The most common genetic alteration in human prostate cancer (PC) is a gene fusion involving the androgen-regulated TMPRSS2 gene and the coding sequence of a member of the ETS family of transcription factors, ERG, which occurs in approximately half of all human prostate cancers. The TMPRSS2-ERG fusion is not only a prostate cancer-initiating event but is also required for the survival of ERG-expressing prostate cancer cells.

(Data source: Kumar-Sinha C,et al. Nat Rev Cancer. 2008)
November 2023,Nature Communications s published a research paper titled "SND1 binds to ERG and promotes tumor growth in genetic mouse models of prostate cancer". The research protein ERG interacts with the SND1/MTDH complex through the Tudor domain of SND1, promotes the nuclear localization of SND1/MTDH , and significantly enhances the growth-promoting function of prostate cancer cells.

(Article publication information)
ERG interacts with the SND1/MTDH protein complex:
Epitope-tagged ERG was subjected to affinity purification-mass spectrometry (AP/MS) studies, and high-precision mass spectrometry analysis identified 216 putative ERG-interacting proteins, which were confirmed by co-expression and co-immunoprecipitation experiments in HEK293 cells to reveal the protein interaction between ERG and SND1/MTDH.

The Tudor domain of SND1 interacts with the N-terminal domain of ERG:
Co-immunoprecipitation experiments were performed using a panel of recombinant ERG and SND1 fragments expressed in HEK293T cells. The N-terminal region of ERG showed strong binding to SND1. Expression of individual ERG domains revealed that the N-terminal domain of ERG and, to a lesser extent, the ETS domain of the G-cell terminal end of ERG interacted with SND1.

SND1 is required for ERG-mediated promotion of cell proliferation in human prostate epithelial cells:
Immortalized non-tumorigenic human prostate epithelial RWPE-1 cells were knocked down for endogenous SND1 or MTDH using two independent shRNA constructs for each gene. Knockdown of SND1 or MTDH did not affect colony size in control RWPE-1-GFP cells, but they significantly reduced colony size in RWPE-1-ERG cells. Expression of exogenous ERG increased the size of 3D colonies grown in organoid culture conditions in the presence of Matrigel.

ERG promotes SND1 nuclear localization and stimulates prostate epithelial cell proliferation:
ERG protein was mainly localized in the nucleus, but most SND1 was present in the cytoplasm of prostate epithelial cells. Cytoplasmic/nuclear fractionation of RWPE-1-ERG and control RWPE-1-GFP cells showed that SND1/MTDH levels were significantly increased in the nuclei of ERG-overexpressing cells.
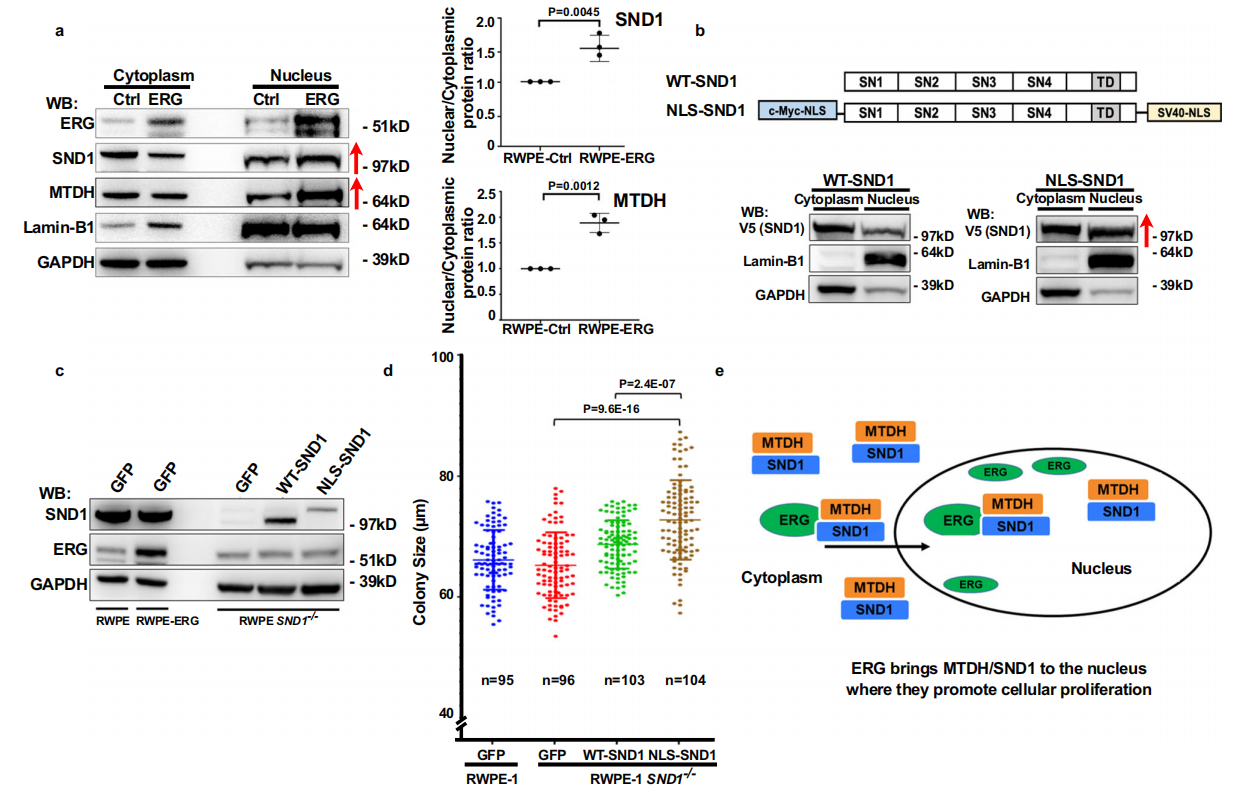
Ablation of endogenous SND1 negatively impacts PC growth in vivo:
They used PB-Cre4/ Pten fl/fl/ERG mice, which develop high-grade prostatic intraepithelial neoplasia and prostate adenocarcinoma at a young age (8-12 months after birth). Ablation of SND1 had a significant negative impact on prostate tumor development and growth: prostate weight and prostate cancer incidence were significantly reduced in PB-Cre4/ten fl/fl/ERG/Snd1 fl/fl mice.

Summary:
This study discovered that ERG interacts with the MTDH/SND1 protein complex and that overexpression of endogenous ERG in prostate epithelial cells increases the nuclear localization of MTDH/SND1 and promotes prostate cell growth in vitro and in vivo. Loss-and gain-of-function experiments revealed a previously unknown ERG-mediated transformation mechanism. These findings also highlight the important role of SND1 in prostate cancer.
