Coagulation factor (FXII ), also known as F12 or HAE, is a serum glycoprotein primarily expressed in hepatocytes. It participates in initiating blood coagulation, fibrinolysis, and the production of bradykinin and angiotensin. FXII directly regulates cellular responses; FXII/FXIIa induces the expression of inflammatory mediators, promotes cell proliferation, and enhances the migration of neutrophils and lung fibroblasts. Its role in diseases such as hereditary angioedema and surface-triggered thrombosis is significant, as is its role in developing treatments for thrombotic inflammatory diseases.

(Data source: Maas C, et al. Blood. 2018)
F12 activation and F12-driven contact system
The FXII-driven contact system is a network of proteases and inhibitors that integrates four main pathways: the complement system, the coagulation cascade, the fibrinolytic system, and the kallikrein-kinin system. This system is called the "plasma contact system" because it is activated when FXII "contacts" an anionic surface , leading to a conformational change and the formation of active FXIIa.
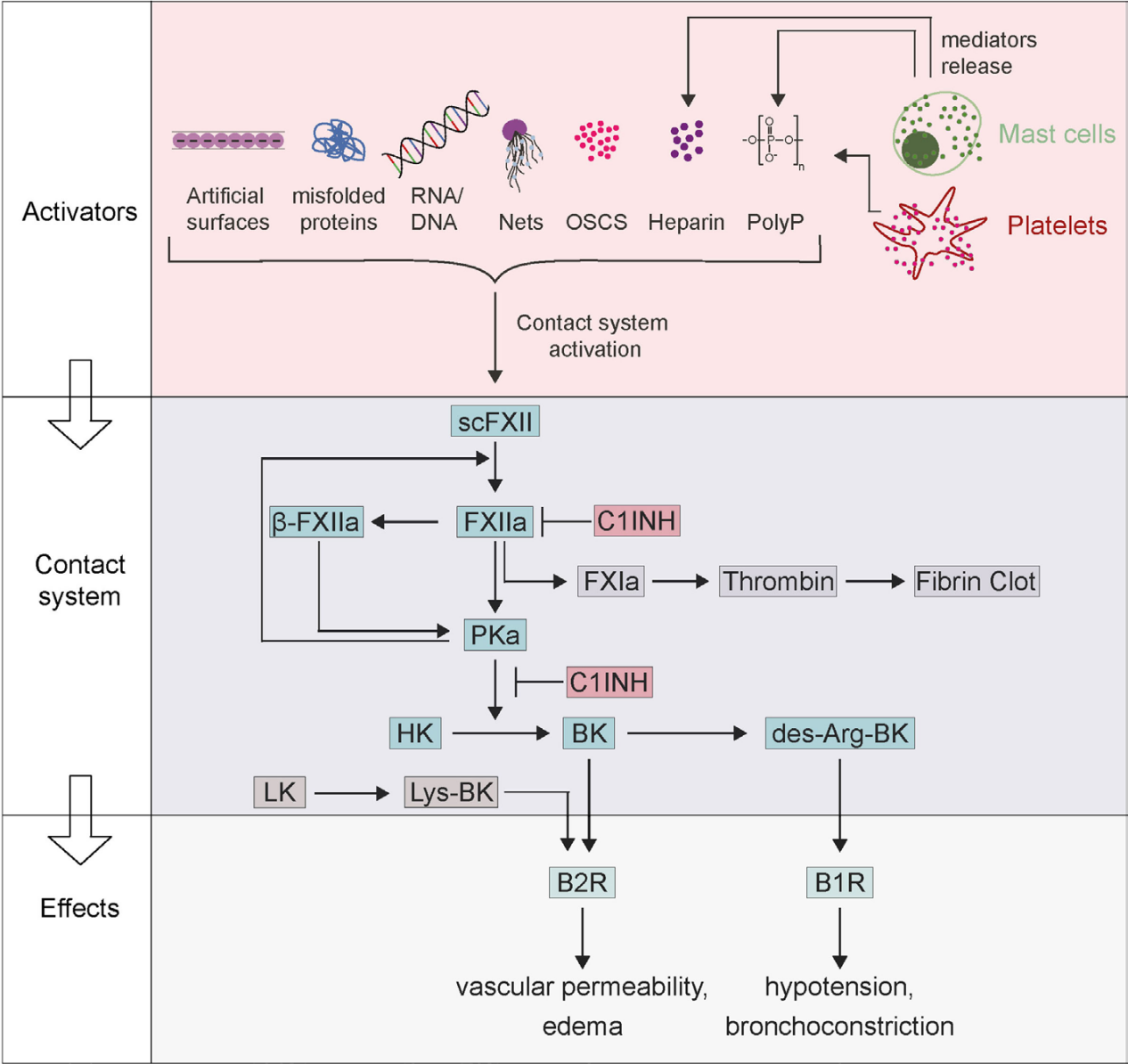
(Data source: Bender L, et al. Front Immunol. 2017)
Upon contact with anionic surfaces such as extracellular RNA, endotoxins, neutrophil extracellular traps (NETs), polyphosphates released by activated platelets, collagen exposed by damaged endothelial cells, heparin secreted by mast cells, and phosphatidylserine on apoptotic cells, FXII is self-activated to αFXIIa (hereinafter referred to as FXIIa). FXIIa cleaves plasma prokallikrein (PK) into plasma kallikrein (PKa), thereby activating additional FXII molecules (FXII heteroactivation) and amplifying the generation of FXIIa. PKa further cleaves FXIIa, leading to the formation of βFXIIa.
FXIIa activates the kallikrein-kinin system, leading to the release of bradykinin (BK) from high-molecular-weight kininogen (HK). Furthermore, it initiates the intrinsic blood coagulation pathway by activating FXI, initiates fibrinolysis via PKa-mediated urokinase activation, and activates C1 esterase (the first component of the macromolecular C1 complex in the classical complement cascade). C1INH inhibits not only FXIIa but also components of the PKa and C1 complex, thereby controlling BK production and the classical complement system. Recent findings suggest that the ability to activate PK and FXI is not limited to FXIIa. FXII, in its single-chain form, can also convert PK to PKa and FXI to FXI; the latter only occurs during platelet polyphosphate release. The ability to activate the PK and classical complement cascade is also present in βFXIIa; however, βFXIIa does not trigger the intrinsic blood coagulation pathway because it does not contain the heavy chain responsible for binding to anion surfaces. The production of two different forms of FXIIa may partially explain why FXII regulates the kallikrein-kinin system or intrinsic blood clotting pathways under certain pathological conditions.

(Data source: Cohn DM, et al. J Intern Med. 2024)
The structure of F12 and its receptor
F12 is a secreted protein primarily produced in the liver and present in plasma as a single-chain zymogen. Upon contact with a negatively charged surface, FXII is converted to its active form, FXIIa, which has a double-chain structure. The FXIIa heavy chain consists of six domains: a fibronectin type II domain, an epidermal growth factor (EGF)-like domain, a fibronectin type I domain, a second EGF-like domain, a kringle domain, and a proline-rich region. The FXIIa light chain contains a His393-Asp442-Ser544 catalytic triplet. The heavy and light chains of FXIIa are interconnected by disulfide bonds. The FXIIa heavy chain contains putative cell and surface binding sites, as well as Zn2+, FXI, heparin, and fibrin binding regions.
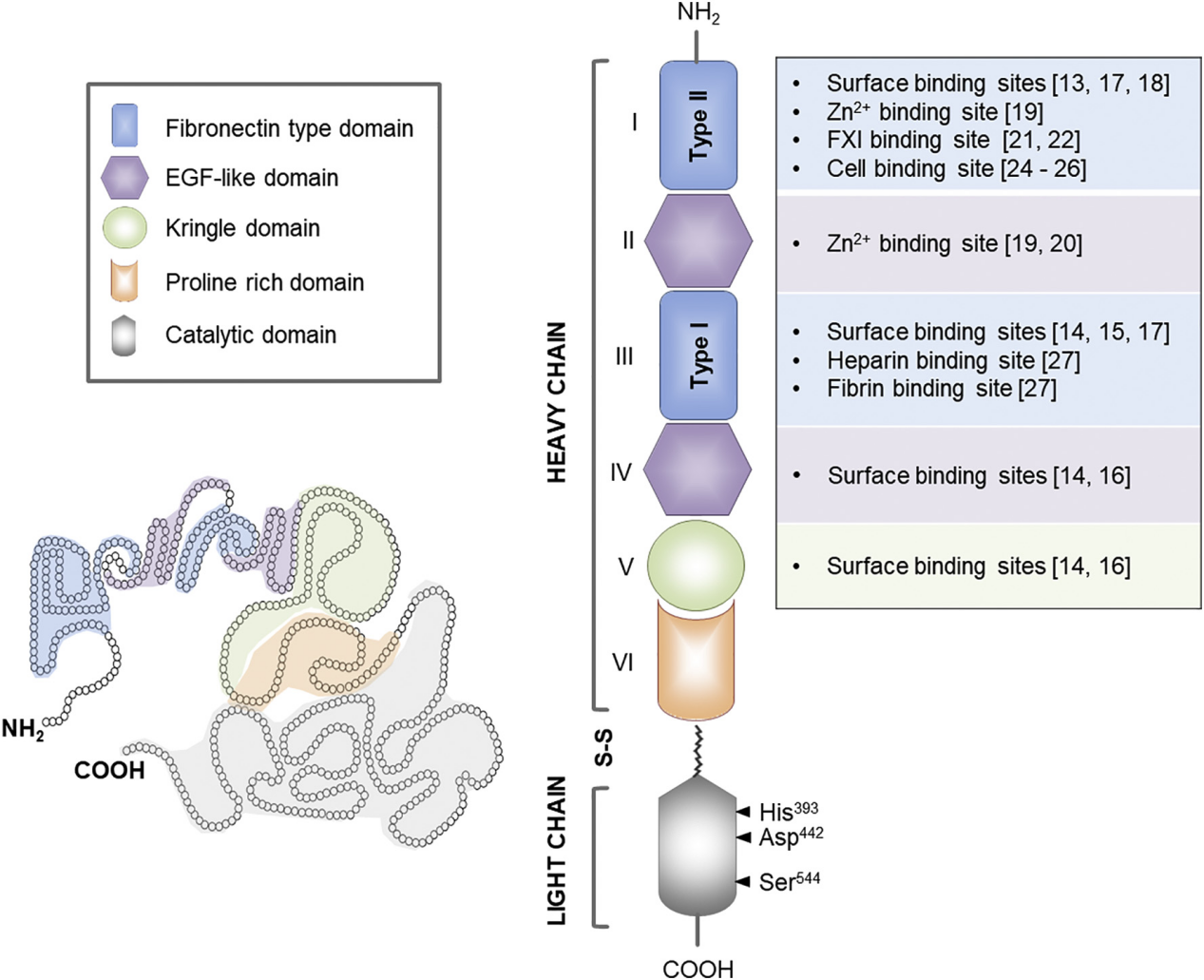
(Data source: Maas C, et al. Blood. 2018)
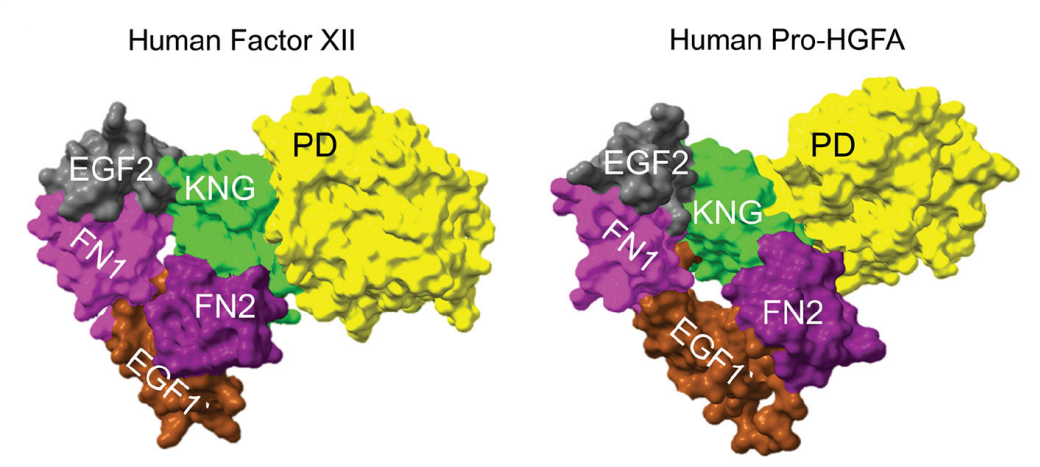
(Data source: Shamanaev A, et al. Semin Thromb Hemost. 2024)
Regulation of F12 in inflammatory cells
FXII can interact with neutrophils, blood monocytes, bone marrow-derived macrophages, and dendritic cells. FXII can induce blood monocytes/macrophages to transform into the M2 phenotype by secreting substances such as IL-4, IL-8, IL-10, and transforming growth factor (TGF)-β, and has been found to promote the expression of substances such as IL-8, IL-1β, IL-6, leukemia inhibitory factor, CXCL5, and TNF-α in human lungs.
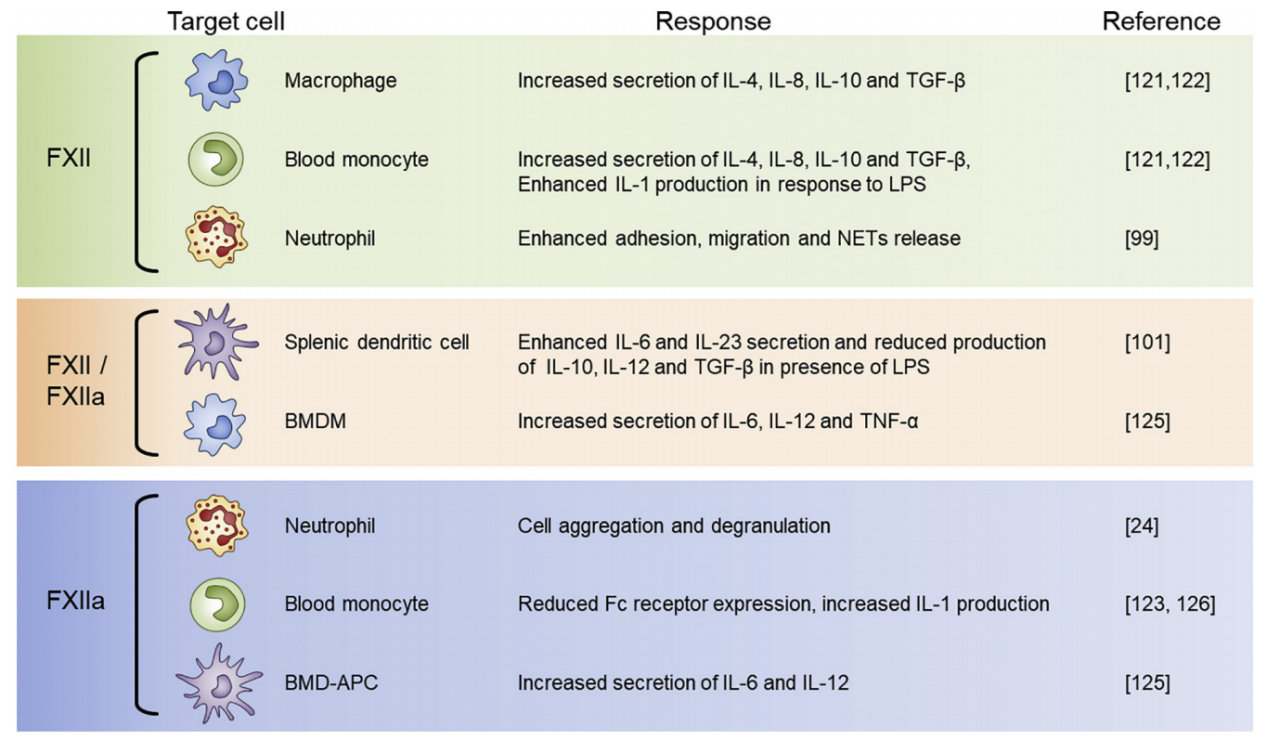
(Data source: Maas C, et al. Blood. 2018)
F12 targeted therapy
REGN9533 is a monoclonal antibody targeting F12 developed by Regeneron Pharmaceuticals for the treatment of blood disorders and is currently in Phase I clinical trials. NCT06665828 is a Phase 1, randomized, double-blind, placebo-controlled study evaluating the safety, tolerability, pharmacokinetics, and pharmacodynamics of a single escalation dose of REGN9533 in healthy adults.
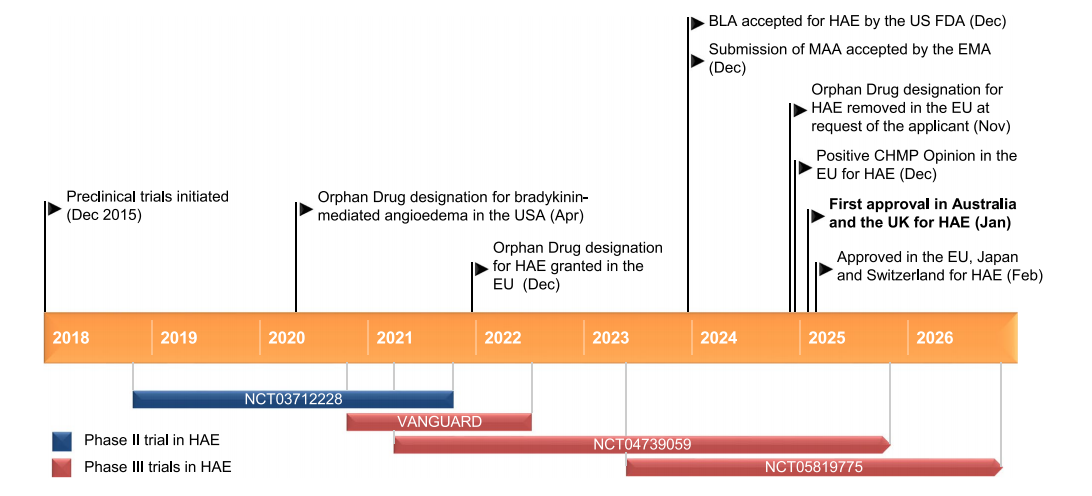
Garadacimab is a fully human IgG4/λ recombinant monoclonal antibody against activated coagulation factor XII, developed by CSL Boehringer Ingelheim, for the prevention of hereditary angioedema attacks. Garadacimab binds to and inhibits the catalytic domain of FXIIa (and βFXIIa). Inhibition of FXIIa can prevent hereditary angioedema attacks by blocking the kallikrein -acid thrombin cascade, which involves activating prokallikrein to kallikrein and producing bradykinin, which causes swelling and inflammation during hereditary angioedema attacks.
In January 2025, Garadacimab received its first approval in Australia and the UK for the prevention of hereditary angioedema in adults and adolescents (≥12 years of age). Furthermore, in February 2025, Garadacimab received the same indication approval in the EU, Japan, and Switzerland. Regulatory review of Garadacimab is currently underway in the US and Canada.
(Data source: Fung S. Drugs. 2025)
