Celiac disease (CeD), also known as gluten-sensitive enteropathy, is an autoimmune disease of the small intestine associated with the ingestion of gluten. Gluten is present in foods containing wheat, barley, or rye. When celiac disease patients consume foods containing gluten, their gluten-specific CD4+ T cells recognize gluten against the backdrop of disease-associated human leukocyte antigen (HLA) molecules (most patients carry HLA-DQ2.5). This triggers an immune allergic reaction in the small intestinal mucosa, leading to its destruction and the inability of the small intestine to absorb nutrients from food.
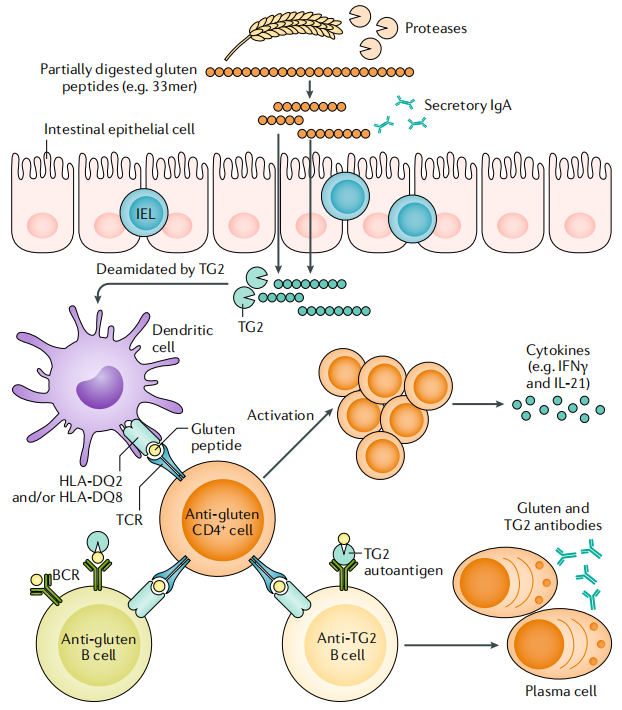
(Data source: Katri L, et al. Nat Rev Dis Primers. 2019)
So far, there are no approved drugs to treat CeD, and the only available treatment is a lifelong gluten-free diet (GFD). Given the central role of gluten-specific T cells in the pathogenesis of CeD, researchers at Chugai Pharmaceutical Co., Ltd. recently reported the development of a broadly reactive, high-affinity bispecific TCR-like antibody, DONQ52, which binds to over 25 different gluten pHLA-DQ2.5 epitopes, encompassing all immunodominant epitopes, and blocks gluten-dependent immune activation in vitro and in vivo while maintaining systemic immunity, potentially offering a novel approach for interventional therapy in CeD.

pHLA-DQ2.5 construct expression:
HLAa chain and β chain expression vectors were constructed separately to produce soluble MHC class II molecule HLA-DQ2.5:33mer (LQLQPFPQPELPYPQPELPYPQPELPYPQPQPF) complex for antibody development immunogens and screening (33mer gliadin peptide contains multiple pathogenic epitopes, such as DQ2.5-glia-α1a, DQ2.5-glia-α2 and DQ2.5-glia-α1b).
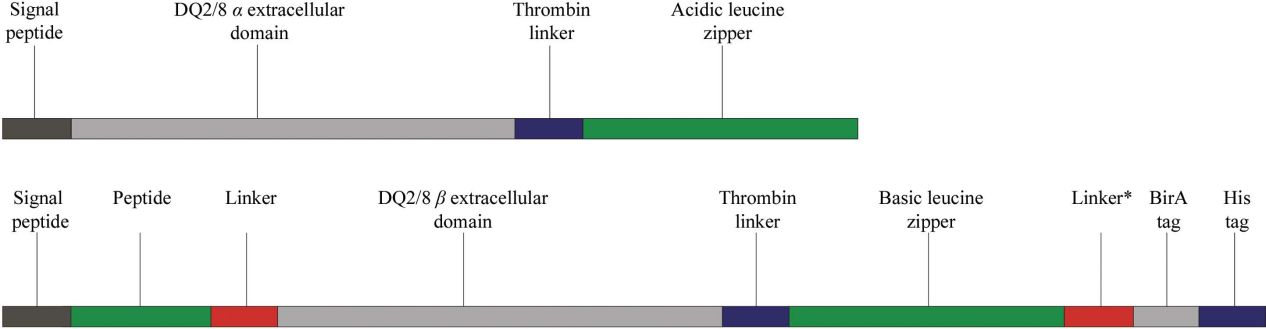
(Data source: Bianchi M, et al. Immunity. 2018)
Single B cell rabbit monoclonal antibody screening:
After immunizing rabbits with recombinant HLA-DQ2.5:33mer, B cells from the immunized rabbits were subjected to fluorescence-activated single-cell sorting (FACS) using the company's in-house rabbit monoclonal antibody development platform. B cells that bind to HLA-DQ2.5:33mer were further screened for binding to various HLA I peptides (pHLA II). Antibodies DQN0344xx and DQN0385ee, which broadly cross-react with gluten pHLA-DQ2.5, were selected as candidate molecules.

(Data source: Chugai Pharmaceutical Co., Ltd.)
DONQ52 dual-antibody modification:
The two original antibodies, DQN0344xx and DQN0385ee, were humanized. While maintaining antibody specificity, pharmacokinetics (PK), physicochemical properties, and immunogenicity, extensive mutation optimization was performed on the heavy and light chains of the antibodies. The optimal and effective mutations in DQN0344xx and DQN0385ee were screened one by one, and the DONQ52 bispecific antibody was generated using the FAST-Ig bispecific antibody platform. At the same time, the Fc was modified to enhance binding to neonatal Fc receptors under acidic pH conditions, improve PK, and reduce binding to Fc gamma receptors and C1q to eliminate ADCC and CDC effector functions.
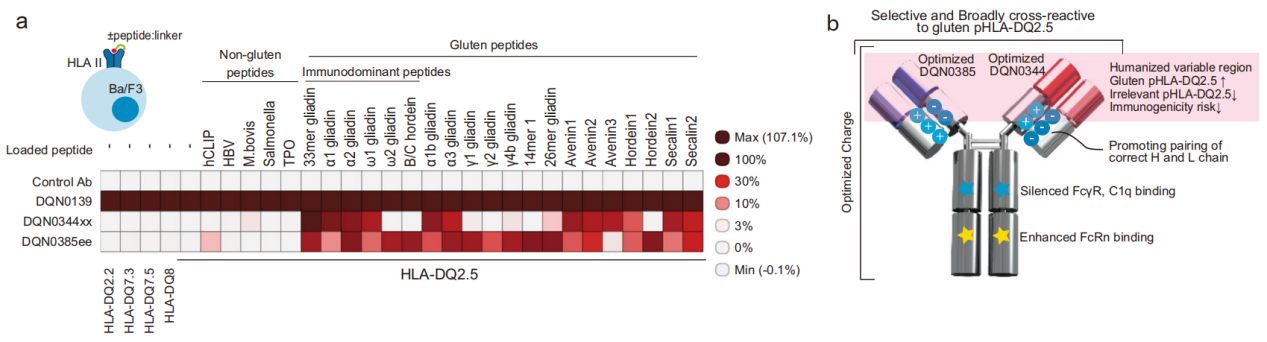
In vitro characterization of DONQ52:
The binding selectivity of DONQ52 was tested using a panel of diverse peptide HLA-DQ2.5 complexes. DONQ52 showed broad reactivity against pathogenic gluten peptide HLA-DQ2.5 complexes, while demonstrating no substantial binding to any of the gluten-unrelated peptide HLA-DQ2.5 complex molecules tested.

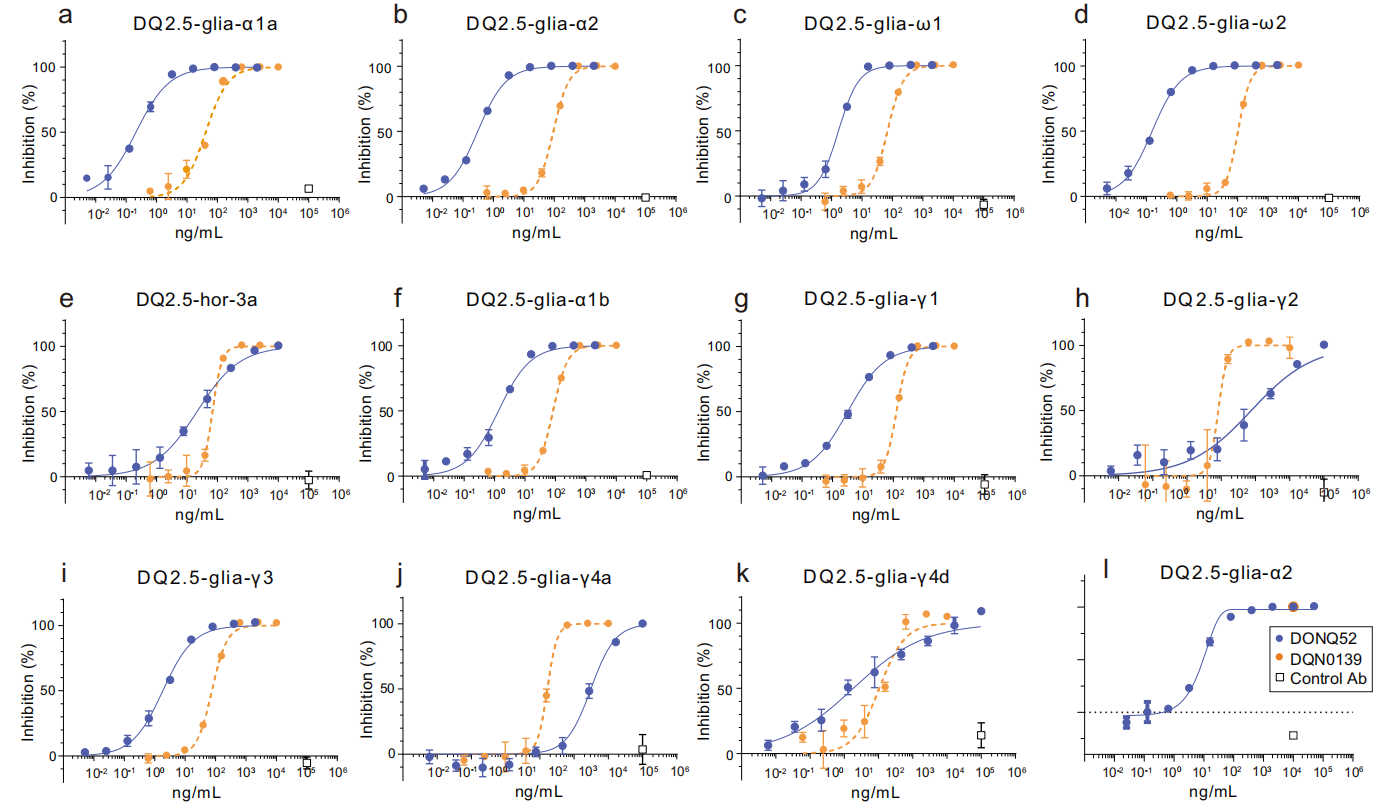
DONQ52 broadly neutralizes in vitro:
DONQ52 dose-dependently inhibited IL-2 production by human CD4+ T cells expressing specific TCRs and had neutralizing effects against five immunodominant epitopes (DQ2.5-glia-α1a, DQ2.5-glia-α2, DQ2.5-glia-ω1, DQ2.5-glia-ω2, and DQ2.5-hor-3a) and six additional CeD-associated epitopes (DQ2.5-glia-α1a, DQ2.5-glia-γ1, DQ2.5-glia-γ2, DQ2.5-glia-γ3, DQ2.5-glia-γ4a, and DQ2.5-glia-γ4d).
Structural basis of DONQ52 cross-reactivity:
Analysis of the X-ray crystal structure of the complex between the antibody fragment Fab and the antigen revealed that each arm of DONQ52 structurally recognizes multiple gluten epitopes with different amino acid sequences: Tyr100a (Tyr100b, Tyr99) in CDR3-VH on DQN0344xx and Tyr-50 (Tyr32) in CDR2-VL (CDR1-VL) on DQN0385ee appear to play a key role in the broad binding to various gluten peptide HLA-DQ2.5 complexes.

DONQ52 Pharmacokinetics:
Single-dose PK analysis showed that DONQ52 has a long half-life and high subcutaneous bioavailability without affecting gluten intake.
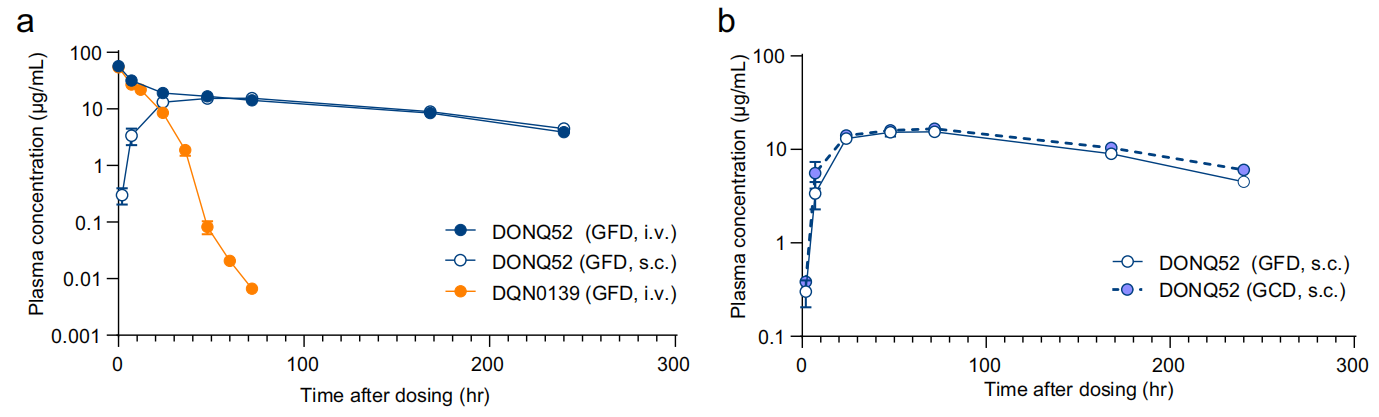
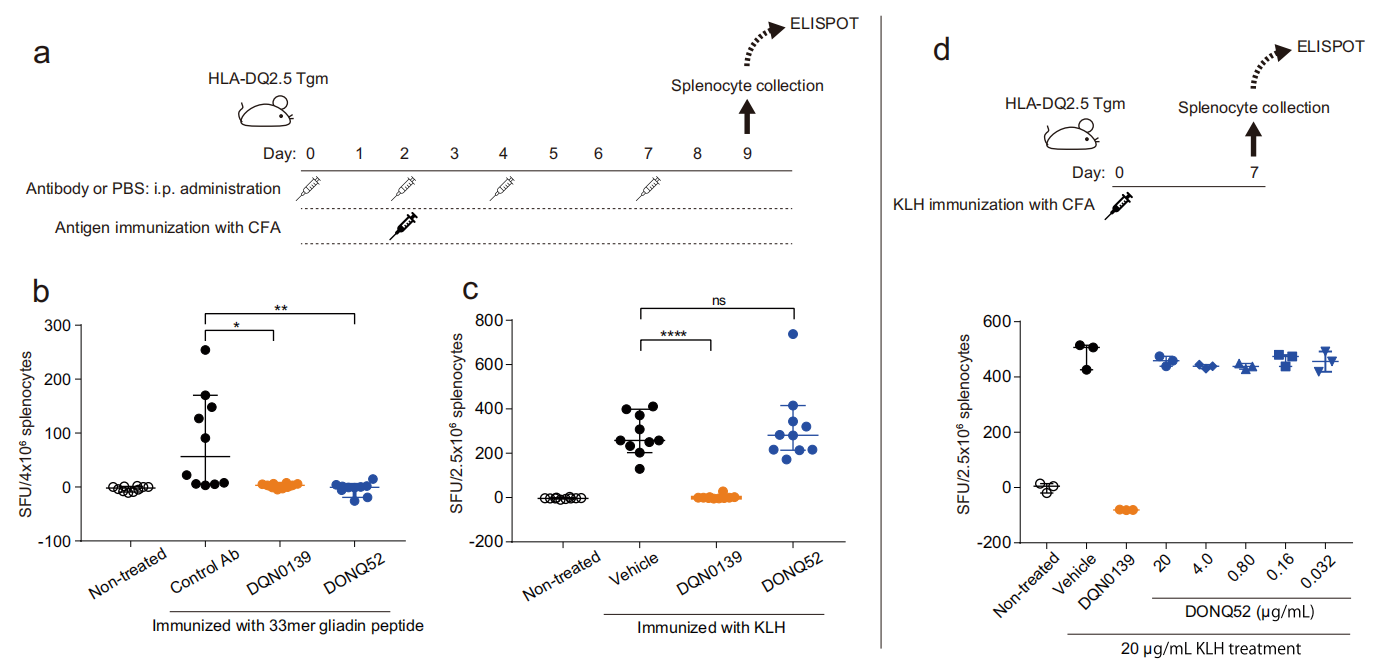
DONQ52 in vivo activity:
In vivo immunization of mice with HLA-DQ2.5:33mer or KLH protein induced antigen-reactive T cells. After intraperitoneal administration, DONQ52 specifically blocked 33mer protein peptide-dependent T cell induction without affecting the response to KLH.
Summary:
DONQ52 is a novel bispecific TCR-like antibody that can broadly neutralize more than 25 different gluten peptide HLA-DQ2.5 complexes, providing an effective, safe and patient-friendly potential treatment for CeD. Phase I clinical trials are currently underway.

