CD8 is a transmembrane glycoprotein that acts as a coreceptor for the T cell receptor (TCR). Like the TCR, CD8 binds to major histocompatibility complex (MHC) molecules but has specificity for MHC class I proteins. Together with the TCR, the CD8 coreceptor plays a role in T cell signaling and assists in cytotoxic T cell (CTL)-antigen interactions.
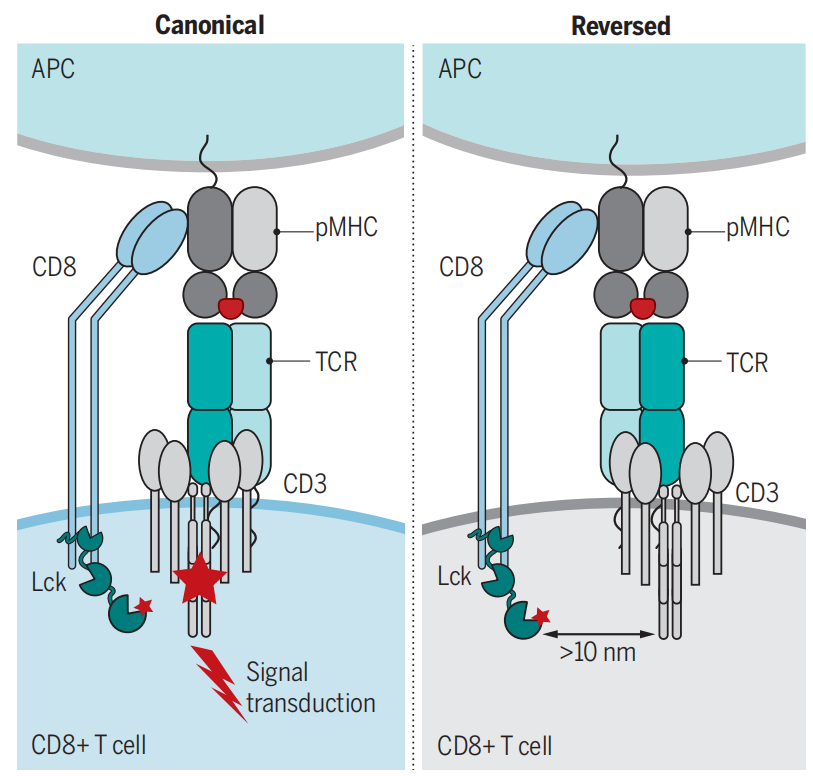
(Data source: Zareie P, et al. Science. 2021)
Definition and structure of CD8:
There are two isoforms of CD8: α and β, each encoded by a different gene. In humans, both genes are located on chromosome 2 at position 2p12. The most common form of CD8 consists of CD8-α and CD8-β chains, both members of the immunoglobulin superfamily, with an immunoglobulin variable region (IgV)-like extracellular domain and an intracellular tail attached to the cell membrane. Less common CD8-α chain homodimers are also expressed on some cells. Each CD8 chain has a molecular weight of approximately 34 kDa.
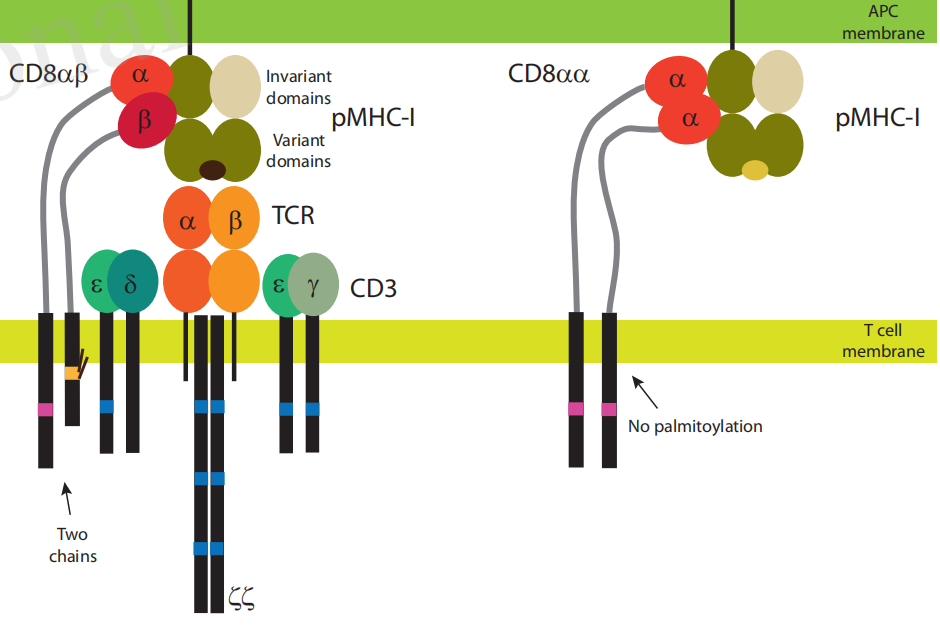
(Data source: Glatzová D,et al. Front Immunol. 2019)
Tissue Distribution:
The CD8 coreceptor is primarily expressed on the surface of cytotoxic T cells, but can also be found on natural killer cells, cortical thymocytes, and dendritic cells. The CD8 molecule is a marker for the cytotoxic T cell population.
Regulation of immune response:
The extracellular IgV-like domain of CD8-α interacts with the α3 portion of class I MHC molecules. This affinity brings the T cell receptor of cytotoxic T cells and target cells into close contact during antigen-specific activation. In addition to facilitating cytotoxic T cell-antigen interactions, the cytoplasmic tail of the CD8 coreceptor interacts with Lck (lymphocyte-specific protein tyrosine kinase) , leading to the activation of transcription factors such as NFAT, NF-κB, and AP-1.
Once CD8+ T cells are activated, they undergo a series of replication and differentiation steps to prepare for a targeted immune response. Contact of CTLs with target infected or malignant cells induces apoptosis:
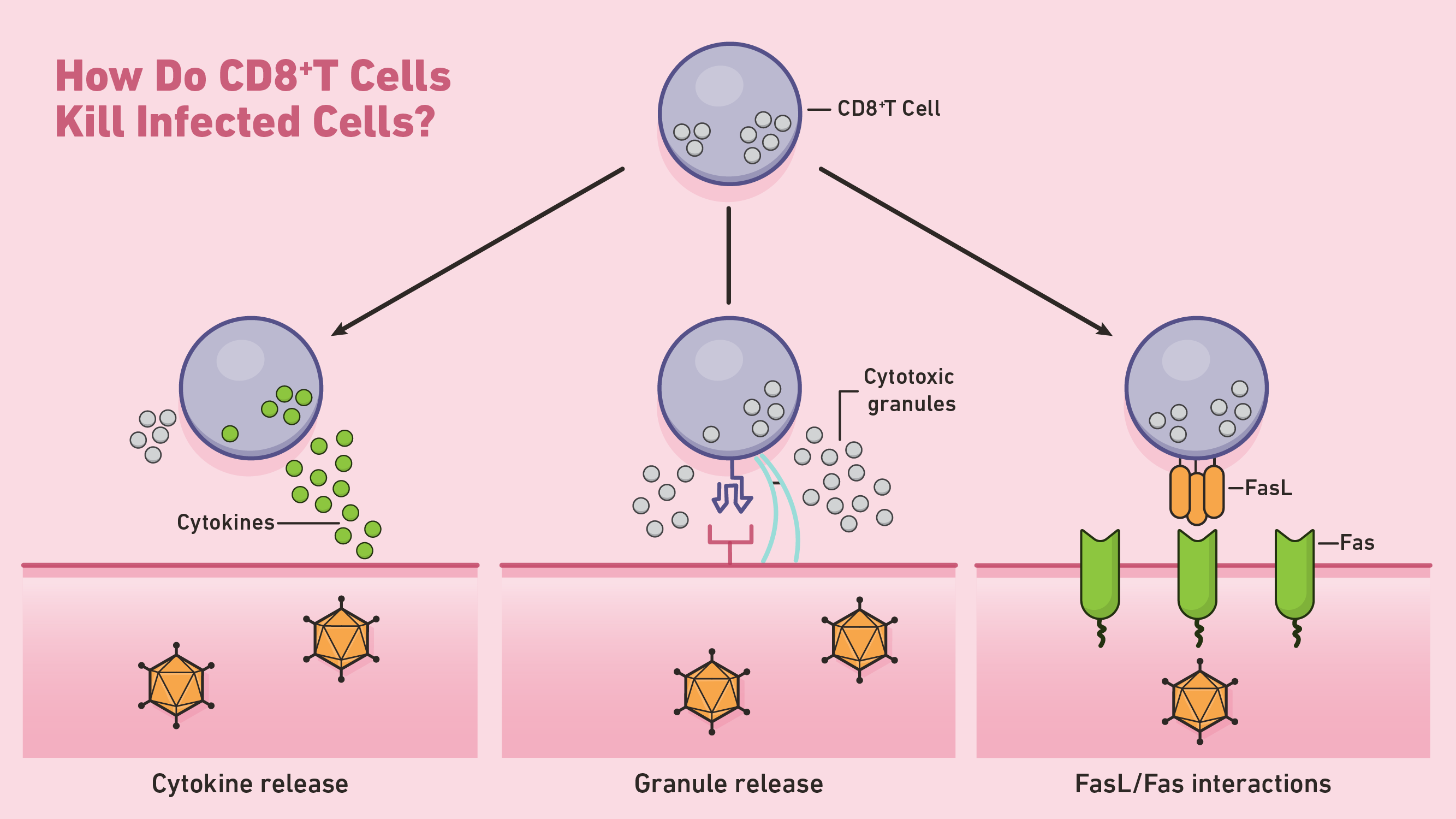
(Data source: technology networks)
I. One way CD8+ T cells initiate apoptosis is through the secretion of perforin and granzymes, two cytotoxic proteins. Perforin is a cytolytic protein that forms pores in the target cell's membrane. CTLs exploit these pores to direct the release of granzymes, a class of serine proteases that continue the apoptotic process within the cytoplasm. These proteases shut down the target cell by cleaving viral and cellular proteins involved in normal cell maintenance. The apoptotic target cell is then cleared by nearby phagocytes.
II. CTLs can also induce apoptosis through the Fas-Fas ligand interaction. Whenever Fas ligand (FasL) binds to the Fas receptor on a target cell, a series of signaling molecules initiate apoptosis by activating caspase proteases. These caspases perform most of the proteolysis in the later stages of apoptosis.
III. CD8+ T cells can also indirectly kill target cells by releasing cytokines such as TNF-α.
Cytotoxic CD8+ T cells play a key role in eliminating intracellular infections and malignant cells and provide long-term protective immunity. A better understanding of CD8+ T cells could enhance our ability to design effective vaccines or manipulate cell-mediated immune responses in real time.
