CD33 is a cell surface protein belonging to the sialic acid-binding immunoglobulin-like lectins (Siglecs) family. It is a transmembrane protein that plays an important role in mediating cell-cell interactions and immune regulation. CD33 is generally expressed on monocytes, macrophages, dendritic cells, and mature myeloid cells, and is particularly expressed on leukemic cells in acute myeloid leukemia (AML), making it an ideal target for AML treatment. In the central nervous system, CD33 is expressed on microglia, which is associated with the clearance of Aβ plaques in Alzheimer's disease (AD). Due to its potential role in AD, CD33 may become a target for the development of new treatments.
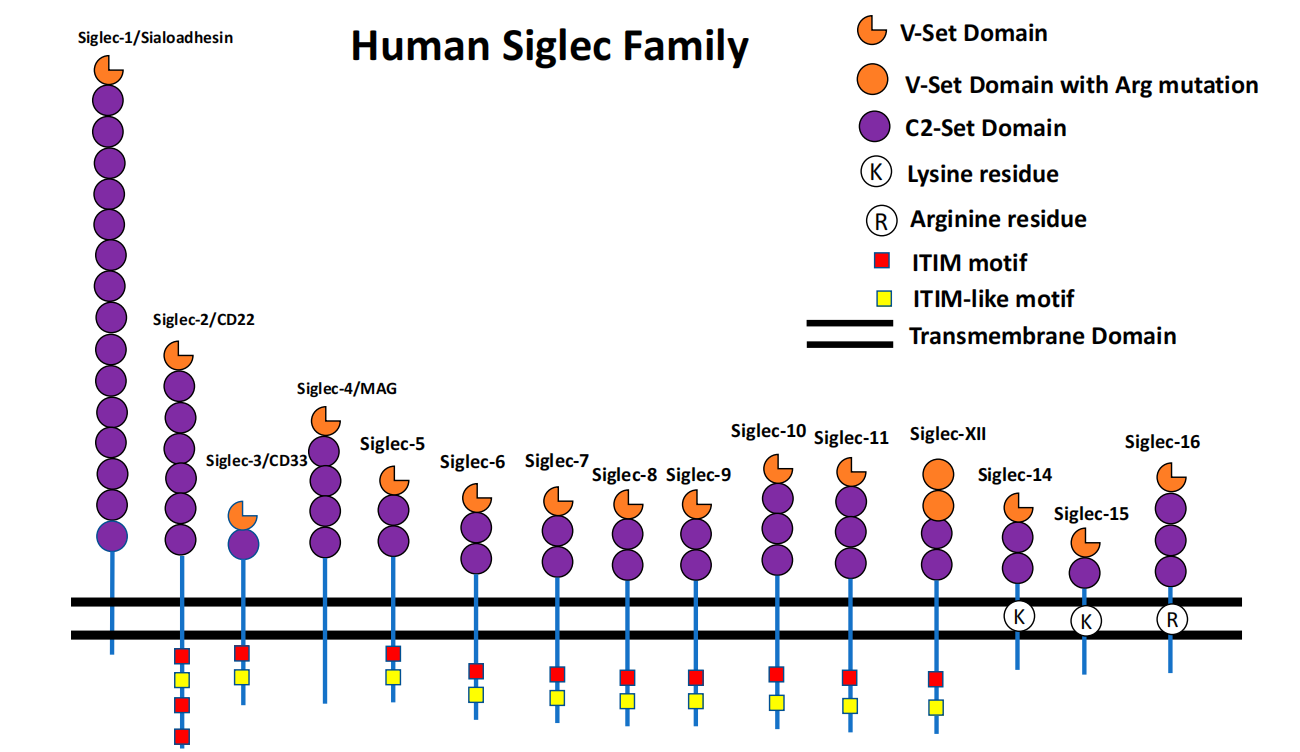
(Data source: Shoib Sarwar Siddiqui, at al. Cells. 2019)
CD 33 structure:
CD33 is a glycosylated transmembrane protein with a molecular weight of approximately 67 kDa. At its N-terminus, CD33 includes an extracellular V-set immunoglobulin-like domain, which is primarily responsible for recognizing sialic acid, and one or more C2-type immunoglobulin-like domains. The C2-type domain plays a supporting role in CD33's function and may participate in cell-cell interactions and signal transduction. The cytoplasmic tail of this transmembrane glycoprotein contains two conserved tyrosine-based inhibitory signaling motifs (ITIMs). ITIMs are the main domains of intracellular inhibitory signal transduction. These motifs play a key role in CD33-mediated signaling, regulating the activity of immune cells by recruiting and activating intracellular tyrosine phosphatases and other signaling molecules.

(Data source Teng Jiang, at al. Mol Neurobiol. 2014)
CD33 signaling pathway and regulation:
The function of CD33 depends on cell surface receptor-ligand interactions and cell type. Because CD33 is expressed on immune cells, a crucial role for CD33 is its ability to regulate immune cell function. Within immune cells, cellular processes such as phagocytosis, cytokine release, and apoptosis are co-regulated by activating receptors containing immunoreceptor tyrosine-based activation motifs (ITAMs) and inhibitory receptors containing ITIMs. Upon ligand binding, tyrosine residues within the inhibitory receptor ITIMs become phosphorylated and serve as docking sites for proteins containing Src homology (SH)2 domains, such as SHP phosphatases. This leads to dephosphorylation of cellular proteins, thereby downregulating activated signaling pathways involving phosphorylation, such as those induced by ITAMs. CD33 is an inhibitory receptor. It can be constitutively activated by sialic acid-containing glycoproteins and glycolipids. The activated CD33 receptor recruits inhibitory proteins, such as SHP phosphatases, through its ITIM domain, thereby inhibiting cellular functions such as phagocytosis.
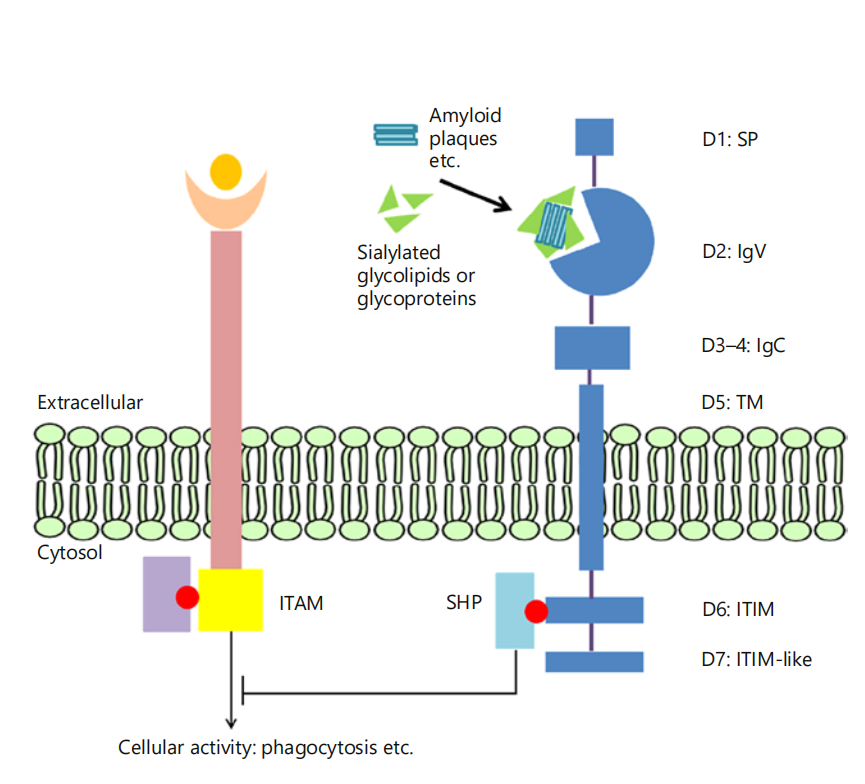
(Data source: Lingzhi Zhao. Gerontology. 2018)
Clinical value of CD33:
The clinical value of CD33 is mainly reflected in its potential as a therapeutic target, especially in the treatment of acute myeloid leukemia (AML) and Alzheimer's disease (AD).
Treatment of acute myeloid leukemia (AML): CD33 is a key marker on the surface of AML cells. Due to its high expression on AML cells and low or absent expression on normal hematopoietic stem cells, CD33 has become an ideal target for AML treatment. The antibody-drug conjugate (ADC) gemtuzumab ozogamicin is a CD33-targeted therapy. It binds to CD33 and is internalized by AML cells, subsequently releasing cytotoxins that cause cancer cell death. Gemtuzumab ozogamicin is the first and only CD33-targeted drug approved by the US Food and Drug Administration (FDA) for the treatment of AML.

(Data source: Noa E Wijnen, at al. OncoTargets and Therapy. 2023)
With the rapid development of immune-directed therapies, such as chimeric antigen receptor (CAR)-T cells, which have shown outstanding clinical activity against B-cell malignancies including acute lymphoblastic leukemia (ALL), CD33-targeted CAR-NK therapy provides a potentially effective treatment for AML by specifically recognizing the CD33 antigen on AML cells, activating and enhancing the killing ability of NK cells.
Treatment of Alzheimer's disease (AD): In AD, CD33 is associated with impaired clearance of Aβ plaques. Because CD33 expression on microglia is associated with Aβ clearance, therapeutic strategies targeting CD33 may help improve Aβ clearance and thus slow the progression of AD.
Research into CD33 as a therapeutic target has driven the development of novel therapeutics, including bispecific antibodies, radioimmunoconjugates, and CAR-modified immune effector cells. Clinical trials of these therapies are ongoing, aiming to improve therapeutic efficacy and minimize side effects. Overall, the clinical value of CD33 lies in its potential as a therapeutic target, particularly in the treatment of AML and other hematologic cancers. By targeting CD33, more precise treatment regimens can be designed to enhance efficacy and minimize effects on normal cells. As research deepens, the application of CD33 in the treatment of other diseases may also become apparent.
