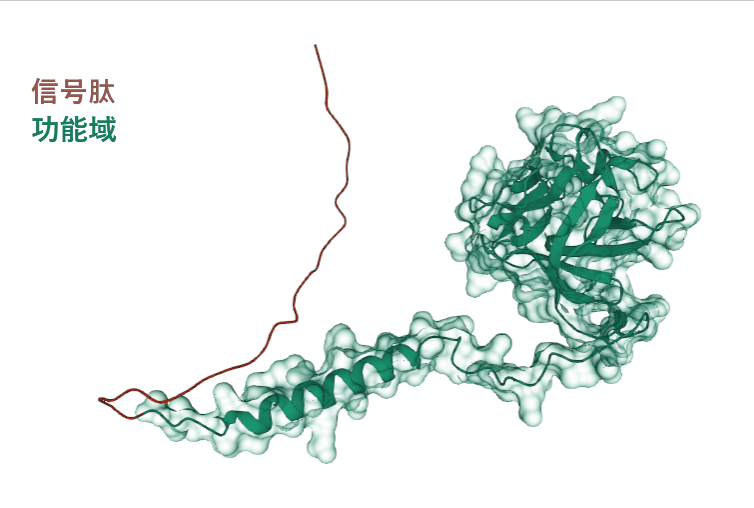
FGF 4 is composed of 206 amino acids and is a secretory extracellular protein. The main functional domain is the 31-206 segment, and the 1-30 segment is a signal peptide that guides protein secretion.

(Data source: Uniprot)
In adults, FGF4 is primarily found in tumors such as gastric cancer, Kaposi's sarcoma, and breast cancer, and is not produced under normal physiological conditions. FGF4 was the first fibroblast growth factor to be defined as an oncogene and is expressed throughout embryonic development.
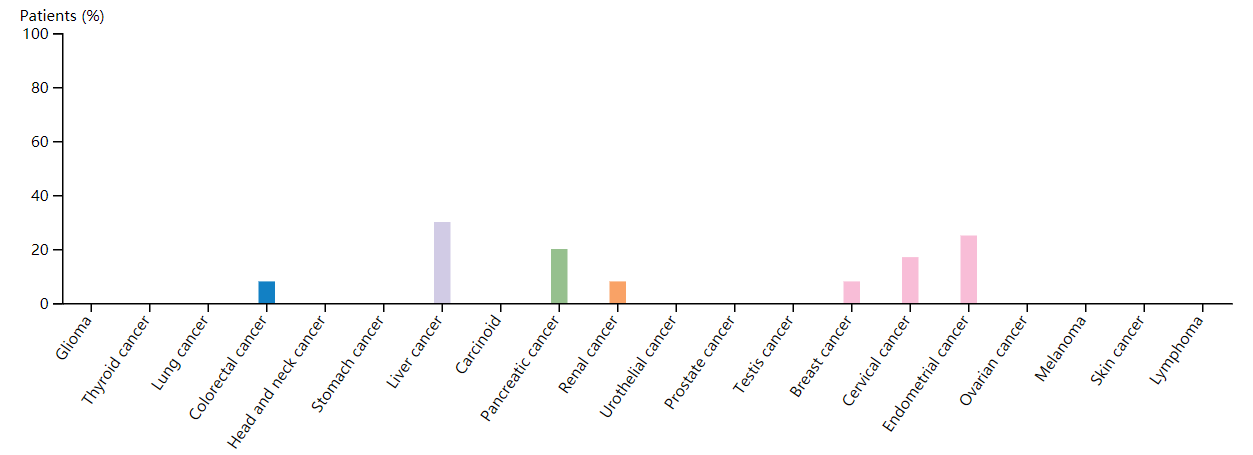
(Data source: Protein Atlas)
Most members of the FGF family have been identified as potential regulators of glucose homeostasis. A low threshold for FGF-induced FGF receptor 1c (FGFR1c) dimerization and activity is sufficient to elicit glucose-lowering activity. Long-term treatment with recombinant FGF4 (rFGF4) protein improves insulin resistance and suppresses adipose macrophage infiltration and inflammation. Unlike FGF1 (a pan-FGFR ligand), FGF4 has a more restricted binding specificity for FGFR1c. Its potent anti-hyperglycemic and anti-inflammatory properties demonstrate its significant potential for the treatment of type 2 diabetes (T2D) and related metabolic disorders.
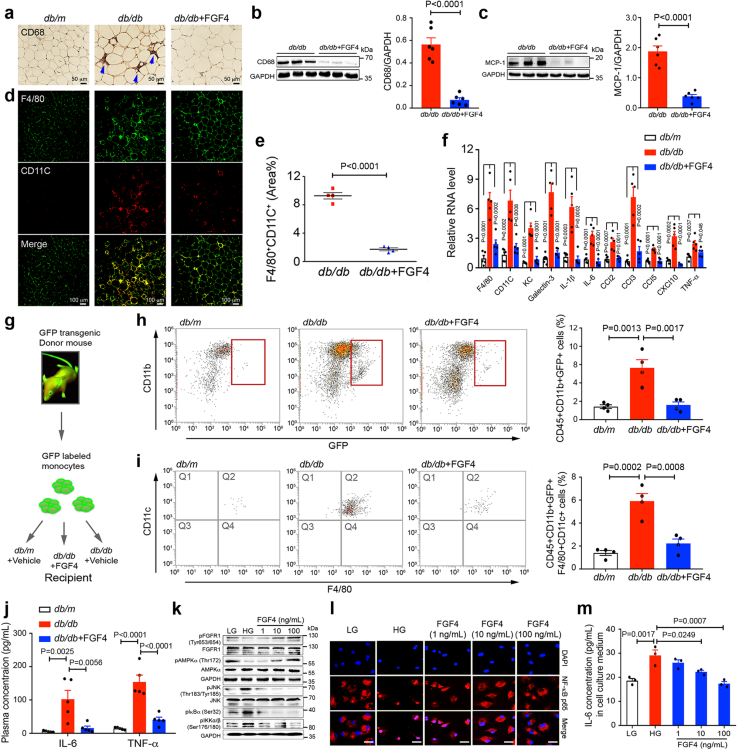
(Data source: Ying L, et al. Nat Commun. 2021)
rFGF4 protein also effectively normalizes the adverse metabolic phenotype of mice with high-fat diet-induced gestational diabetes (GDM) and is highly effective in treating neural tube defects in embryos in the GDM model. Mechanistically, rFGF4 treatment inhibits proinflammatory signaling cascades and neuroepithelial cell apoptosis in both GDM models.
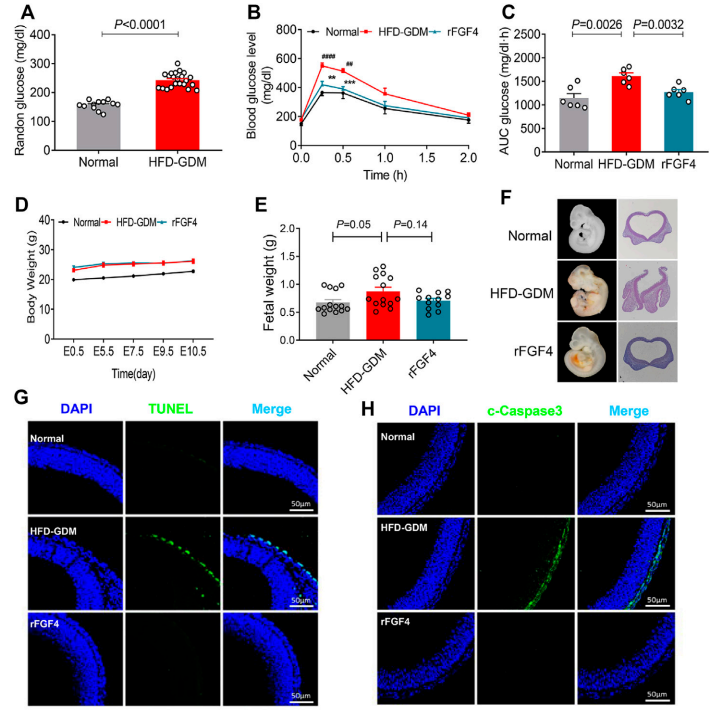
(Data source: Fan M, et al. Front Pharmacol. 2022)
Hepatic FGF4 expression is negatively correlated with pathological grade in both human patients and mouse models of nonalcoholic fatty liver disease (NAFLD) and nonalcoholic steatohepatitis (NASH) induced by dietary challenge: Hepatic FGF4 deficiency exacerbates hepatic steatosis and liver damage induced by an obesity-inducing high-fat diet , while pharmacological administration of rFGF4 alleviates hepatic steatosis, inflammation, liver damage, and fibrosis in the livers of these mouse models. These beneficial effects of FGF4 are primarily mediated by activation of hepatic FGF receptor (FGFR) 4, which promotes calcium influx and, in turn, activates the CaMKK2-AMPK-Caspas6 pathway, leading to enhanced fatty acid oxidation, reduced hepatocyte apoptosis, and attenuated liver damage.
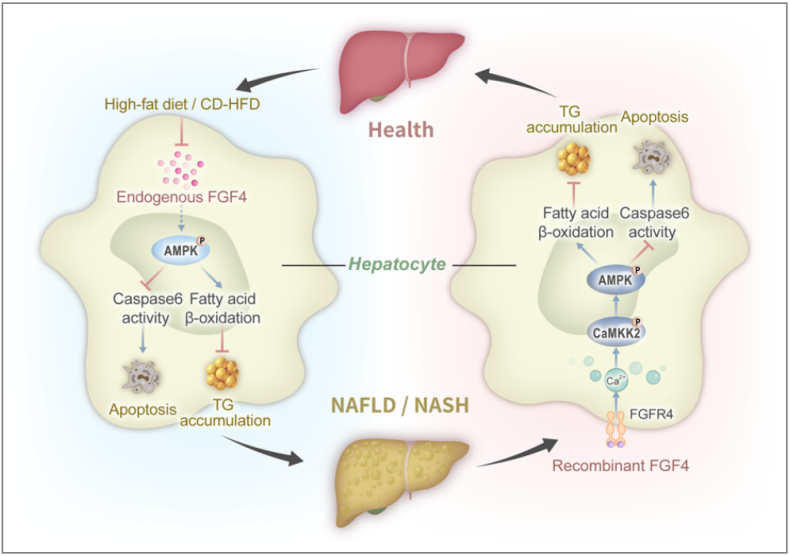
(Data source: Song L, et al. Hepatology. 2022)
Earlier studies have found that rFGF4 can stimulate the expression of matrix metalloproteinase 9 (MMP9) and VEGF receptor 1 in mouse skin fibroblasts in vitro. It combines with VEGF-A to promote fibroblast migration and accelerate wound healing in diabetic mice.
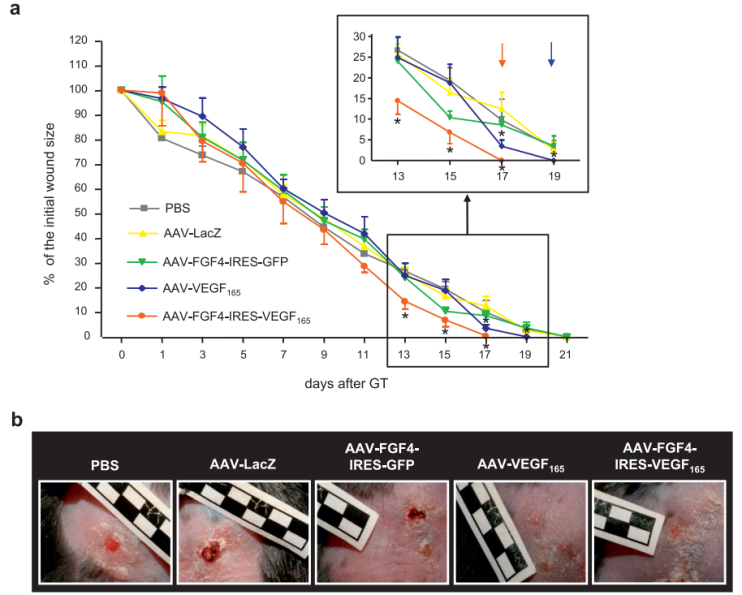
(Data source: Jazwa A, et al. Genet Vaccines Ther. 2010)
In human and mouse wound models, FGF4 is significantly upregulated at the wound edge in response to epidermal keratinocyte re-epithelialization . Blockade of FGF4 results in delayed re-epithelialization in human ex vivo skin wounds, whereas rFGF4 treatment promotes re-epithelialization and wound repair. rFGF4 primarily promotes p38 MAPK-GSK3β-mediated Slug stabilization by reducing Slug ubiquitination, thereby promoting keratinocyte migration and proliferation, which promotes wound re-epithelialization.
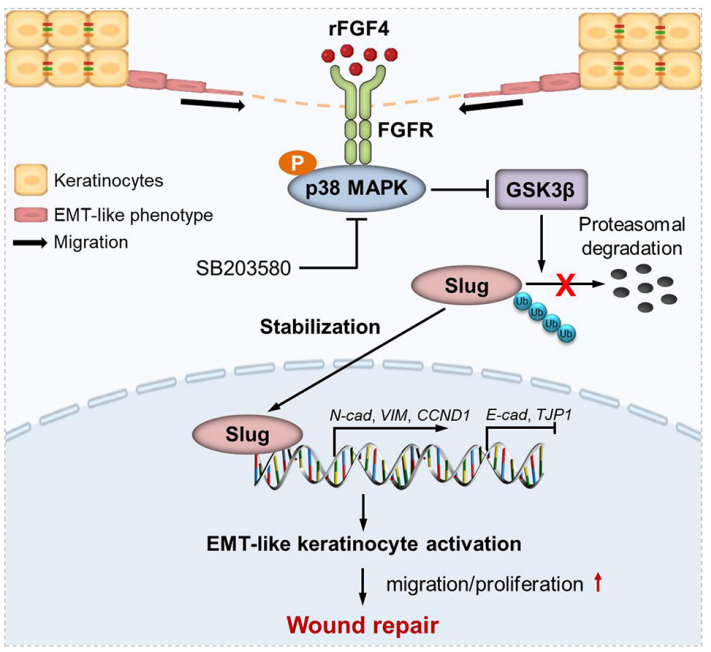
(Data source: Sun J, et al. J Invest Dermatol. 2022)
Given the involvement of the FGFR (fibroblast growth factor receptor) family in the pathogenesis of various cancer types, the selection of short peptide sequences derived from FGF4 that binds to FGFR1 as cancer-targeting agents presents an interesting strategy for expanding the portfolio of targeted anticancer drugs. One such short FGF4 peptide was successfully conjugated to monomethyl auristatin E (MMAE), demonstrating significant specific toxicity against FGFR1-expressing lung cancer cell lines.
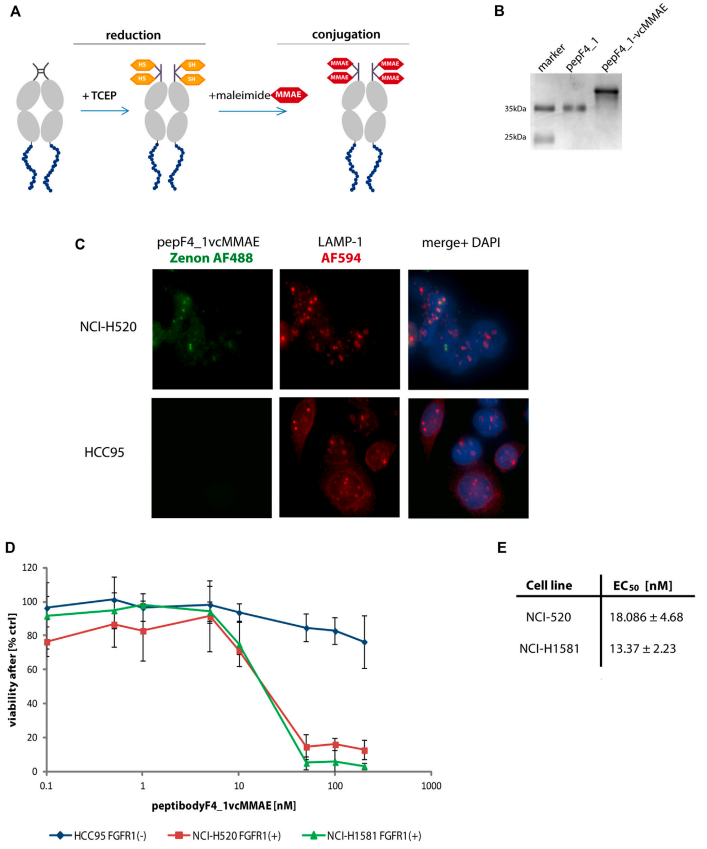
(Data source: Jendryczko K, et al. Front Pharmacol. 2021)
As an aside, FGF4 also has a history of drug development: Collateral and Cardium developed the gene therapy drug Ad5FGF-4 (adenovirus type 5 carrying the FGF4 gene), which leverages FGF4 to promote angiogenesis and improve myocardial ischemia. However, two Phase IIb/III clinical trials of this gene therapy were terminated before completion due to a high placebo response and the trial design being deemed unlikely to demonstrate benefit. Post-hoc subgroup analyses showed that only female patients showed substantial benefit. Ultimately, the gene therapy was shelved for over a decade. Although it has been re-introduced into clinical trials, recruitment is not promising.
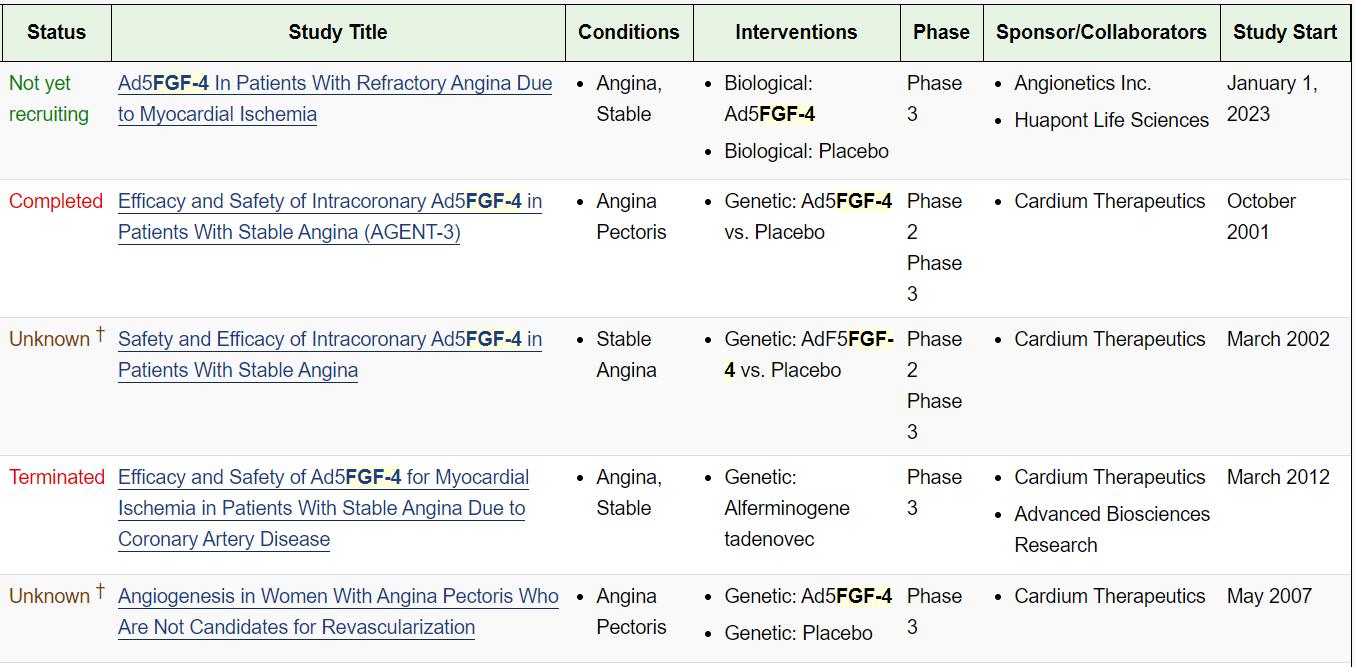
(Data source: Clinical trials)
There have been no major breakthroughs or new discoveries in FGF4 research so far. We hope that scientific researchers will continue to delve deeper and achieve results in the field of glucose metabolism as soon as possible.
