CD38 is a transmembrane glycoprotein with exoenzyme activity that also functions as a receptor and adhesion molecule. CD38 is expressed at low levels in normal lymphocytes, myeloid cells, and non-hematopoietic tissue cells, but is highly expressed in MM cells. Therefore, CD38 is an ideal target for MM treatment.
CD38 distribution
on multiple myeloma (MM) cells and plays a regulatory role in the occurrence and development of tumors. In addition, CD38 is highly expressed in B cells, macrophages, dendritic cells (DCs), innate lymphocytes (ILCs), natural killer (NK) cells, T cells, neutrophils, and monocytes.
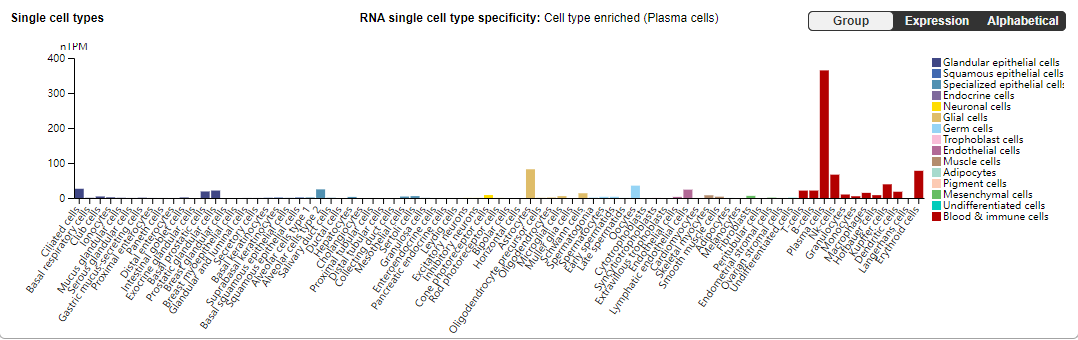
(Data source: the Human Protein Atlas)
CD38 structure and enzyme function:
CD38 is a type II transmembrane glycoprotein with a single transmembrane segment and a molecular weight of 46 kDa. It consists of a long extracellular region at the C-terminus, a single transmembrane domain, and a short cytoplasmic tail at the N-terminus. The tertiary structure of CD38 reveals that it is composed of two distinct domains: the NH2-terminal domain, composed of a bundle of α-helices and two short β-strands; and the COOH-terminal domain, consisting of a β-segment composed of four parallel β-strands surrounded by two long α-helices and two short α-helices. These two distinct domains are connected by a hinge region consisting of three peptide chains, including residues 118-119, 143-144, and 200-201. The relative conformation of these two distinct domains is further stabilized by a disulfide bond formed between Cys-119 and Cys-201. The overall structure is L-shaped, with the NH2-terminal domain primarily composed of α-helices and the COOH-terminal domain primarily composed of β-strands. This structure is very similar to the secondary structure of Aplysia cyclase.

(Data source: Malavasi F, et al. Physiol Rev. 2008)
CD38 is a bifunctional extracellular enzyme with both cyclase and hydrolase activities, involved in nucleotide metabolism. CD38 is primarily expressed on immune cells and metabolizes nicotinamide nucleotides (NAD+ and NMN) into ADPR and cADPR, leading to calcium mobilization. While intracellular CD38 is primarily present in the cytoplasm and cell membranes of organelles, the vast majority of CD38 activity occurs extracellularly, leading to the degradation of NAD+ precursors (such as NMN) required for NAD+ synthesis. The extracellular activity of CD38 has widespread implications for NAD+ homeostasis in the context of infection, metabolic dysfunction, aging, and tumor biology. The products of the catalytic reaction, such as ADPR and cADPR, serve as important second messengers in cells, affecting cell growth, insulin release, T cell activation, and other functions by regulating cellular calcium.

(Data source:Hogan KA, et al. Front Immunol)
Mechanism of action of CD38 antibodies
CD38 monoclonal antibodies (mAbs) have become an effective treatment for MM through multiple mechanisms, including complement-dependent cytotoxicity, antibody-dependent cell-mediated cytotoxicity, antibody-dependent cellular phagocytosis, programmed cell death, enzyme regulation, and immune regulation.

(Data source: Gao L, et al. Br J Cancer. 2023)
Clinical CD38-targeted antibodies
In 2015, Daratumumab (Darzalex), a fully human anti-CD38 antibody developed by Johnson & Johnson, was approved for the treatment of patients with relapsed and drug-resistant multiple myeloma. Daratumumab became the first CD38 antibody on the market.
On March 2, 2020, Sanofi's oncology drug Isatuximab received FDA approval. This is the second CD38 antibody drug after Daratumumab, also for the treatment of multiple myeloma. Currently, both Daratumumab and Isatuximab have shown significant efficacy in the treatment of multiple myeloma.
Recently, a new humanized CD38 monoclonal antibody called SAR442085 has significantly enhanced affinity for activated Fcγ receptors (Fcγr), thereby enhancing anti-tumor activity against multiple myeloma. Compared with daratumumab and isatuximab monoclonal antibodies, SAR442085 has been shown to have a higher binding affinity for CD38. These observations highlight the preclinical efficacy of SAR442085 and support further clinical trials of this novel anti-CD38 antibody in patients with multiple myeloma.
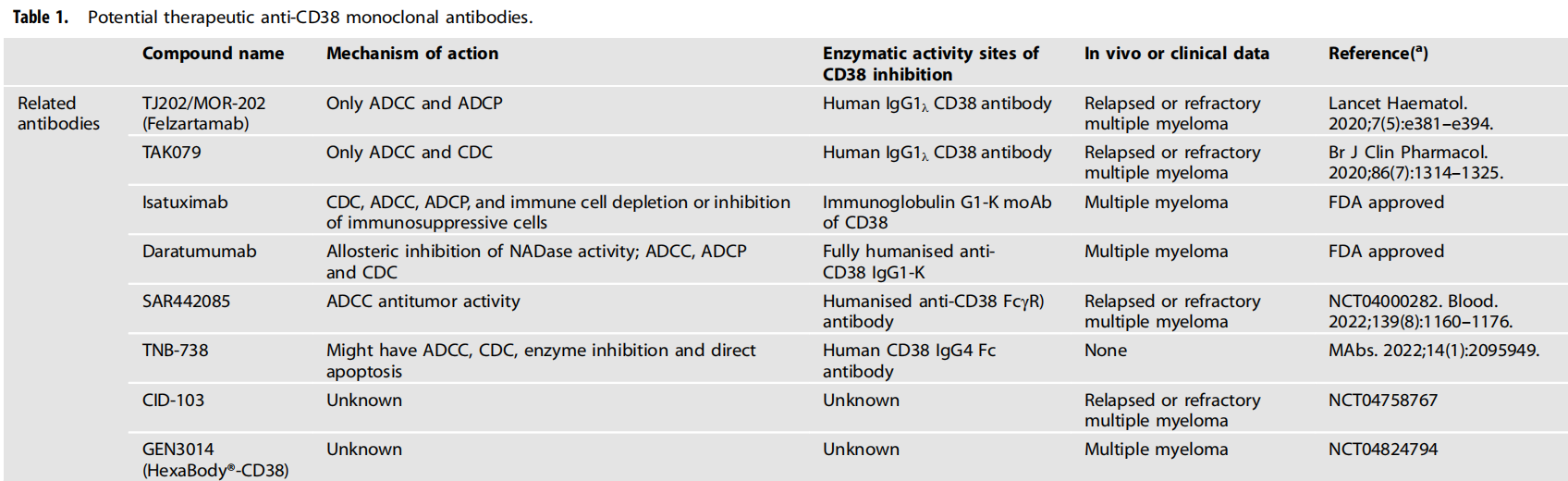
(Data source: Gao L, et al. Br J Cancer. 2023)
Potential markers of CAR-T cell exhaustion
In recent years, CAR-T cell therapy has become one of the most promising treatments for hematological malignancies and autoimmune diseases. CD38 has been identified as a potential marker for CAR-T cell exhaustion. Inhibiting CD38 enzymatic activity can enhance the cytotoxicity and anti-tumor efficacy of CAR-T cells. Inhibiting CD38 can reverse CAR-T cell exhaustion and improve CAR-T cell function. Mechanistically, CD38 inhibition inhibits glycolysis through specific signaling pathways, indicating the potential clinical application of CD38 inhibition in enhancing CAR-T cell therapy.

(Data source: Huang Y, et al. Cell Rep Med. 2024)
