The DPEP family is an important member of the dipeptidase family, primarily participating in key physiological processes through the hydrolysis of dipeptides. In healthy individuals, DPEP3 expression is almost exclusively confined to the testes, but its expression is abnormally upregulated in various cancers. Some studies have found DPEP3 lacking dipeptidase activity, and its exact biological function remains under investigation. Current research suggests that DPEP3 may play a regulatory role in reproduction, tumors, and inflammation; therefore, DPEP3 may be a potential target for therapeutic research strategies in various diseases.
DPEP dipeptidase family
Dipeptidase-3 (DPEP3) belongs to the membrane-bound dipeptidase family, which has three members: Dipeptidase 1 (DPEP1), Dipeptidase 2 (DPEP2), and Dipeptidase 3 (DPEP3). They primarily participate in key physiological processes by hydrolyzing dipeptides, including glutathione (GSH), leukotrienes, and lipid metabolism; and possess β-lactamase and metalloenzyme activities. Furthermore, the three members of the DPEP family exhibit significant differences in substrate specificity and tissue distribution. For example, DPEP2 shows the highest expression levels in the lungs, heart, and testes, while DPEP3 is only found in the testes.
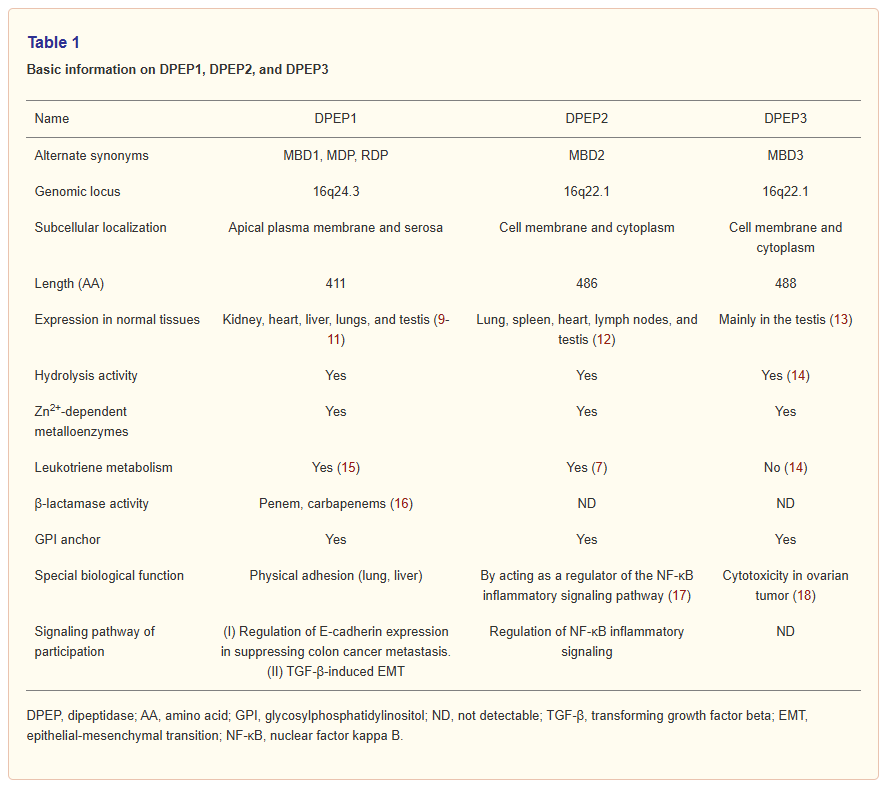
(Data source: Wang L, et al. Transl Cancer Res. 2024)
Biological structure of DPEP3
The DPEP 3 gene is located on chromosome 16 (16q22.1) and encodes 488 amino acids (aa). The initial hydrophobic region consisting of the first 35 amino acid residues is called the signal peptide (SSP). The region from 36 aa to 463 aa is the main chain of the gene, containing an N-glycosylation site linked to asparagine and three disulfide bond sites. The disulfide bond at amino acid 434 can link two DPEP3 monomers to form a dimer structure. In addition, the GPI site at the C-terminus of DPEP 3 ensures its anchoring to the outer cell membrane. Notably, after GPI anchoring, the C-terminus of DPEP 3 contains an extended hydrophobic amino acid sequence as a propeptide (464 aa- 488 aa), which is usually cleaved during DPEP 3 molecule maturation.
Based on X-ray structure, DPEP 3 typically exists in cells as a homodimer. Similar in structure to its family relative DPEP1, the DPEP3 monomer consists of a trisulfide phosphate isomerase (TIM) (β/α)8 barrel fold, with the active site located at the center of the barrel. The secondary structure is stabilized by two intramolecular disulfide bonds: one linking β2 (Cys 146) and β4 (Cys 225) of the β chain, and the other linking β6 (Cys 297) and β7 (Cys 329). Overall, the truncated DPEP3 (78–437) shares 49% sequence identity with DPEP1 (17–385), indicating a high degree of structural conservation between the two enzymes.
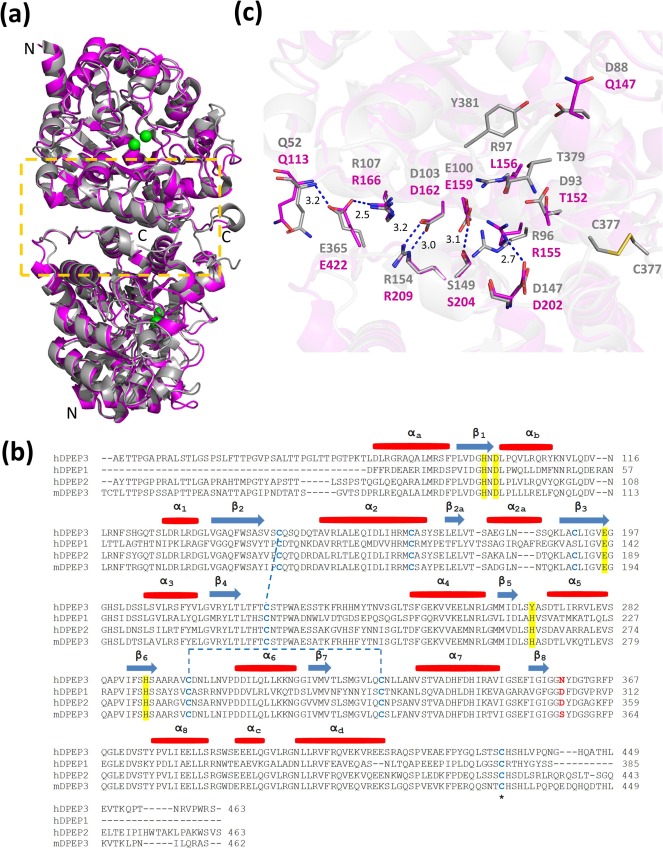
(Data source: Hayashi K, et al. J Struct Biol. 2020)
DPEP3 expression in cancer
DPEP3 protein expression is highly restricted in normal tissues, with its mRNA expressed only in testicular tissue. However, it exhibits aberrant expression in certain types of cancer. Current research has revealed that DPEP3 is a cancer-testis antigen selectively expressed in cancers such as ovarian cancer, non-small cell lung cancer, and melanoma, with particularly high expression in tumor-initiating cells of ovarian cancer.
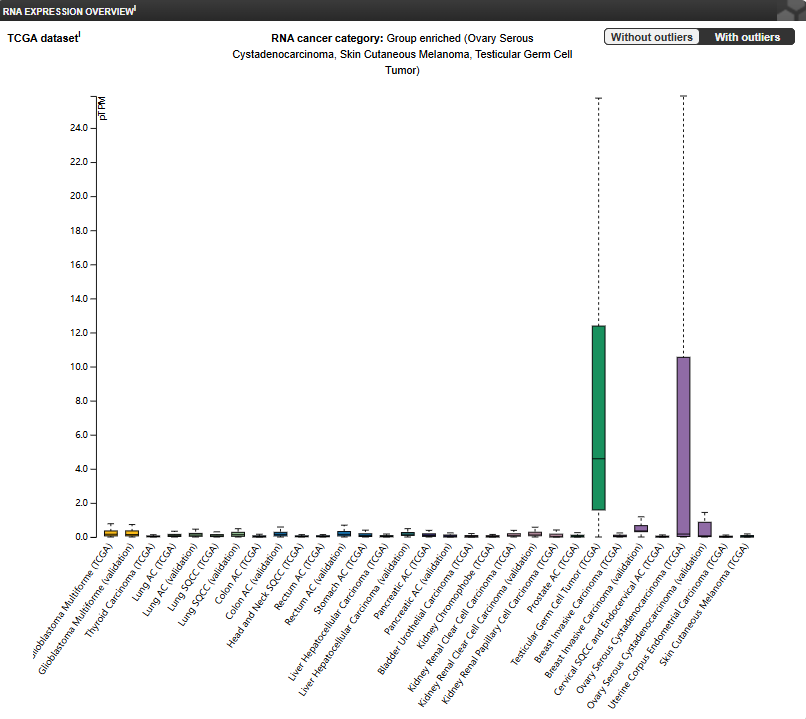
(Data source: The Human Protein Atlas)
Research on DPEP3 in Disease
The DPEP family is an important member of the dipeptidase family, which is specifically used to hydrolyze dipeptides. However, some studies have found that DPEP3 lacks dipeptidase activity and cannot hydrolyze cysteyldiglycine. The exact biological function of DPEP3 is still under investigation. Researchers studying changes in semen metabolism after microsurgical varicocele surgery have found that DPEP3 levels significantly decreased post-surgery, consistent with improved semen quality. This provides new clues for predicting early diagnosis and prognosis in varicocele microsurgery. Furthermore, researchers investigated the expression of a group of carcinoembryonic antigen (CTA) proteins in non-small cell lung cancer (NSCLC). They validated eight CTA proteins (DPEP3, EZHIP, MAGEA4, MAGEB2, MAGEC2, PAGE1, PRAME, and TKTL1) through RNA sequencing and performed immunohistochemistry on NSCLC patient tissues. The results showed that one or more CTAs were expressed in most NSCLC patients, suggesting that the presence of DPEP3 may affect the immune system's response in tumor tissues. Additionally, reports indicate that DPEP3 expression is lower in colon cells of patients with rheumatoid arthritis, suggesting a potential role for DPEP3 in rheumatoid arthritis research.
Clinical drug development for DPEP3
Clinical research on DPEP3 is still in its early stages, but existing literature explores its potential for clinical application. SC-003 is a PBD-conjugated anti-DPEP3 monoclonal antibody. SC-003 has shown strong preclinical efficacy in a DPEP3-positive ovarian cancer PDX (patient-derived xenograft) model. Currently, the study has reported results from a first-in-human phase 1 trial, showing that all patients eventually discontinued treatment, with (n= 47; 64%) discontinuing due to disease progression. Other reasons for discontinuation included adverse events (n=14; 19%), patient withdrawal (n=7; 10%), and investigator decisions (n=6; 8%); 50 patients (68%) died during the study. Upon further evaluation, all patients experienced≥1 adverse event, with 64 (87%) experiencing≥1 drug-related adverse event. Forty-nine (66%) patients experienced ≥1 serious adverse event (SAE); 15 (20%) patients experienced≥1 drug-related SAE. Overall, the most common treatment-related adverse events were fatigue (51%), nausea (51%), decreased appetite (36%), pleural effusion (35%), abdominal pain (34%), and peripheral edema (34%). In the diagnostically selected stage 1b portion (n=42 evaluable patients), increased DPEP3 expression detected by immunohistochemistry (IHC) was associated with enhanced antitumor activity.
Although SC-003 showed efficacy in preclinical studies, it resulted in low response rates and short duration of action in patients with epithelial ovarian cancer (EOC), and was associated with significant toxicities at doses that produced some antitumor activity. This clearly demonstrates the immense challenge of translating a promising target into a successful drug. While the clinical exploration of SC-003 ended in failure, it provided valuable experience and data that point the way for the design of next-generation, more precise DPEP3-targeted therapies.
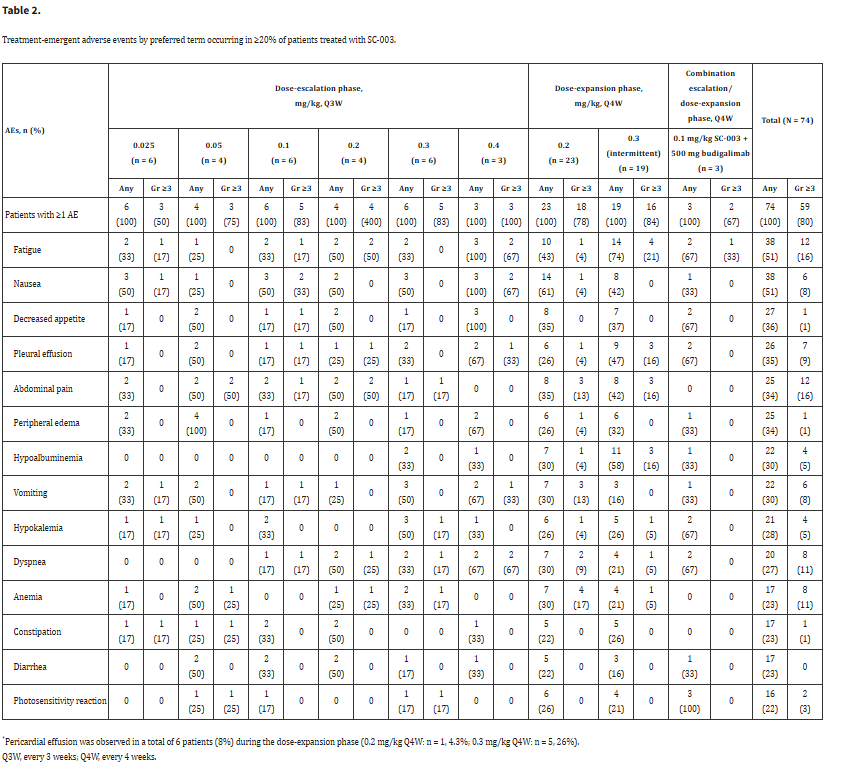
(Data source: Hamilton E, et al. Gynecol Oncol. 2020)
