Background:
Ethylpyrimidine, a typical pyrimidine fungicide, has been widely used for over 50 years to control powdery mildew on various fruits and vegetables. Excessive use of ethylpyrimidine can lead to accumulation of residues on the skins of major agricultural products, reaching 0.012-0.061 meq/kg. Long-term exposure to ethylpyrimidine in humans may lead to elevated blood urea concentrations, increased relative liver weight, and even urinary incontinence and thyroiditis. Children aged 2-14 years face increased non-carcinogenic health risks from long-term exposure to ethylpyrimidine.
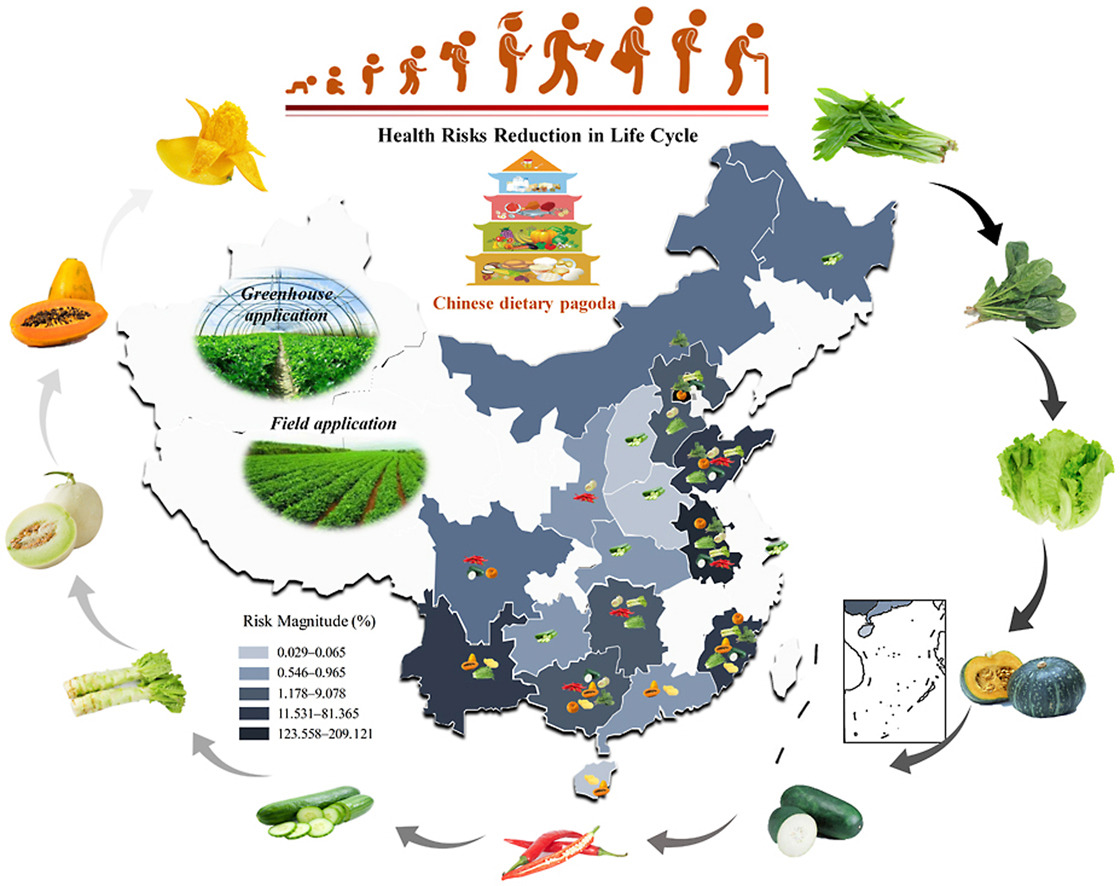
(Data source: hen Z, et al. Chemosphere. 2023)
Currently, the detection of ethoxyphenol is mainly based on liquid chromatography, tandem mass spectrometry, and gas chromatography-tandem mass spectrometry, but these methods require expensive equipment, professional personnel, and time-consuming sample preparation. Therefore, there is an urgent need for an on-site, high-throughput, rapid, highly sensitive, and reliable analytical method to detect ethoxyphenol residues in crops and the environment.
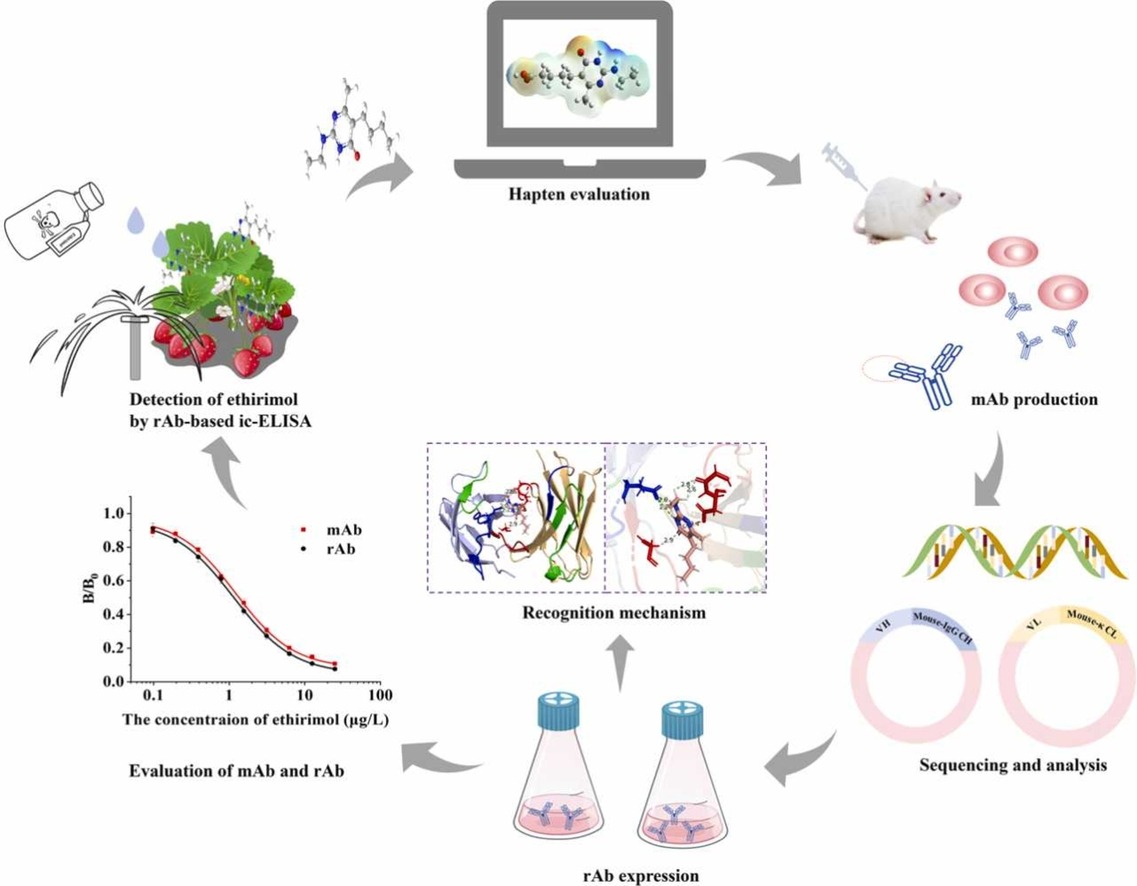
(Data source: Lei X, et al. J Hazard Mater. 2024)
Recently, a team from the Institute of Agricultural Product Quality Standards and Testing Technology of the Chinese Academy of Agricultural Sciences published their latest research results in J Hazard Mater: they obtained a new monoclonal antibody against ethoxypyrimidine, studied the structural mechanism of the recombinant antibody recognizing the new hapten of ethoxypyrimidine through molecular simulation, and developed an indirect competitive enzyme-linked immunosorbent assay (ic-ELISA) method to quickly and conveniently detect ethoxypyrimidine in real samples.

Hapten design:
For small molecules, introducing a suitable spacer arm can ensure optimal exposure of characteristic groups to the immune system while maintaining coupling distance from the carrier protein. Researchers employed two opposite orientations for the ethynol spacer arm and designed two hapten structures, hapten01 and hapten02, accordingly. Surface electrostatic potential analysis revealed that in hapten02, N6, N7, and O16 were more prominently exposed, promoting immune recognition. Therefore, hapten02 was selected as the synthetic hapten molecule.

Preparation of immunogens & screening of antigens:
Small molecules possess antigenicity but lack immunogenicity. To induce immunogenicity, small molecules must be conjugated to a carrier protein to form a complete antigen. The active ester method was used to conjugate the hapten Hapten 02, a hapten of ethoxybenzol, to the carrier proteins BSA and OVA to synthesize the complete antigen. The molecular weights of Hapten-BSA and Hapten-OVA at their respective absorption maxima were 75991.440 and 47756.602, respectively. These values exceeded the absorption maxima of the BSA/OVA standard (67510.359 and 44721.036, respectively), indicating successful coupling of the hapten to the BSA/OVA surface.

Monoclonal antibody development and validation:
Using hybridoma technology, cell cloning and screening were performed, and the monoclonal cell line 14G5F6 was selected for subsequent experiments. The heavy chain of this antibody is of the IgG1 isotype, while the light chain is of the kappa isotype. The amino acid sequence of the variable region of mAb-14G5F6 was obtained through amplification and sequencing. This was then integrated into the expression vector PSTEP2-Mouse-IgG1-CH/κ-CL for recombinant expression to obtain rAb-14G5F6. The IC50 value of mAb-14g5f6 was 1.35 μg/L, with a detection range (IC20-IC80) of 0.34-6.26 μg/L and a correlation coefficient (R2) of 0.999 . These results confirm the rationality of the hapten structure. The IC50 value of rAb-14G5F6 was 1.15 μg/L, the detection range (IC20~IC80) was 0.27~4.89 μg/L, and the correlation coefficient (R2) was 0.999 , showing consistent recognition sensitivity with that of the parental mAb-14G5F6.
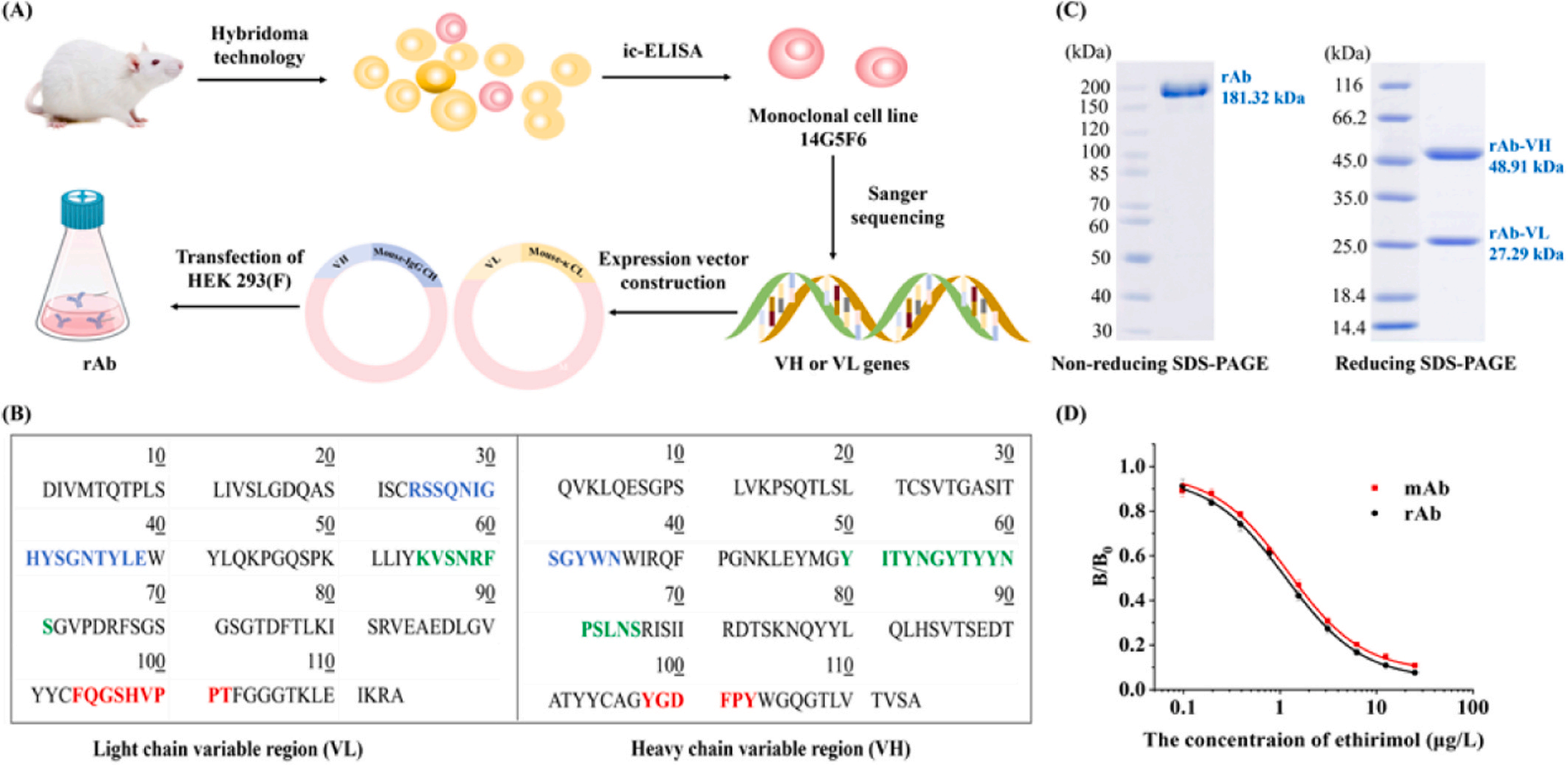
Antibody binding kinetics SPR assay:
Single-cycle SPR screening revealed that ethiprimol rapidly bound to mAb-14G5F6 and rAb-14G5F6 on the chip within a 60-second binding period, while other analogs showed weak or no binding. During the dissociation process, ethiprimol dissociated relatively slowly from the chip, while other analogs dissociated rapidly, indicating that the antibody had a greater binding affinity for ethiprimol than the other analogs. Multi-cycle kinetic analysis revealed KD affinity constants of 5.45×10⁻¹⁰mol/L for mAb-14G5F6 and 5.71×10⁻¹⁰mol/L for rAb-14G5F6. Both antibodies exhibited exceptional specificity and affinity for ethiprimol, consistent with the results from the ic-ELISA assay.

Immunoassay method development:
An immunoassay for the detection of ethynol in irrigation water and strawberry samples was developed using the rAb-14G5F6 antibody. The limit of detection (LOD) for the rAb-14G5F6 ic-ELISA assay was 0.036 mg/kg, significantly lower than the EU maximum residue limit for strawberries (0.3 mg/kg), meeting the monitoring requirements for ethynol in strawberries. The correlation curve slope (R 2) was close to 1 compared with the results obtained by liquid chromatography-mass spectrometry/mass spectrometry, demonstrating the reliability and accuracy of the rAb-14G5F6-based ic-ELISA.

Antibody molecular mechanism:
Based on homology modeling principles, the antigen-binding region of rAb-14G5F6 was predicted. The hydrogen atoms around N6 and N7 of ethipirol , as well as O16 and the pyrimidine ring, are key to the specific recognition of the ethipirol antibody. The hydrogen atoms at N6 and N7 of ethipirol act as hydrogen donors, forming traditional hydrogen bonds. The hydrogen atom at C9 acts as a hydrogen donor, forming a carbon-hydrogen bond with GLY108 in the antibody's CDRH3 as a hydrogen acceptor. Furthermore, GLY107 in CDRL3 acts as a hydrogen donor, forming a carbon-hydrogen bond with O16 of ethipirol as a hydrogen acceptor. These forces work together to form a stable antibody-ethipirol complex. A strong ionic bond is established between ASP (H114) of CDRH3 and the central pyrimidine ring of ethipirol, reinforcing the antibody's characteristic "fast association and slow dissociation" kinetics for ethipirol.

Summary:
In this study, the researchers successfully developed a high-throughput, low-cost small molecule ethidium bromide immunoassay based on a novel recombinant antibody, filling the gap in rapid detection methods for ethidium bromide. The expression of the recombinant antibody in mammalian cells retains the special characteristics of the original monoclonal antibody, including affinity, specificity, tolerance and actual sample detection capabilities.
