Fibroblast growth factor receptor 3 (FGFR3) is a tyrosine protein kinase that acts as a cell surface receptor for fibroblast growth factor and is involved in regulating cell proliferation, differentiation, and apoptosis. It plays a crucial role in regulating chondrocyte differentiation, proliferation, and apoptosis and is essential for normal skeletal development. It regulates osteogenesis and postnatal bone mineralization through osteoblasts. It promotes chondrocyte apoptosis and may also promote cancer cell proliferation. Genomic alterations in FGFR3 are potent oncogenic factors in bladder cancer.
Expression distribution of FGFR3
FGFR3 is primarily expressed in astrocytes, basal cells, suprabasal keratinocytes, and bile duct cells. It is abnormally overexpressed in a variety of cancers, such as multiple myeloma, bladder cancer, cervical cancer, and gastric cancer.

(Data source: Uniprot)
The structure of FGFR3 and its receptor
FGFR3 is a single-pass transmembrane protein. Its extracellular region contains three immunoglobulin-like domains (IgI-IgIII), which are responsible for binding to FGF ligands (such as FGF1 and FGF9) with the assistance of heparin sulfate proteoglycans. The transmembrane region stabilizes the receptor's localization on the cell membrane. The intracellular region possesses tyrosine kinase activity and contains an ATP-binding site and autophosphorylation sites (such as Y647/Y648). Upon FGF ligand binding, FGFR3 dimers form, triggering autophosphorylation of the intracellular kinase domain and activating downstream signaling pathways.
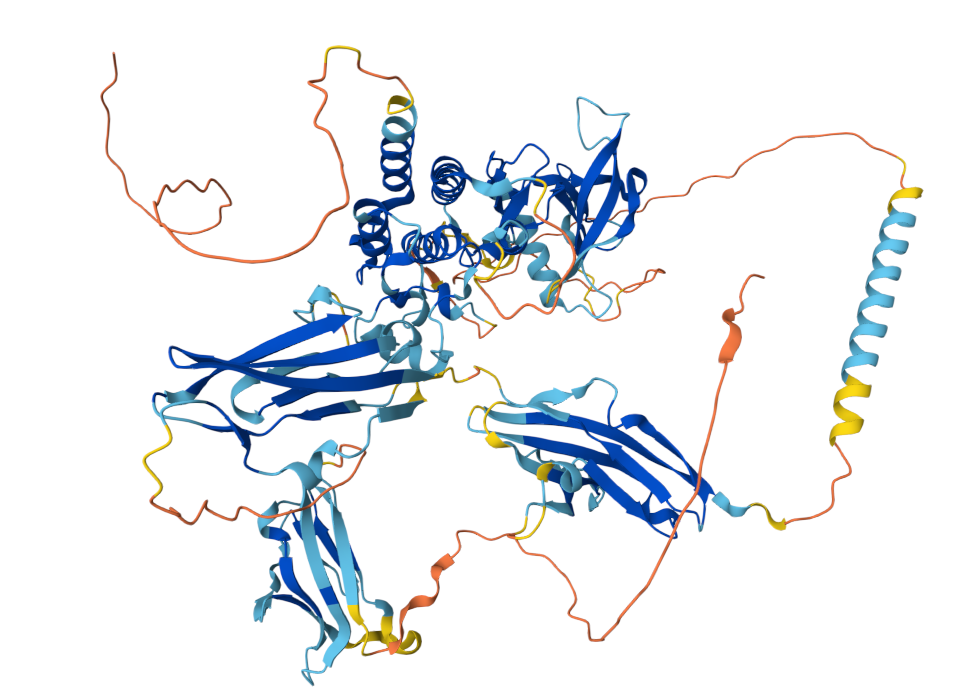
(Data source: AlphaFold)
FGFR3 signaling pathway and regulation
When fibroblast growth factors (FGFs) bind to the extracellular domain of FGFRs, FGFR3 undergoes conformational changes, triggering dimerization and transphosphorylation of its intracellular C-terminal tyrosine kinase domain. Different tyrosine residues in the intracellular C-terminal fragment of FGFR can trigger distinct downstream signaling pathways. When FGFRs become active, they may recruit complexes from the RAS or PI3K/AKT pathways through their essential adaptor FRS2α, thereby initiating signaling through the ERK or AKT pathways. Active FGFRs may also play a role in activating the JAK/STAT pathway. Furthermore, FGFRs activate PLCγ by inducing the hydrolysis of phosphatidylinositol-4,5-bisphosphate (PIP2) to inositol triphosphate (IP3), leading to calcium influx or triggering protein kinase C (PKC) signaling via diacylglycerol (DAG).
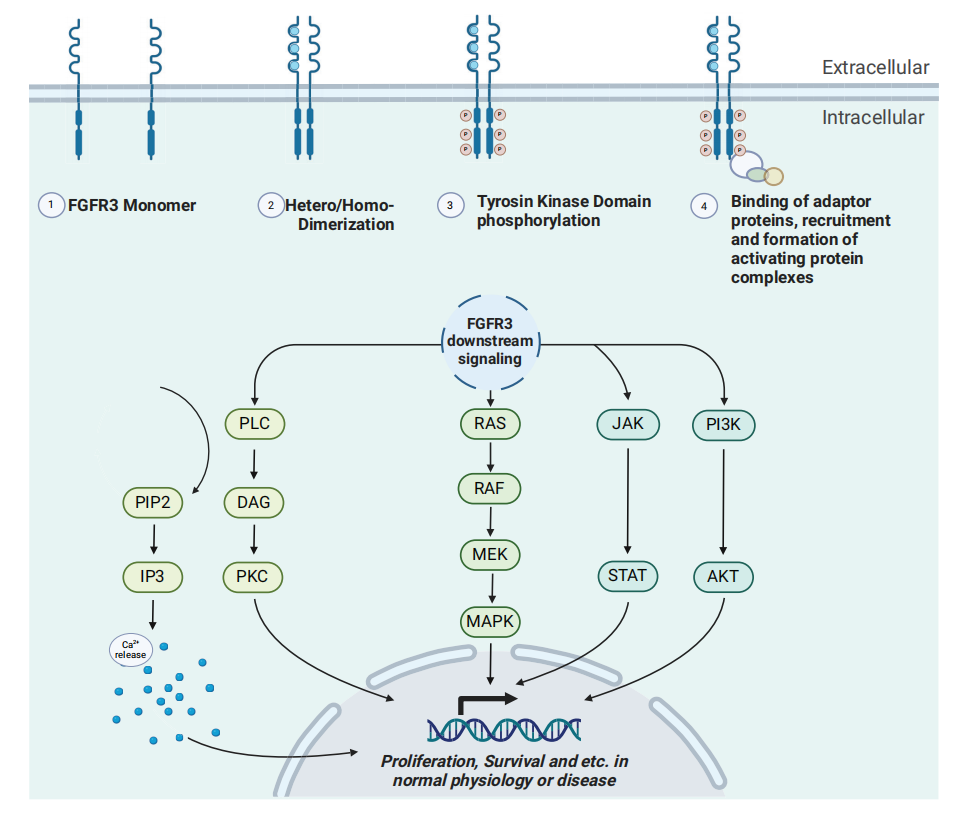
(Data source: Noeraparast M, et al. Cancer Commun (Lond). 2024)
FGFR3 alterations and their role in bladder cancer and other diseases
Fibroblast growth factor receptor (FGFR) alterations, particularly FGFR3 mutations, are among the most common alterations in BLCA. Point mutations predominate, followed by less common structural variants (approximately 14:1 ratio) that fusion with other genes, such as TACC3. Three prominent mutational hotspots within the FGFR3 coding sequence are concentrated at amino acid residues S249, R248, and Y373, leading to constitutive activation of FGFR3.
FGFR3 mutations promote cell proliferation and tumorigenesis by activating downstream signaling pathways such as RAS-RAF-MEK-MAPK, PI3K/AKT, calcium signaling, and JAK/STAT pathways.
A positive feedback loop between FGFR3 and MYC has also been discovered. FGFR3 activates MYC expression through p38α MAPK and stabilizes MYC protein through AKT-mediated GSK3β phosphorylation, thereby promoting tumor cell proliferation.

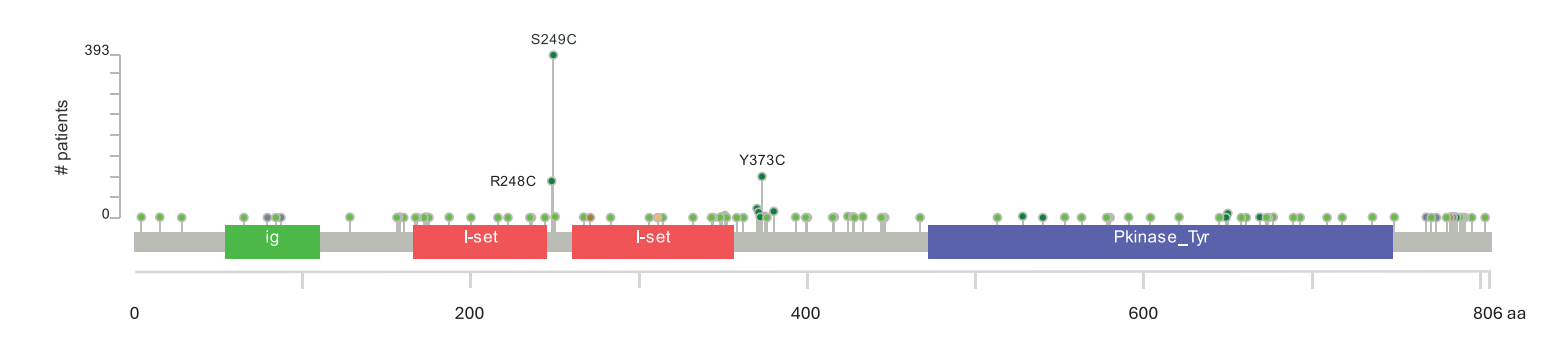
(Data source: Noeraparast M, et al. Cancer Commun (Lond). 2024)
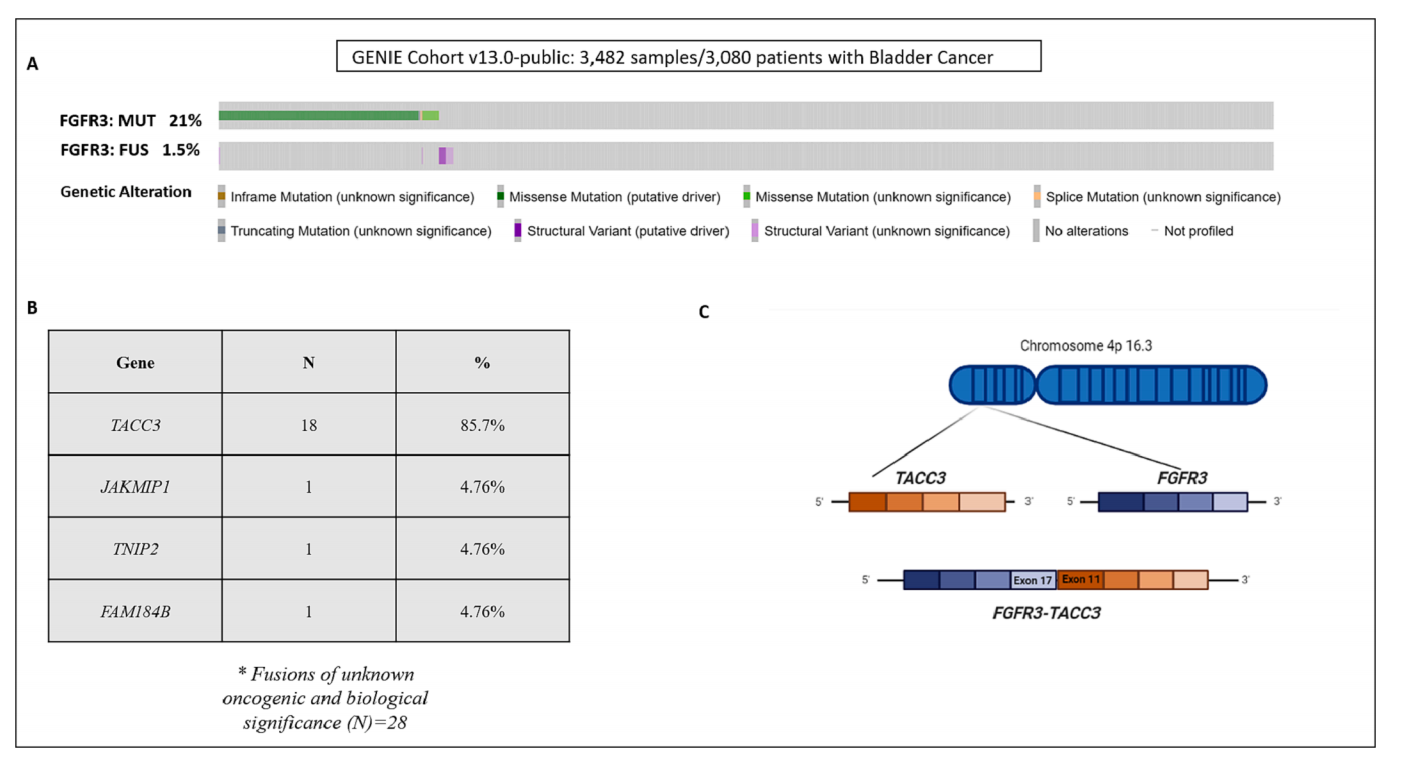
(Data source: Ascione CM, et al. Cancer Treat Rev. 2023)
FGFR3-targeted therapy
FGFR3-targeted therapies include small-molecule tyrosine kinase inhibitors (TKIs) with high affinity for the ATP-binding pocket. Based on their mechanism of action, these inhibitors can be divided into multi-target TKIs, including nonspecific inhibitors with affinity for several other receptors. For example, dovitinib and derazantinib, while these drugs have some inhibitory effects on FGFR3, are often associated with high toxicity and adverse reactions due to their multi-target nature. Specific, selective FGFR-TKIs, such as erdafitinib, rogaratinib, pemigatinib, infigratinib, and futibatinib, offer greater selectivity and lower toxicity and are currently the focus of research.
Other targeted therapies are being studied for the treatment of patients with advanced or metastatic bladder cancer, including monoclonal antibodies and antibody-drug conjugates (ADCs).

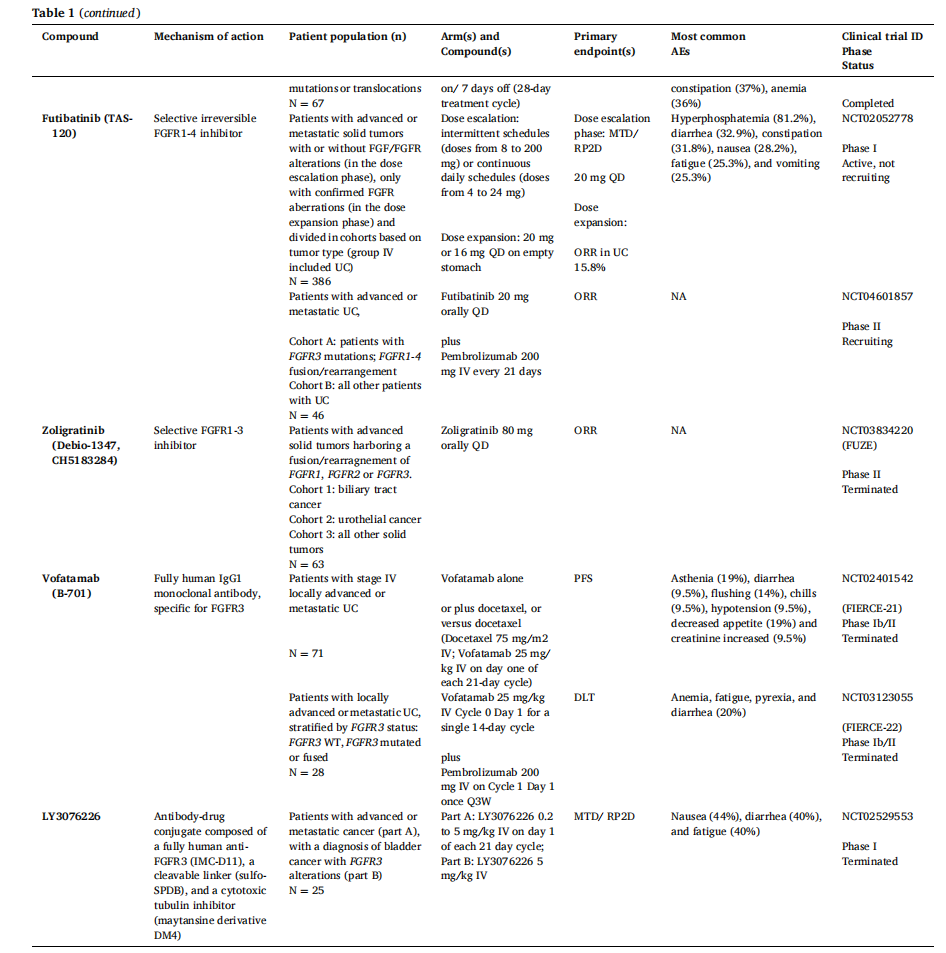
Vofatamab (B-701) is a fully humanized IgG1 monoclonal antibody targeting FGFR3. Preclinical studies have demonstrated antitumor activity in xenograft models of bladder cancer and multiple myeloma, as well as antiproliferative effects against FGFR3-positive cancer cell lines. Two separate trials are investigating vofatamab, either in combination with chemotherapy (FIERCE-21, NCT02401542) or with pembrolizumab (FIERCE-22, NCT03123055). While preliminary data from both studies appear encouraging, the pharmaceutical company has paused the trials pending complete results.
R3Mab (MFGR1877S) is a recombinant human antibody that selectively binds to the IgII and IgIII domains of FGFR3 but not to other FGFRs. In vitro, R3Mab significantly inhibited thymidine incorporation in bladder RT112 cells and reduced the viability of RT4 cells. In a bladder cancer model, R3Mab inhibited ligand phosphorylation and activation of downstream signaling pathways, consistent with previous results. R3Mab demonstrated potent antitumor activity in RT112 mouse xenografts and multiple myeloma cell lines.
(Data source: Ascione CM, et al. Cancer Treat Rev. 2023)
LY3076226 is an antibody-drug conjugate composed of the fully humanized anti-FGFR3 antibody IMC-D11 linked to the cytotoxic tubulin inhibitor emtansine derivative DM4 via a cleavable linker, sulfo-SPDB. The antibody binds to FGFR3 and blocks ligand-dependent signaling in tumor cell lines. In vivo studies demonstrated antitumor activity resulting from the cytotoxic effects of DM4, leading to cell cycle arrest, cell death, and tumor stasis. Although a Phase 1 study (NCT02529553) evaluated the safety, efficacy, and pharmacokinetics (PK) of LY3076226 in patients with urothelial carcinoma and FGFR3 alterations, development of the drug was halted due to pipeline priorities.
Drug resistance is a major challenge in FGFR3-targeted therapy. Mechanisms of resistance include secondary mutations, bypass activation, and downregulation of negative regulatory mechanisms. Combining FGFR3 inhibitors with other drugs (such as immune checkpoint inhibitors and chemotherapeutics) may improve efficacy and overcome drug resistance.
