Background
Making immunoglobulin G (IgG) antibodies autonomously internalize and localize to the cytoplasm of target cells (termed cytoplasmic-penetrating antibodies, CTs) is challenging but holds great promise. The main obstacle to CT entry into the cytoplasm is limited endosomal escape.
On April 11, 2025, Professor Yong-sung Kim of Seoul National University in South Korea published an article titled " Engineering IgG antibodies for intracellular targeting and drug delivery " in J Control Release. The researchers developed a second-generation (2G) CT, designated in2CT4.1, which features an endosomal pH-responsive endosomal escape motif (RW/E motif) containing an arginine-tryptophan pair and a Glu patch located between the CH3 and CL domains of the IgG1/ k antibody . This motif selectively disrupts the endosomal membrane at acidic endosomal pH to facilitate cytosolic entry, while remaining inactive at neutral pH. The 2G CT, in2CT4.1, achieved efficient cytosolic localization at nanomolar concentrations, demonstrating approximately threefold higher endosomal escape efficiency compared to first-generation CTs. The potential of the 2G CT was demonstrated by constructing a cytosolic α-tubulin-targeting CT in in2CT4.1 using the α-tubulin-specific variable domain.

Designing a pH-responsive endosomal escape motif in the CH3 domain of HC
Three key mutation regions were identified at the base tip of the CH3 domain to create a pH-responsive endosomal escape motif (RW/E motif) in the IgG CH3 domain: the AB loop (residues 353-362aa); the EF loop (residues 411-423aa); and the G strand (residues 439-437aa). These regions enhance the affinity of the homodimeric CH3 structure, and mutations in these regions do not affect Fc function.
The key role of the E-patch in the pH-dependent behavior of the R-W/E motif. Based on MD simulations, the authors proposed a mechanism by which the RW/E motif responds to endosomal acidic pH : at neutral pH, the negative charge of the E-patch repels the plasma membrane, preventing the binding of adjacent RW pairs. Conversely, in the acidic endosomal environment, partial protonation of the E-patch reduces its negative charge, enabling the RW pair to interact with the endosomal membrane and destabilize it, thereby promoting endosomal escape.

Cytoplasmic localization of CTs with RW/E motifs within the CH3 domain
Using split-GFP complementation assays, all six designed CT variants (in2CT1.N) exhibited higher GFP complementation fluorescence than 1G CT, with in2CT1.1 and in2CT1.9 showing the highest signals, indicating that in2CT1 possesses the ability to penetrate the cytoplasm. Control antibodies lacking endosomal escape ability, such as inCT (AAA) and traditional therapeutic antibodies, showed no GFP complementation fluorescence.
Using the SLEEQ assay to assess endosomal escape efficiency, in2CT1.1 and in2CT1.9 CTs exhibited significantly enhanced escape efficiency at 0.5 μM for 12 hours, indicating that the R-W/E motif significantly enhances the endosomal escape ability of CTs.
In2CT1.1 and in2CT1.9 showed a 3-fold increase in the uptake of peptide blue compared to inCT, consistent with the 2-fold enhancement observed in GFP and luciferase complementation experiments. In2CT1.1 w/o E allowed dye uptake at both pH 5.5 and pH 7.4, demonstrating a key role for the E-patch in selective endosomal escape under acidic conditions.

Incorporation of the RW/E motif into the CL domain confers cytosol-penetrating ability to CT
The CL domain has short α-helices in the AB and EF loops, similar to the CH3 domain . The RW/E endosomal escape motifs from the CH3 domains of in2CT1.1 and in2CT1.9 were incorporated into the structurally equivalent positions in the CL domain of inCT(AAA), creating the in2CT2.1 and in2CT2.2 variants. In a cytoplasmic penetration assay, in2CT2.1 demonstrated cytoplasmic penetration efficiency comparable to that of inCT but approximately two-fold lower than that of in2CT1.1. in2CT2.1 exhibited pH-dependent membrane pore-forming activity in Ramos cells. The RW/E endosomal escape motif can be efficiently incorporated into the CL domain, conferring cytoplasmic permeability to CTs without compromising their biophysical properties.

Characteristics of in2CT4.1 containing RW/E motifs in both CH3 and CL domains
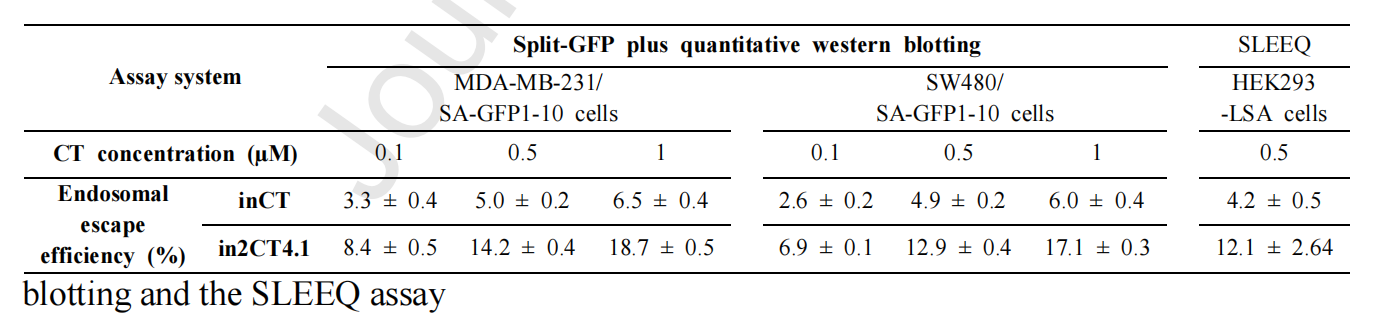
2G CTs were constructed by introducing an RW/E motif between CH3 and CL. In2CT4.1 showed significant GFP complementary fluorescence in MDA-MB-231/SA-GFP1-10 cells. Compared with the precursor clones in2CT1.1 and in2CT2.1, the cytoplasmic penetration efficiency of in2CT4.1 was significantly improved, which was increased by about 2.6 times.

Pulse-chase experiments revealed that in2CT4.1 escape from endosomes occurs primarily in the more acidified late endosomes, rather than the early endosomes. In2CT4.1 interacts with membranes and induces membrane pores for endosomal escape only at slightly acidic pH.

Differential scanning calorimetry was used to assess the thermal stability of in2CT4.1 . The midpoints of thermal denaturation for the CH2 domain (Tm1 ) and the Fab/CH3 domain ( Tm2 ) were approximately 70°C and 81°C , respectively, consistent with the characteristics of full-length IgG1/κ antibodies. Mutations in the RW/E motifs within the CH3 and CL domain loops of in2CT4.1 did not affect its interaction with FcRn, which primarily binds to the CH2-CH3 interface of the Fc region. 2G CT (in2CT4.1) retained FcRn binding ability, and its serum half-life was comparable to that of a wild-type IgG1/κ antibody , demonstrating that 2G CT retains the inherent properties of antibodies.

Advantages of the 2G CT platform
It can directly target cytosolic proteins by binding to specific VH/VL domains or by integrating into the CH3/CL constant domains of receptor-targeting antibodies, thereby conferring cytosolic penetration while maintaining antigen specificity. This versatility offers a significant advantage over 1G CT, in which the endosomal escape motif is confined to the VH/VL domains, limiting targeting flexibility. 2G CT can effectively deliver bacterial toxins into the cytoplasm of tumor cells, killing target tumors.

Summarize
κ antibody with intrinsic cytoplasmic penetrability, by introducing pH-responsive endosomal escape motifs into the CH3 domain of the heavy chain and the CL domain of the light chain. The 2G CT platform enables the development of therapeutic antibodies targeting previously refractory cytoplasmic proteins and is a valuable tool for applications requiring precise delivery of functional payloads to the cytoplasm. It effectively delivered bacterial toxins into the cytoplasm of tumor cells, killing target tumors. Compared to 1G CT, 2G CT demonstrated a three-fold increase in endosomal escape efficiency while maintaining desirable biophysical properties, including thermal stability and FcRn binding affinity, consistent with typical IgG/ κ antibodies. Future studies should focus on expanding the application of the 2G CT platform to deliver a range of therapeutic payloads, including nucleic acids and enzymes, and optimizing the endosomal escape motif to enhance efficiency. Furthermore, evaluating the long-term stability and immunogenicity of 2G CT-based therapies in clinical settings is crucial for translating this technology into effective treatments.
