Background
Bispecific antibodies (bsAbs) are a promising drug modality that can simultaneously bind to two distinct epitopes on the same or different antigens. So far, 14 bsAb drugs have been approved for marketing. Despite this success and the diverse formats designed to date, producing bsAbs with high purity and stability remains challenging.
On March 25, 2024, Swedish researcher Nyesiga B et al. published an article in mAbs titled“RUBY® -a tetravalent (2+2) bispecific antibody format with excellent functionality and IgG-like stability, pharmacology and developability properties.”The study developed a unique bispecific antibody format called RUBY, which has the characteristics of full-length IgG in terms of stability, manufacturability, half-life and functionality, while having a 2+2 geometry, that is, it can simultaneously bind to two targets in a bivalent manner , providing new ideas for the development of tumor-targeting bispecific antibodies and new therapeutic concepts and clinical research.

Structural design of RUBY
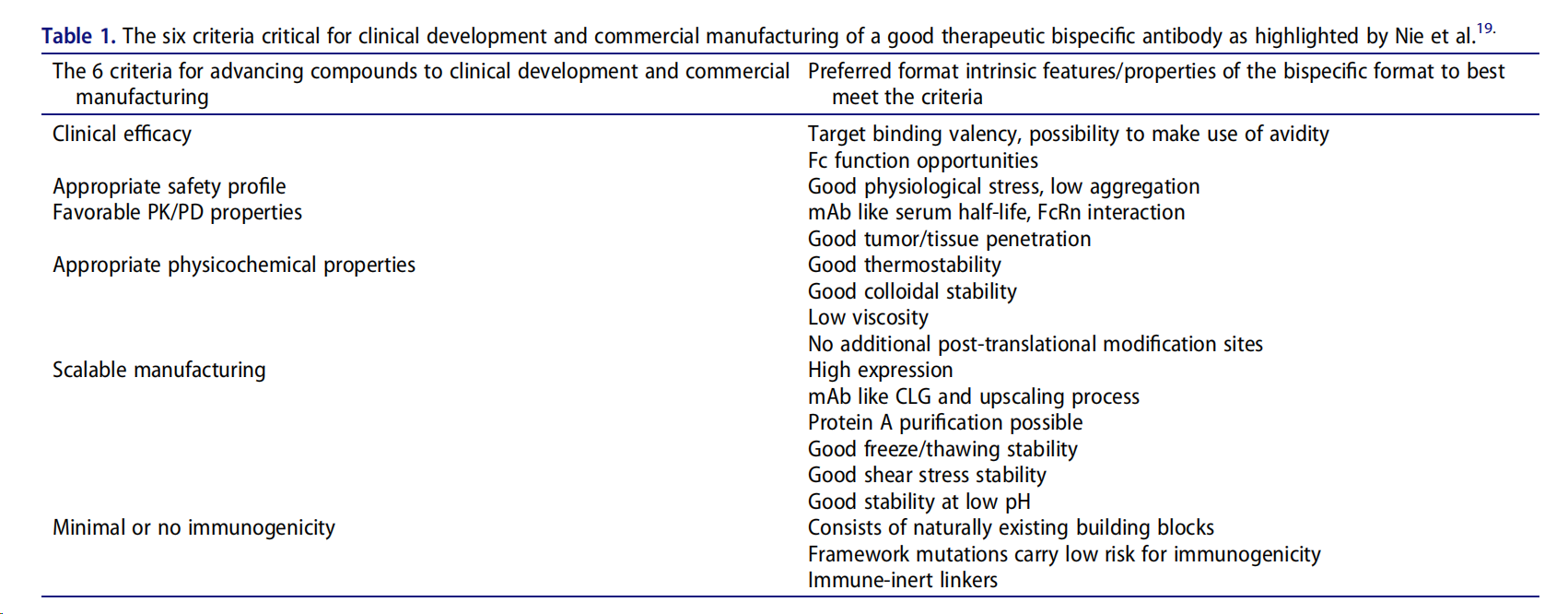
RUBY's molecular design utilizes a unique architecture consisting of an IgG linked to two Fab domains, connected at the C-terminus of the IgG via a polypeptide linker (3×G4S). The RUBY bsAb is therefore composed of six polypeptide chains, comprising two copies of three different polypeptide chains: 1) a long chain consisting of the IgG heavy chain and the light chain of the Fab; 2) a light chain bound to the VH1 and CH1 domains of the IgG; and 3) a light chain bound to the short heavy chain of the Fab attached to the IgG. Because the Fabs are attached via their light chains, the classic problem of light chain mispairing is eliminated in RUBY; correctly paired products will contain all correct chains. This is a valuable feature, as light chain mispairing can lead to significant variations in drug potency. A second key feature of the RUBY bsAb format is the use of interface mutations between the VH-CH1 and VL-CL pairs to minimize the formation of aggregates and soluble antigen-binding fragments, as well as the introduction of mutations to reduce the risk of immunogenicity, thereby reducing the risk of MHC II-binding peptides.
Stability validation of RUBY bsAb
Selected RUBY bsAbs underwent developability assessment, including studies measuring temperature, shear stress, colloidal, freeze-thaw, serum, and pH stability.
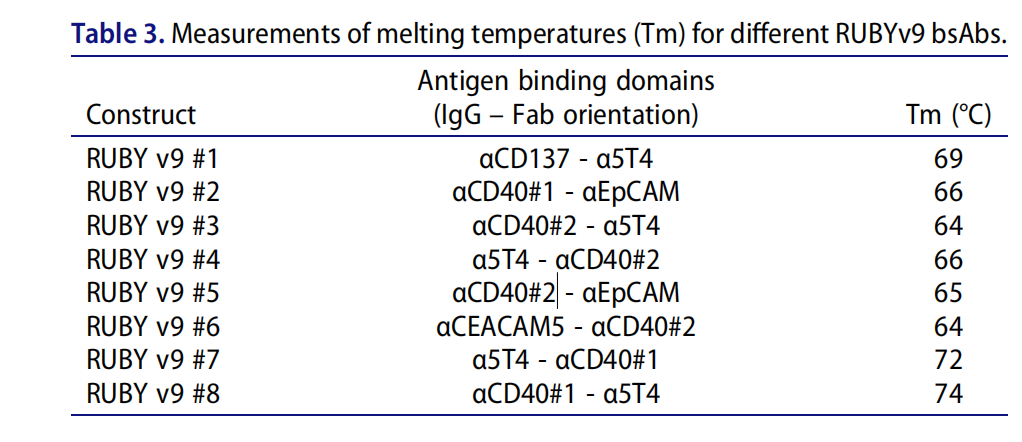
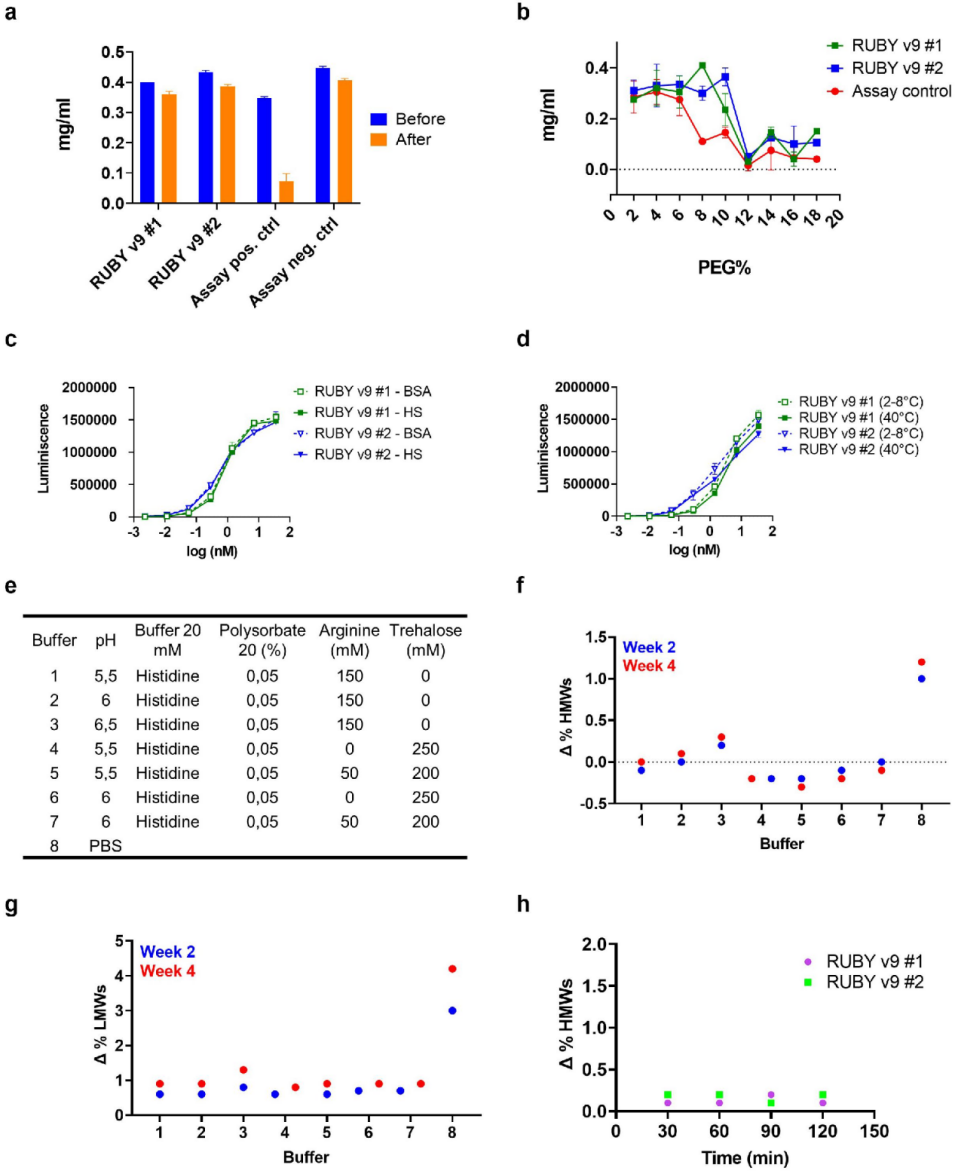
The bsAbs exhibited excellent thermal stability between 64°C and 74°C , as well as good shear stress and colloidal stability (PEG concentrations exceeding 9% were required to induce 50% protein loss). Serum stability was verified by dual ELISA, demonstrating good stability under physiological conditions , with no degradation at high temperatures or after freeze/thaw. They were also highly stable at high protein concentrations and were insensitive to low pH. These results further demonstrate the potential of RUBY as a universal format for stabilizing bsAbs.
Antigen-binding properties and functions of RUBY bsAb
As verified by dual ELISA, RUBY bsAb was able to bind to its respective targets simultaneously, and the position of the antigen-binding domain in IgG or attached Fab had little effect on its antigen-binding ability.
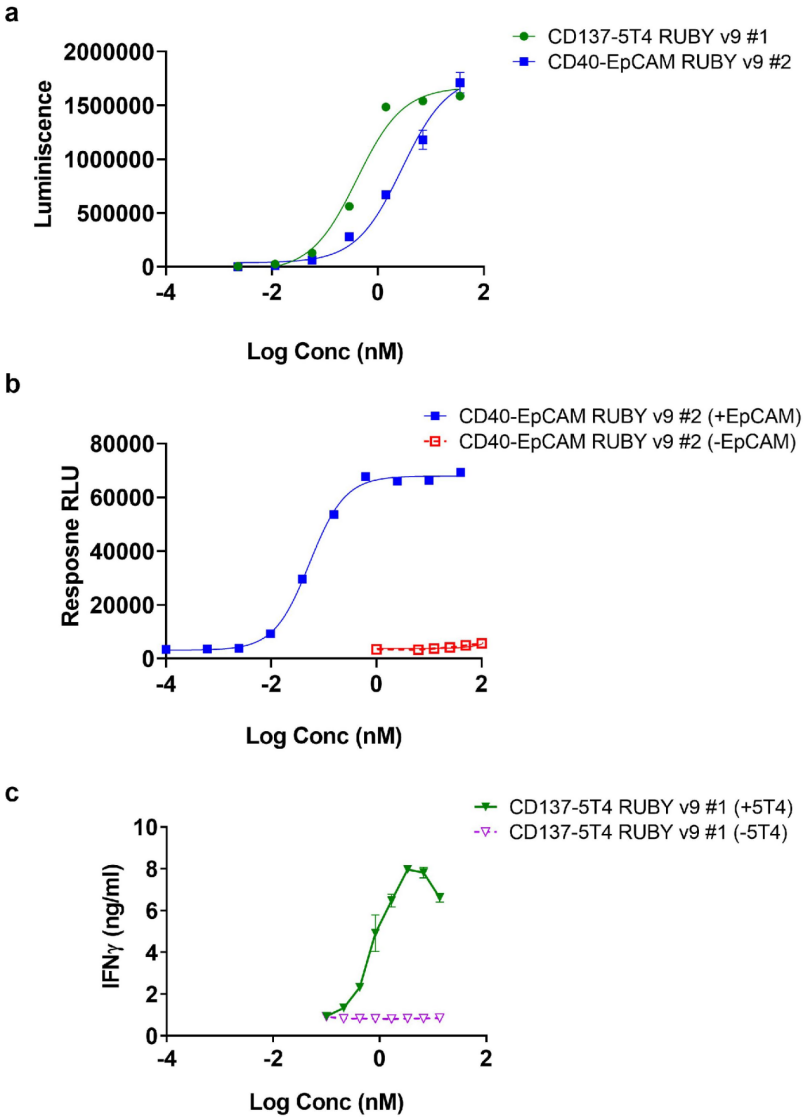
The CD40-EpCAM RUBY bsAb demonstrated conditional CD40 activation: the bsAb induced activation of CD40-expressing cells only in the presence of EpCAM expressed on CHO cells, whereas no activation was observed in a similar setting with CHO cells lacking EpCAM expression. The CD137-5T4 RUBY bsAb induced potent T cell activation, measured by a dose-dependent increase in interferon (IFN)-γ release in the presence of 5T4. In the absence of 5T4, there was no dose-dependent increase in T cell activity driven by the bsAb construct. T cell activation was mediated by CD137 crosslinking induced by 5T4-dependent clustering triggered by the bsAb. These data demonstrate the advantages of the RUBY format for generating conditionally functional bsAbs designed to tether immune cells to tumor cells and restrict activity to target tissues, thereby minimizing the risk of systemic toxicity.
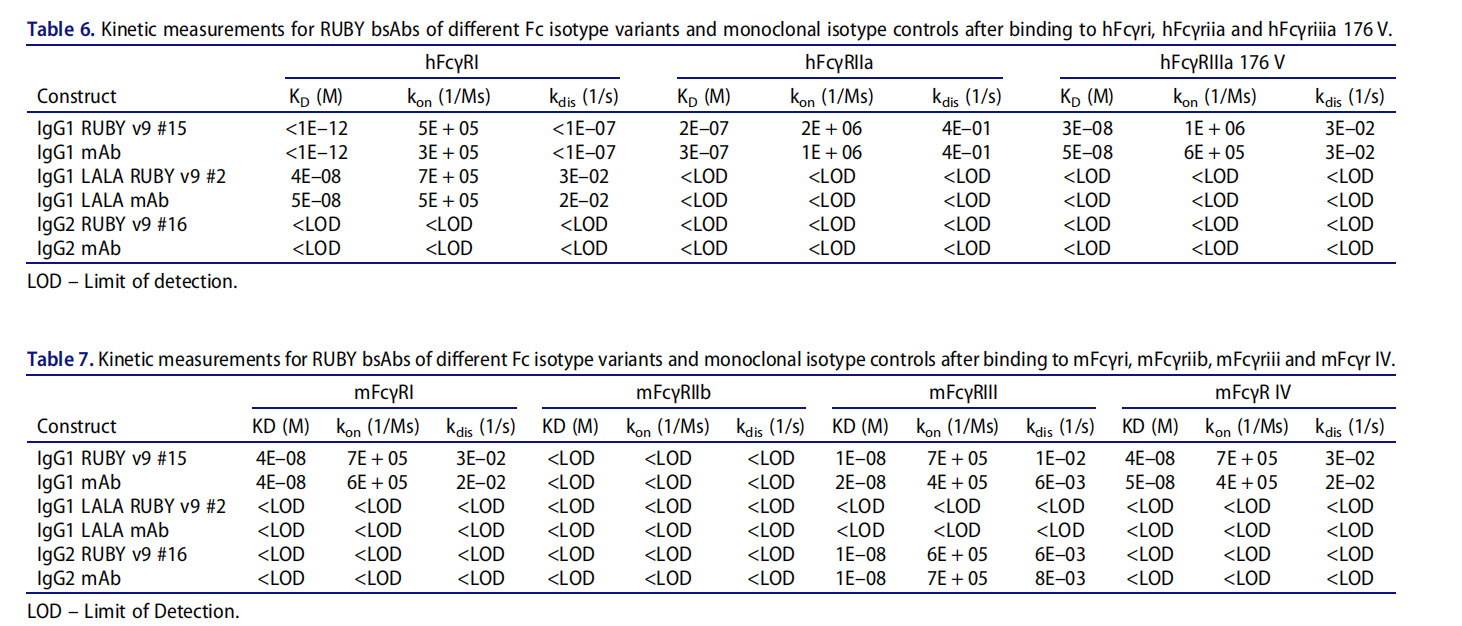
FcγR binding properties of RUBY bsAb
The RUBY bsAb has similar interactions with human Fcγ receptors; the IgG1 construct showed comparable binding to mFcγRI, FcγRIIa, and hFcγRIIIa 176 V, with characteristics comparable to monoclonal antibodies, allowing the development of biological functions that rely on this interaction.
Validation of RUBY bsAb in mice and cynomolgus monkeys
RUBY bsAbs exhibited FcRn binding and pharmacokinetic characteristics similar to those of monoclonal antibodies in mice and cynomolgus macaques. Furthermore, by evaluating the pharmacokinetic characteristics of RUBY bsAbs carrying LALA-silenced IgG1 Fc in mice, the serum concentrations of RUBY bsAbs were similar to those of the corresponding monospecific antibodies, with minimal difference in half-life. In cynomolgus macaque experiments, RUBY bsAbs also exhibited pharmacokinetic properties similar to those of monoclonal antibodies, and their stability and pharmacokinetic performance in different species were similar to those of monospecific antibodies. The addition of a C-terminal Fab and the introduction of mutations into the variable and constant domains of RUBY bsAbs had little or no effect on the PK properties of these bsAbs compared to monospecific antibodies.


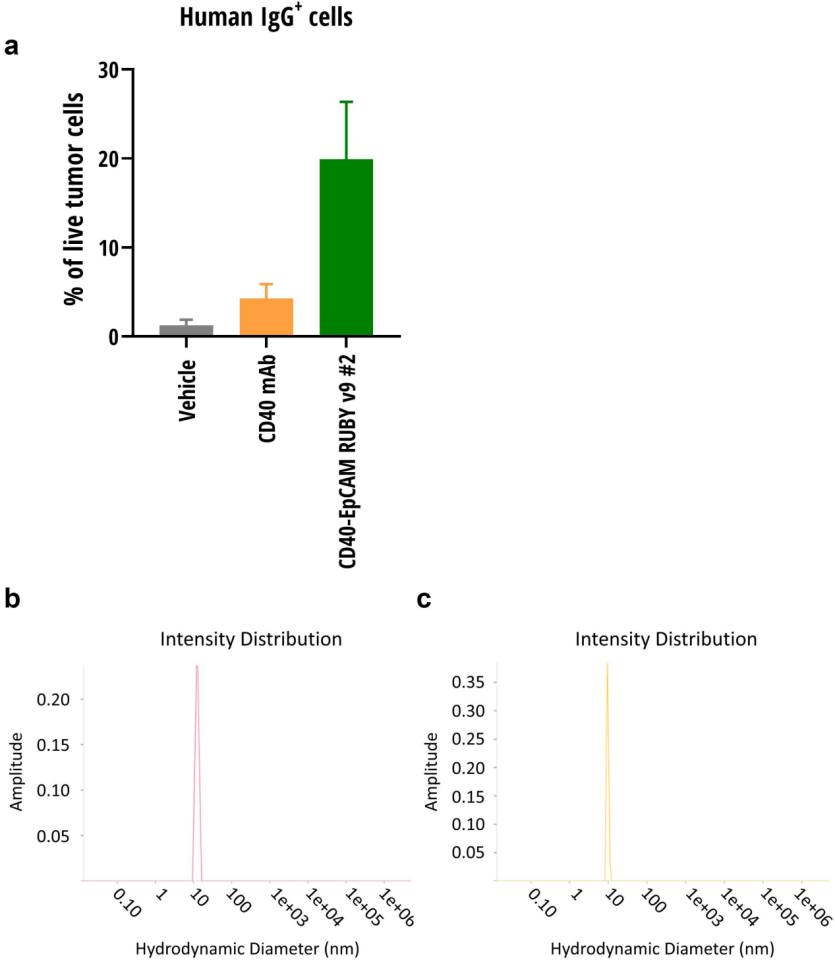
RUBY bsAbs localize to tumors in vivo
RUBY bsAbs demonstrate efficient tumor cell localization, effectively reaching and enriching in tumors expressing relevant antigens, while treatment with monospecific antibodies targeting CD40 results in fewer tumor cells expressing IgG. The ability of monospecific or bispecific antibodies to localize to tumors depends on several factors, such as size, affinity, and expression pattern of the target antigen. Dynamic light scattering (DLS) assessment of the hydrodynamic diameter of RUBY bsAbs reveals little difference compared to monospecific antibodies. The RUBY format is well-suited for developing bispecific antibodies for tumor localization or potentially other relevant tissues.
Summary
In this study, researchers developed a unique bispecific antibody format, called RUBY, with excellent biophysical and biological properties. RUBY employs specific mutations to limit the formation of aggregates and soluble Fab byproducts, reducing the risk of immunogenicity. This format demonstrates efficient tumor localization in vivo, with IgG-like properties and natural bivalent target interactions. Furthermore, RUBY demonstrates promising clinical development potential by improving stability and reducing the risk of immunogenicity.
