The interleukin-2 receptor (IL-2R) is composed of three subunits: α (IL-2Rα , CD25), β (IL-2Rβ, CD122), and γ common (IL-2Rγ, CD132). CD25 is the only subunit of the IL-2 receptor that binds IL-2. These three subunits have different affinities and are all essential. CD25 deficiency can lead to autoimmune diseases such as autoimmune enteritis, viral infections, and various lymphomas. CD25 is a key marker highly expressed on the surface of regulatory T cells (Tregs). Tregs regulate immune responses to maintain immune tolerance and prevent excessive immune responses. High CD25 expression is a key characteristic of Tregs in carrying out their immunosuppressive functions.
CD25 structure:
The CD25 molecule is encoded by the gene IL2RA. It is a single-chain protein containing approximately 272 amino acid residues, including a 21-amino acid residue signal peptide. The mature molecule contains 251 amino acids, multiple cysteines, 2 N-glycosylation sites, and the transmembrane region and intracellular region contain 19 and 13 amino acid residues, respectively.
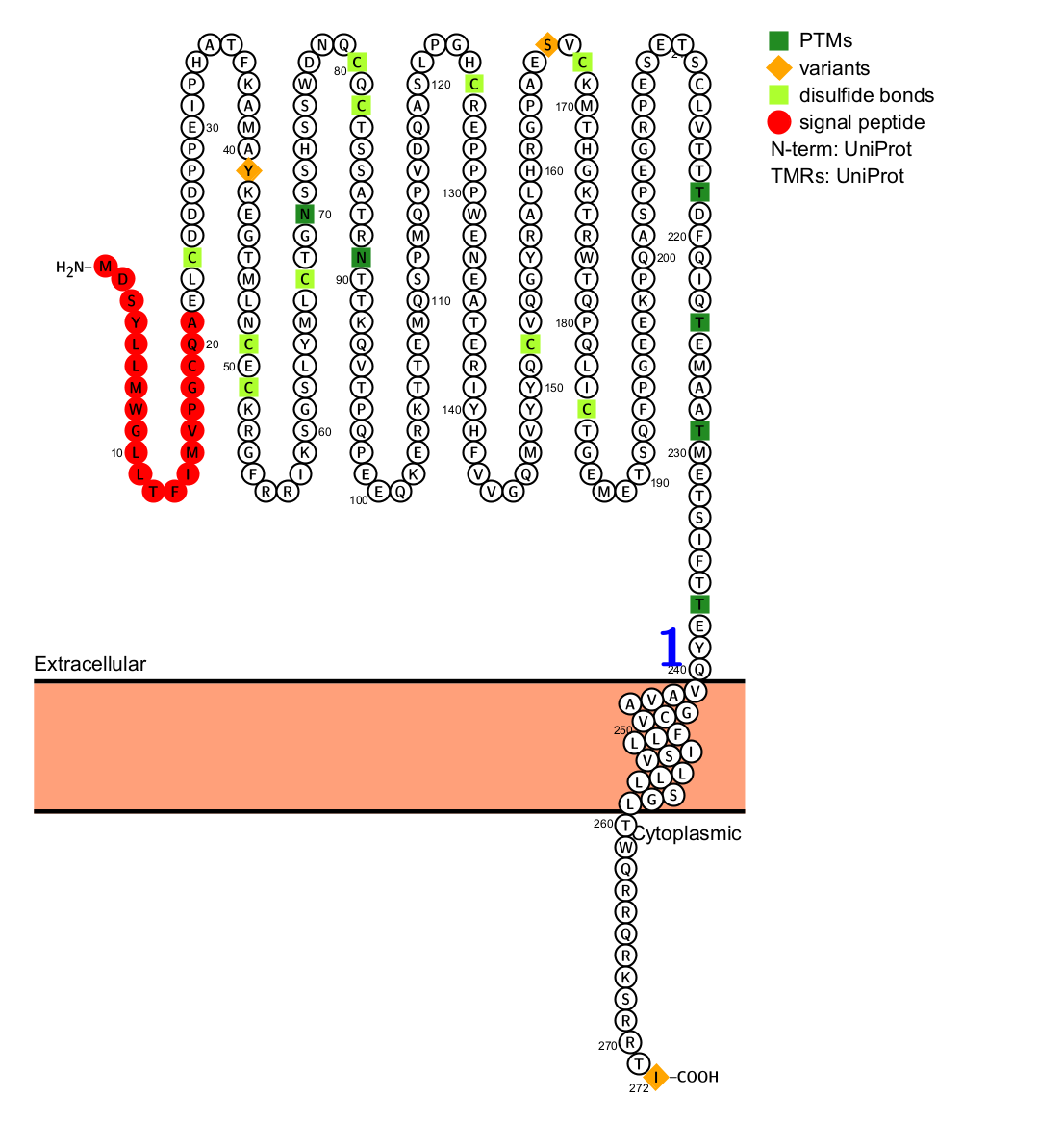
(Data source Uniprot)
The extracellular region of CD25 contains an immunoglobulin (Ig)-like domain, which is the site of its binding to IL-2. This region comprises a single Ig-like fold, consisting of a V-type (variable) region and a C-type (constant) region. This domain is characterized by a specific binding site for the IL-2 molecule.

(Data source Yuxi Wang , et al. Int . J. Cancer . 2022)
CD25 signaling pathway and regulation:
CD25 is the only subunit that binds to IL-2. IL-2, a low-affinity cytokine, initially binds to CD25 (IL-2Rα), forming a low-affinity complex. This binding promotes the formation of a heterodimer of IL-2Rβ and IL-2Rγ, leading to the formation of a high-affinity IL-2R complex. This in turn promotes the formation of IL2Rα/β/γ and IL-2 tetramers, ultimately initiating signal transduction. In cells with high levels of IL2Rβ and IL2Rγ, the heterodimer of IL2Rβ and IL2Rγ can independently initiate signal transduction when bound to IL-2. The binding of IL2Rα and IL-2 alone does not induce signaling. However, upon IL-2 binding to IL-2R, IL-2 primarily triggers the JAK/STAT5, PI3K/Akt/mTOR, and MAPK signaling pathways, which play crucial roles in cell growth, survival, differentiation, and immunity. In cancer treatment, CD25 serves as a potential therapeutic target, potentially enhancing immune responses by modulating the aforementioned signaling pathways, particularly by influencing the function of regulatory T cells (Tregs). Tregs play an important role in maintaining immune homeostasis, but in the tumor microenvironment, their overactivation may suppress anti-tumor immune responses. Therefore, targeting CD25 to deplete Tregs could restore the balance between effector T cells (Teffs) and Tregs, thereby enhancing the efficiency of anti-tumor immune responses.
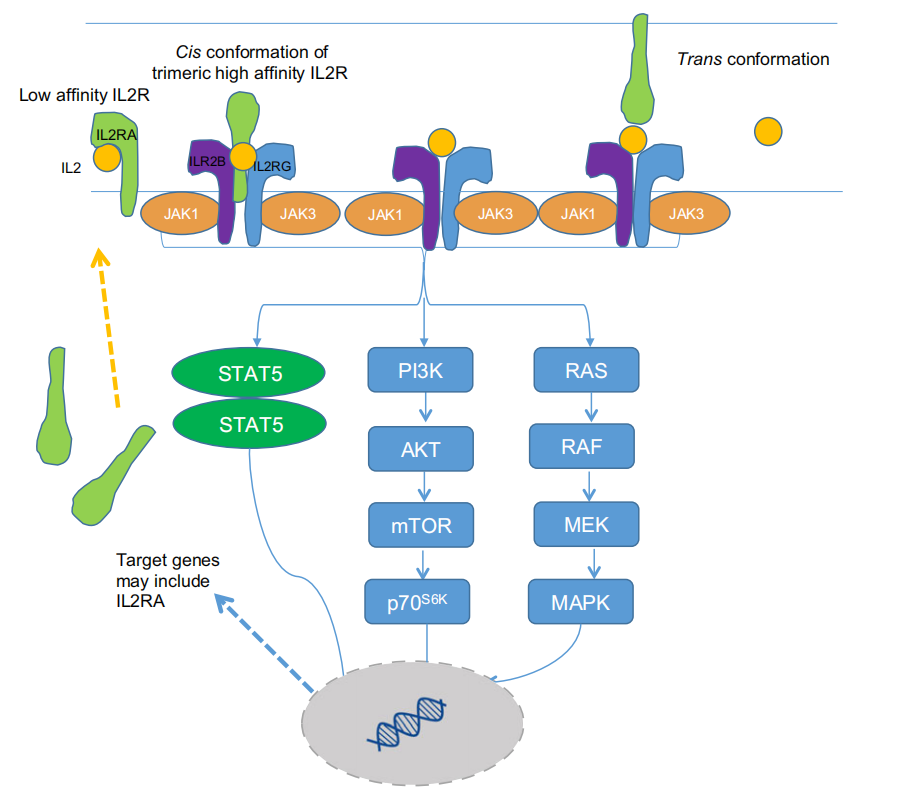
(Data source: JohnA. Hartley, atal. British Journal of Haematology. 2017)
Clinical value of CD25:
Drugs targeting CD25 for tumor treatment: There are currently two main strategies for developing drugs targeting CD25 for tumor treatment. First, recombinant IL-2, mutant IL-2, receptor-biased IL-2, and anti-CD25 monoclonal antibodies can act as immunomodulators to modulate IL-2 signaling, thereby affecting tumor initiation and progression. Second, tumors are killed by delivering cytotoxins to CD25+ cells. These CD25+ cells can be tumor cells or immune cells that regulate tumor cell survival.
A. Recombinant interleukin 2 binding to CD25: Recombinant interleukin 2 (IL2) binding to CD25 may affect tyrosine kinase signaling pathways, PI3K and RAS, and dimerization of STAT5.
B. Anti-CD25 antibodies: Anti-CD25 antibodies modulate cell signaling by competing with IL-2 for binding to an epitope on CD25. These antibodies may also affect antibody-dependent cell-mediated cytotoxicity and complementary cytotoxicity.
C. Anti-CD25 radioimmunoconjugates (RICs): RICs specifically bind to CD25-expressing tumor cells through their antibody moiety. Radiation emitted by the radioisotope causes direct damage to tumor cells or affects adjacent, non-targeted cells through a "crossfire" (bystander) effect. Radiation-induced DNA damage and cellular stress responses lead to apoptosis or necrosis of tumor cells.
D. Anti-CD25 immunotoxins: The antibody portion of the immunotoxin specifically binds to cells expressing CD25. The antibody-toxin complex is internalized by the cell through endocytosis, where it is then released by the acidic environment or by intracellular enzymes. The released toxin causes cell death by inhibiting protein synthesis or disrupting the cell membrane.
E. Anti-CD25 antibody-drug conjugate: The antibody-drug complex is taken up by cells through endocytosis. Inside the cell, the linker is cut by a specific intracellular environment (such as an acidic environment or a specific enzyme), releasing the cytotoxic drug. The released drug exerts its effect inside the cell, blocking cell division or inducing cell apoptosis, thereby killing the target cell.
F. Bispecific antibodies: CD25×CTLA-4 bispecific antibodies , dual antigens targeting the depletion of Treg cells, relieving the inhibition of effector T cells and enhancing their ability to attack tumor cells.
G. CD25-targeted CARNK cell therapy: NK-92 cells engineered with anti-CD25 CAR are able to specifically detect and lyse target cells with appropriate cytokine production and cytotoxic activity.

(Data source: Yuxi Wang, et al. Int. J. Cancer. 2022)

(Data source: Yuxi Wang, et al. Int. J. Cancer. 2022)
