For general detection antibodies, we mainly consider their specificity and affinity, but for functional therapeutic antibodies, in addition to these two indicators, we also need to consider antibody-mediated humoral immunity and innate immune activity.

(Data source: Delidakis G, et al. Annu Rev Biomed Eng. 2022)
The pharmacological properties of antibodies are largely determined by their Fc regions. Interaction with complement C1q initiates complement-dependent cytotoxicity (CDC). Binding to various Fc receptors on leukocytes can induce antibody-dependent cell-mediated cytotoxicity (ADCC) or antibody-dependent cell-mediated phagocytosis (ADCP). Binding to the neonatal Fc receptor (FcRn) can extend the half-life of IgG. For many years, scientists have studied the structures involved in binding to these various ligands with the goal of altering the natural properties of antibodies to enhance (for example, increase tumor cell cytotoxicity or extend half-life) or reduce (for example, avoid unwanted side effects).

(Data source: Delidakis G, et al. Annu Rev Biomed Eng. 2022)
For tumor immunology indications, this antibody-mediated cell killing is very necessary, but when the therapeutic antibody simply acts as a blocker to prevent the interaction of the ligand with its receptor, the Fc effector function is unnecessary because it may mediate high cytotoxicity and kill normal body cells.

(Data source: Vonderheide RH, et al. Clin Cancer Res. 2013)
At present, a large number of different mutant Fcs have been developed to enhance/silence Fc effector functions (data source Liu R, et al. Antibodies (Basel). 2020 Wang X, et al. Protein Cell. 2018).

1.Enhance Fc effect:
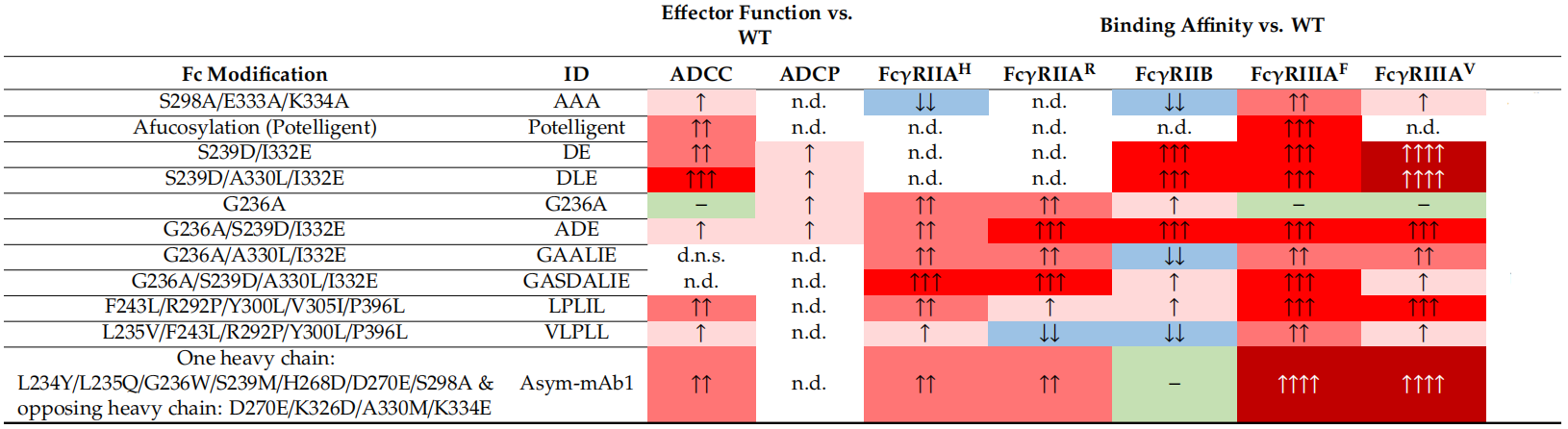

2.Silencing Fc effect:

3. Extend half-life:

4. Bispecific antibody heterodimerization: