CD36 is a scavenger receptor expressed in multiple cell types that mediates lipid uptake, immune recognition, inflammation, molecular adhesion and cell apoptosis. It plays an important role in the treatment of blood diseases and cancer and is a very potential disease treatment target.
Distribution of CD36
CD36 is expressed in a variety of tissues, including endothelial cells, cardiomyocytes, renal tubular epithelial cells, hepatocytes, adipocytes, platelets, and macrophages, and is involved in many pathophysiological processes, including immune regulation and metabolic regulation.
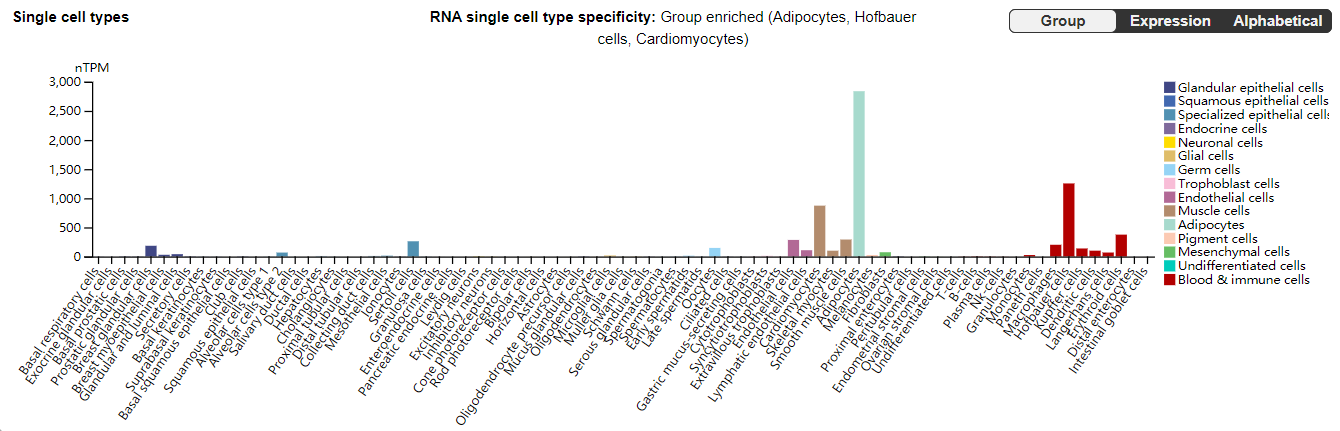
(Data source: The human protein atlas)
Structure of CD36:
The CD36 gene is located on chromosome 7q11.2 and consists of 15 exons. The CD36 protein has 472 amino acids and a protein size of approximately 53 kDa, but due to glycosylation, its actual molecular weight is 88 kDa . CD36 contains two phosphorylation sites and four palmitoylation sites located at the NH2 and COOH termini. Two ubiquitination sites are also found at the COOH terminus. The protein contains two transmembrane domains, a large glycosylated extracellular domain, and two short cytoplasmic tails at the N and C termini.
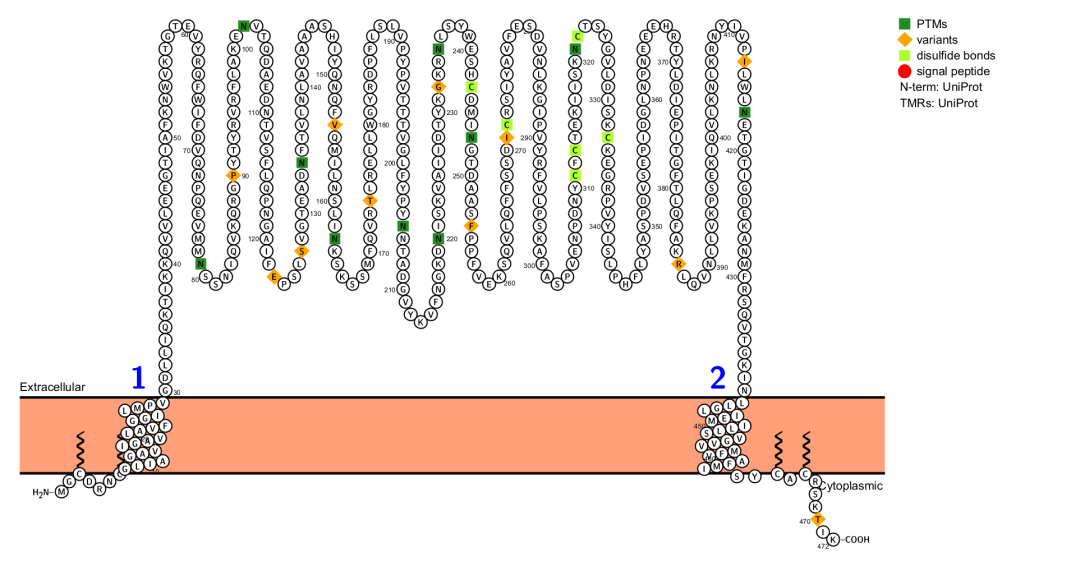
(Data source : Uniprot)
CD36 signaling pathway and regulation:
CD36 has dual functions as a signal transducer and fatty acid transporter. In response to extracellular signals (such as DAMPs), CD36 assembles and interacts with other membrane receptors (including NKA, TLRs, integrins, and tetraspanins) to form distinct signaling complexes. These signaling complexes then transmit signals to various downstream effector molecules, including SFKs, MAPKs, AMPK, the guanine nucleotide exchange factor Vav, and nicotinamide adenine dinucleotide phosphate oxidases of the NOX family. Activation of these effectors leads to the generation of reactive oxygen species (ROS) and the activation of transcription factors, including peroxisome proliferator-activated protein kinase (PPAR-γ) and nuclear factor-κB. Concurrently, CD36 binds to and promotes the transport of LCFAs across the plasma membrane, thereby influencing fatty acid metabolism and potentially promoting activation of the PPAR-γ pathway. The dual functions of CD36 ultimately integrate to lead to diverse cellular responses, such as migration, immune activation, differentiation, and cell growth/death.

(Data source: Chen Y, et al. J Exp Med. 2022)
CD36 and Disease
CD36 and diabetic cardiomyopathy
In a healthy heart, insulin typically activates the PI3K-Akt signaling pathway, promoting the transport of CD36 from the nucleus to the cell membrane. Simultaneously, the FOXO1 transcription factor promotes CD36 expression, and LCFAs absorbed by CD36 are oxidized as mitochondrial substrates and stored as fat. However, in diabetes, increased insulin levels more strongly activate PI3K-Akt, promoting the transport of more CD36 from the nucleus to the cell membrane. Excessive distribution of CD36 on the cell membrane leads to increased LCFA uptake, which in turn leads to triglyceride accumulation and insulin resistance. Insulin resistance and ROS assembly can worsen cardiac function and contribute to diabetic cardiomyopathy. Therefore, inhibiting the uptake of long-chain fatty acids by targeting CD36 may help improve diabetic cardiomyopathy.
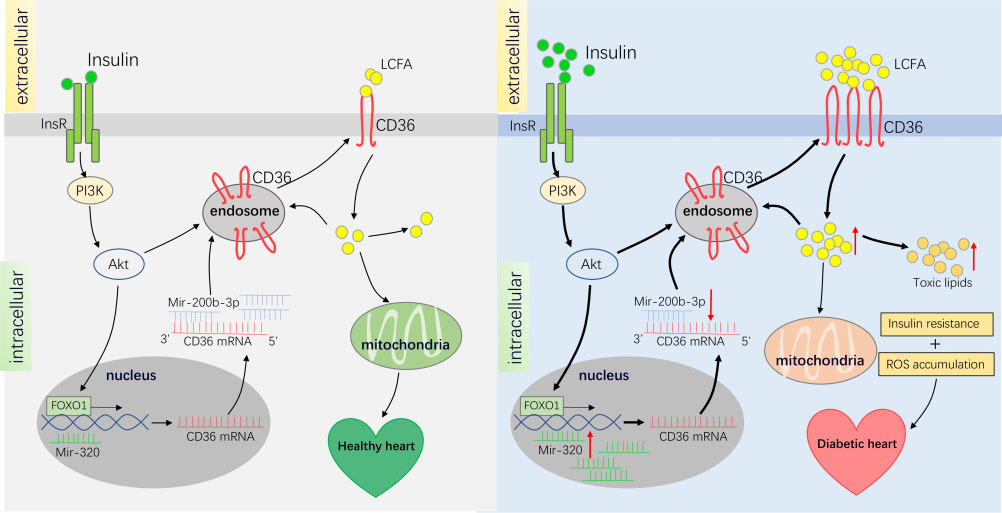
(Data source: Shu H, et al. Cardiovasc Res. 2022)
CD36 and atherosclerosis
CD36 plays a crucial role in atherosclerosis by mediating the uptake of oxidized low-density lipoprotein (oxLDL) in macrophages, leading to the formation of foam cells and inflammation in the arterial wall. Binding of oxLDL to CD36 activates signaling pathways that promote lipoprotein uptake, foam cell formation, and proinflammatory cytokine secretion, contributing to the development of atherosclerosis. Furthermore, CD36-mediated oxLDL uptake in macrophages can lead to metabolic reprogramming toward glycolysis, resulting in increased reactive oxygen species (ROS) and proinflammatory activation. In non-immune vascular cells, such as vascular smooth muscle cells, CD36 signaling in response to oxLDL can promote neointimal hyperplasia, further exacerbating atherosclerosis. These findings suggest that targeting CD36 may be a potential therapeutic strategy for atherosclerosis.
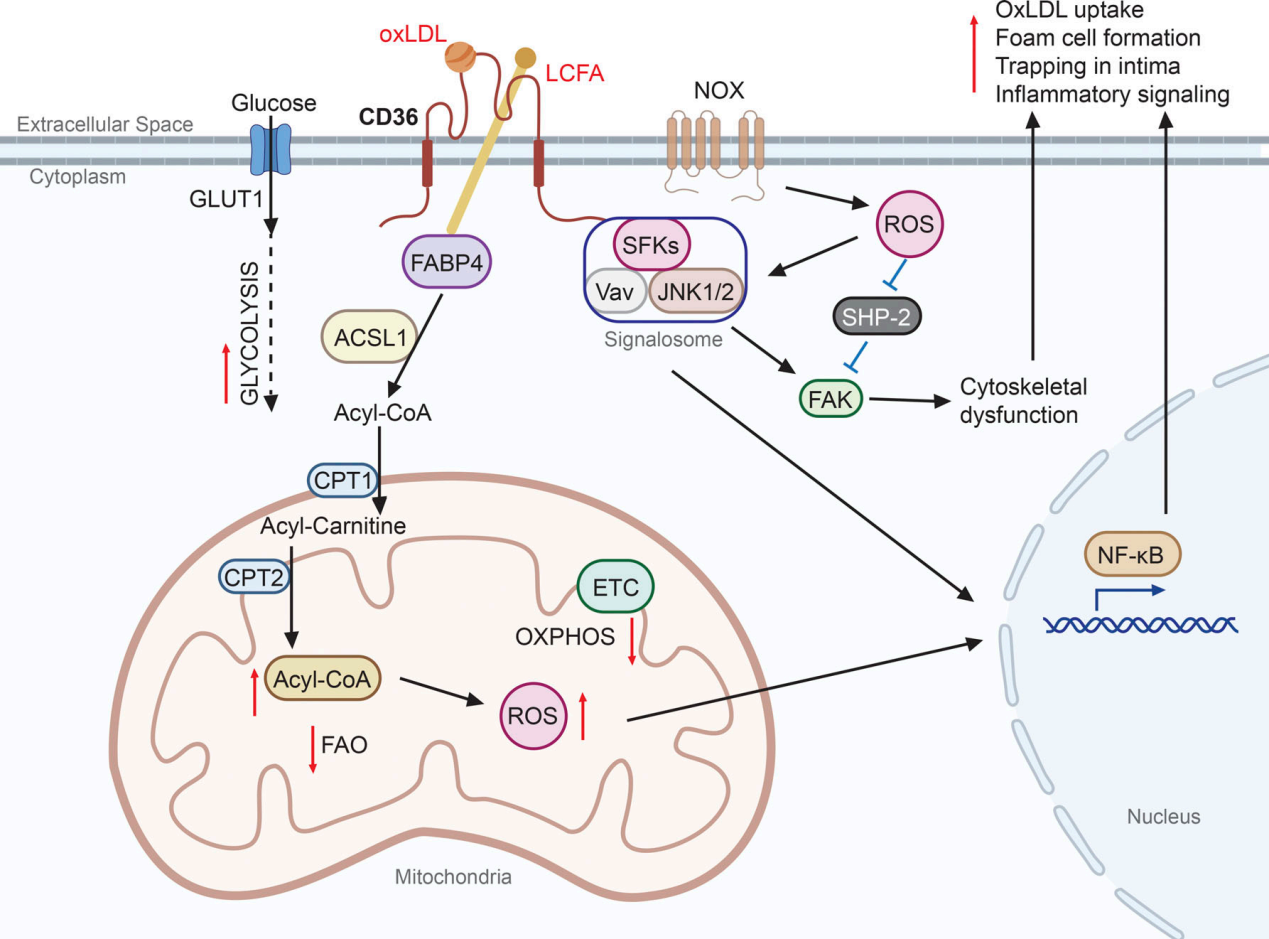
(Data source: Chen Y, et al. J Exp Med. 2022)
CD36 and cancer
CD36 plays a crucial role in immune regulation within the tumor microenvironment. Within tumor tissue, CD36 influences tumor immunity by regulating cytokine production, antigen presentation, phagocytosis, and immune tolerance. In macrophages, CD36 works with TLRs to recognize and phagocytose pathogens and pathogen-associated molecules, such as LPS from various bacteria, thereby participating in the innate immune response. Furthermore, CD36-mediated endocytosis of oxidized low-density lipoprotein (ox-LDL) can trigger sterile inflammation, thereby impacting immune regulation within the tumor microenvironment. CD36 also recognizes phosphatidylserine on the surface of apoptotic cells and, by binding to αvβ5 integrin, activates CD8+ T cells, thereby mediating the killing of malignant or infected cells. Reducing CD36 expression may help tumor cells evade the immune system. These findings suggest that targeting CD36 may be a potential strategy for tumor immunotherapy.
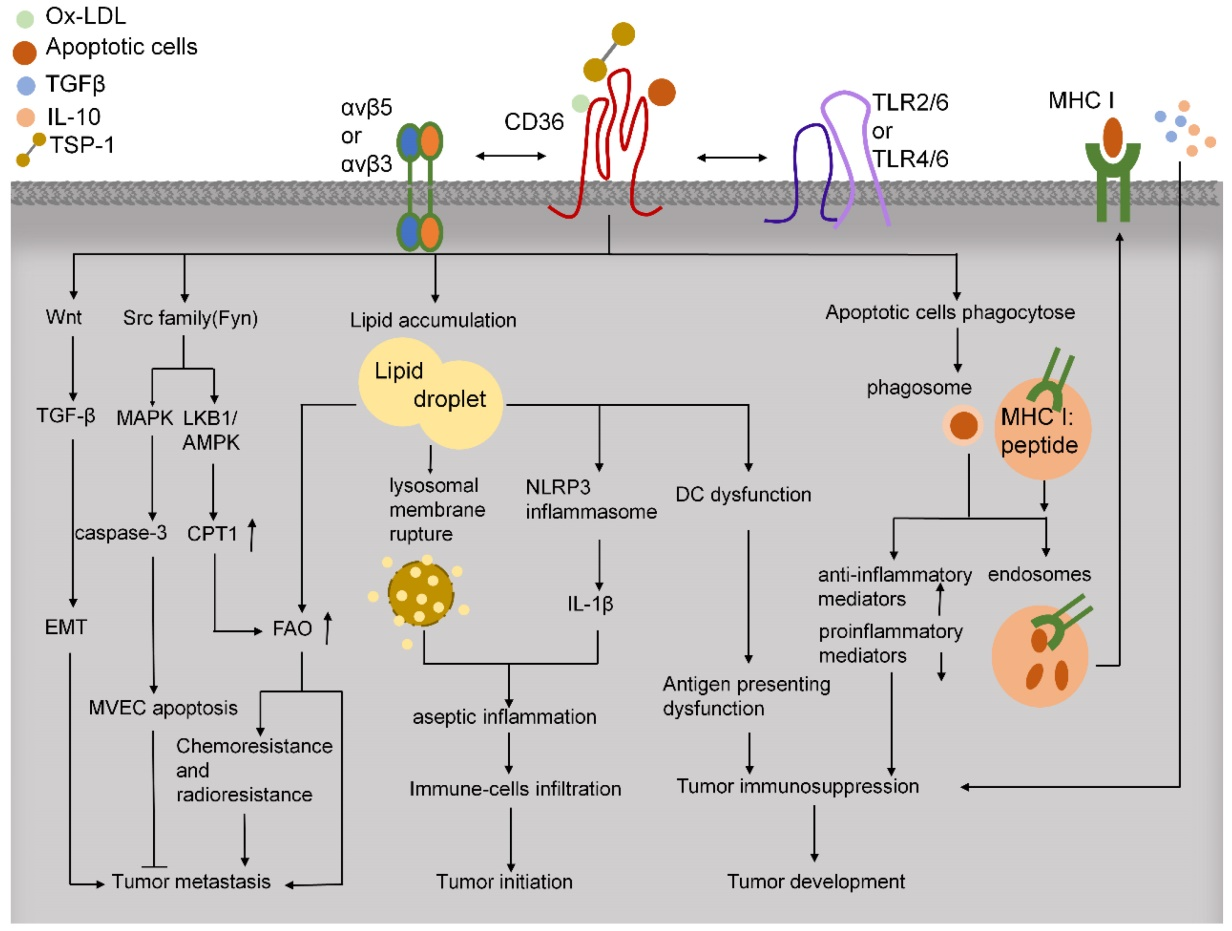
(Data source: Wang J, et al. Theranostics. 2019)
CD36 as a therapeutic target
CD36, a therapeutic target, has a broad expression pattern and diverse roles in various cancers. Historically, most inhibitors developed against CD36 have been small molecule inhibitors or fatty acid analogs. SSO, a fatty acid analog, inhibits CD36 uptake of long-chain fatty acids and oxidized low-density lipoprotein by irreversibly binding to the CD36 FA binding site Lys164. Numerous other structurally diverse small molecules exhibit anti-CD36 activity, including natural products like salvianolic acid (SAB) and MTN, as well as novel approaches to synthetically conjugating lipid tails to platinum-based chemotherapy payloads to generate cytotoxic fatty acid analogs. Furthermore, activation of the TSP-1-CD36-mediated apoptotic signaling pathway is another novel approach to targeting CD36. Currently, VT1021 is the only CD36-targeted drug in clinical trials for cancer treatment, having demonstrated promising results in Phase I/II trials for ovarian cancer, pancreatic cancer, triple-negative breast cancer, and glioblastoma, and has entered Phase II/III trials for glioblastoma. In addition to cancer, CD36 inhibition may also play a role in the treatment of other metabolic diseases, such as atherosclerosis, diabetes, etc. These studies suggest that CD36 inhibition may bring multiple mechanistically different therapeutic benefits to cancer patients.
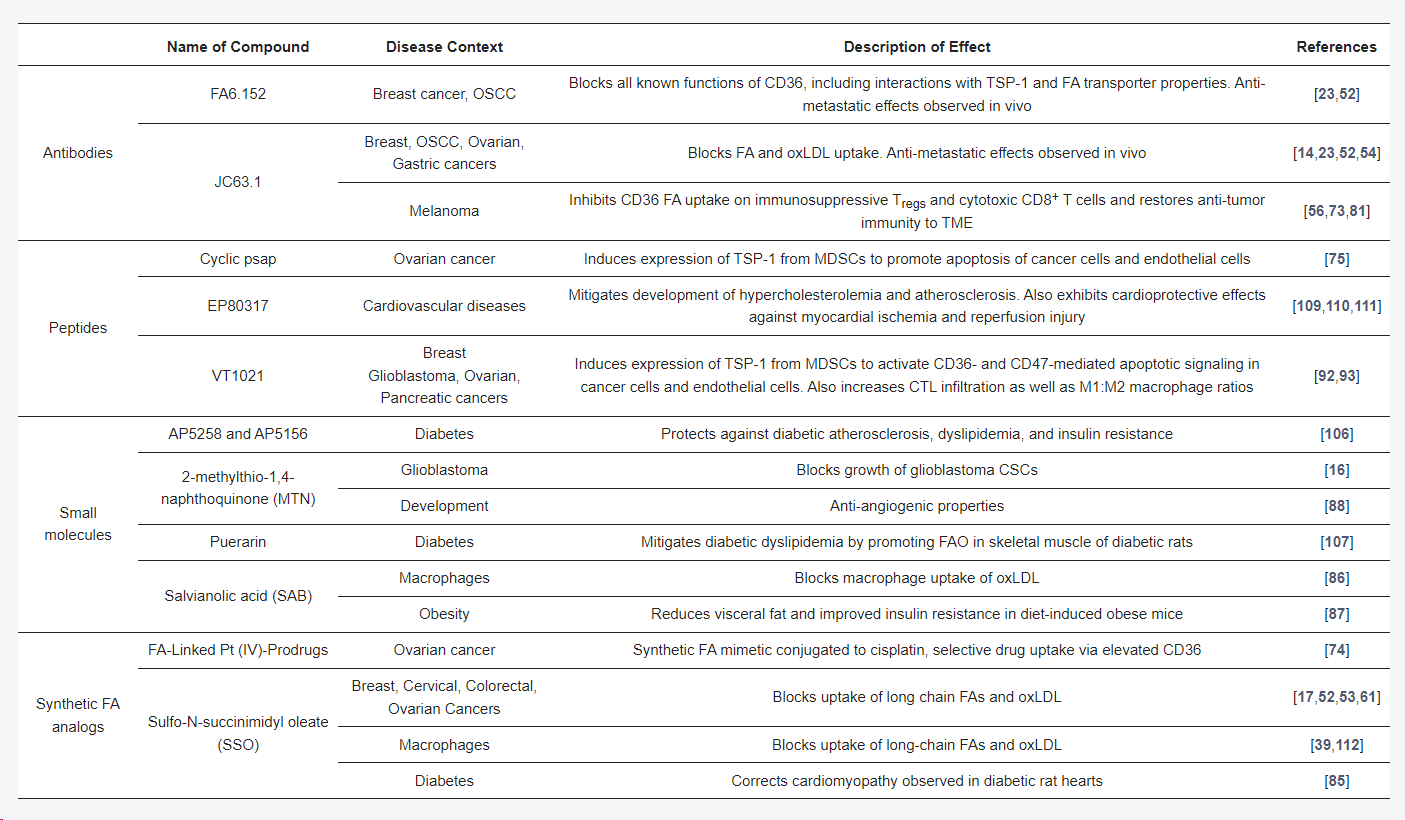
(Data source: Feng WW, et al. Cells, 2023)
