Background
Signaling pathways involving the receptor tyrosine kinase FLT3 and FLT3 ligand regulate the development and activity of dendritic cells (DCs) and other myeloid cells, including monocytes. Increased FLT3/FLT3L signaling and elevated circulating FLT3L levels are associated with multiple autoimmune and inflammatory diseases, including rheumatoid arthritis, Sjögren's syndrome, and systemic lupus erythematosus.
On July 7, 2025, Annie Lau-Kilby's team at Amgen published an article titled "Development and preclinical characterization of AMG 329: a human antibody neutralizing FLT3 ligand" in MAbs. The article describes the derivation and in vitro characterization of AMG 329, an IgG1 monoclonal antibody against human FLT3, and the pharmacodynamic effects of AMG 329 on circulating DCs in cynomolgus monkeys.

Expression distribution and function of FLT3
FLT3 is constitutively expressed on multipotent progenitor cells in the bone marrow but is maintained on pDCs, DC1s, and a subset of DC2s; these cells depend on FLT3L for development and survival.
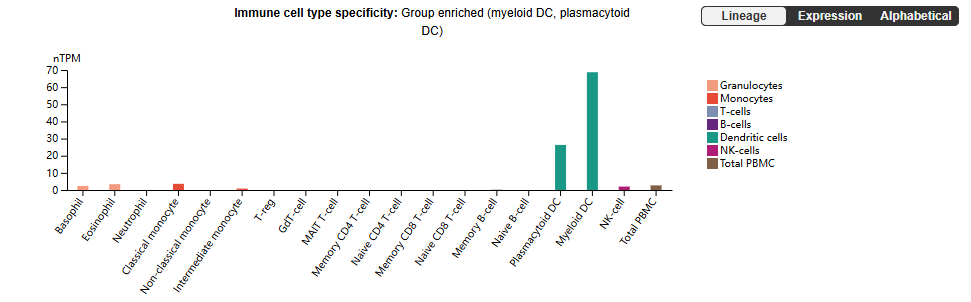
(Data source: Uniprot)
DCs play an important role in activating innate and adaptive immunity, initiating immune responses by interacting with T cells and regulating the balance between immune activation and tolerance. DCs have been considered as potential therapeutic targets for a variety of pathological conditions, including autoimmune diseases. pDCs have attracted particular attention as potential therapeutic targets due to their involvement in a variety of inflammatory and autoimmune diseases and their ability to produce large amounts of type I interferons and proinflammatory cytokines/chemokines.

FLT3 is activated and expressed on monocytes, B cells, and T cells, while FLT3L appears to promote the proliferation and survival of these cells. In patients with autoimmune or inflammatory diseases, inhibiting the FLT3/FLT3L signaling pathway may reduce the pro-inflammatory effects of pDCs, cDCs, and monocytes, thereby slowing disease progression.

(Data source: Swiecki M, et al. Nat Rev Immunol. 2015)
Preparation optimization and in vitro characterization of AMG 329
The parental clone 5D9 of AMG 329 was derived from a human naïve phage display library; Three rounds of solution-phase screening enriched for antibodies that bind to human and cynomolgus macaque (but not mouse) FLT3L and were converted to ScFv-Fc or single-chain Fab. Subsequently, high-throughput HTRF screening was performed against human FLT3/FLT3L for blocking activity. Positive clones were reformatted into human IgG (containing the Fc-silencing triple mutations L234F/L235E/P331S). Two rounds of affinity maturation yielded the high-affinity variant AM40, which, after combining it with an IgG-TM backbone, was named AMG 329 (formerly known as MED1116/VIB-1116/HZN-1116). BLI confirmed its direct blocking of the FLT3L-FLT3 interaction.

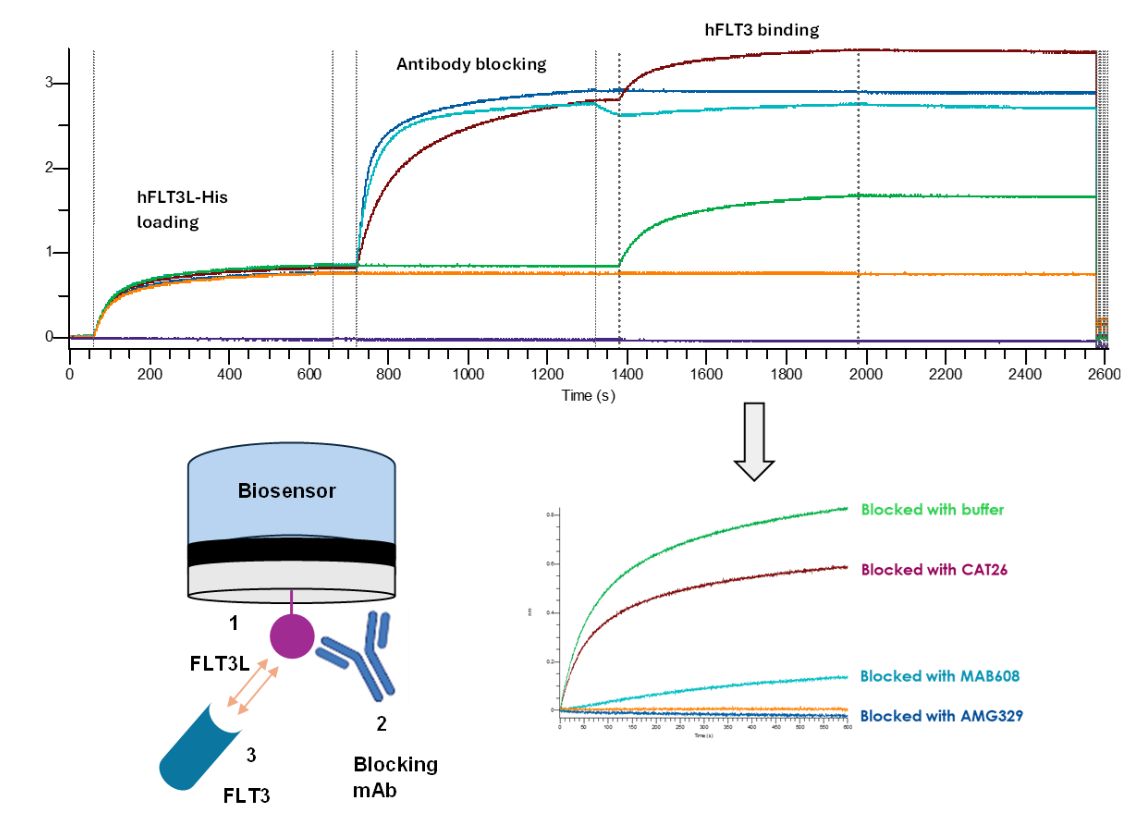
Species cross-reactivity
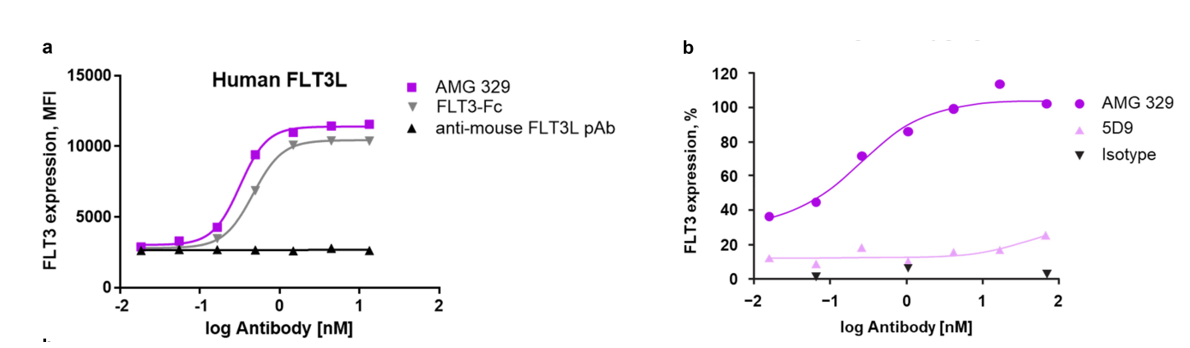
The extracellular domain of FLT3L shares 95% sequence identity between humans and cynomolgus monkeys, and 73% between humans and mice . AMG 329's parent antibody, 5D9, binds to human and cynomolgus monkey FLT3L, but not mouse FLT3L.

Neutralization of FTL3L
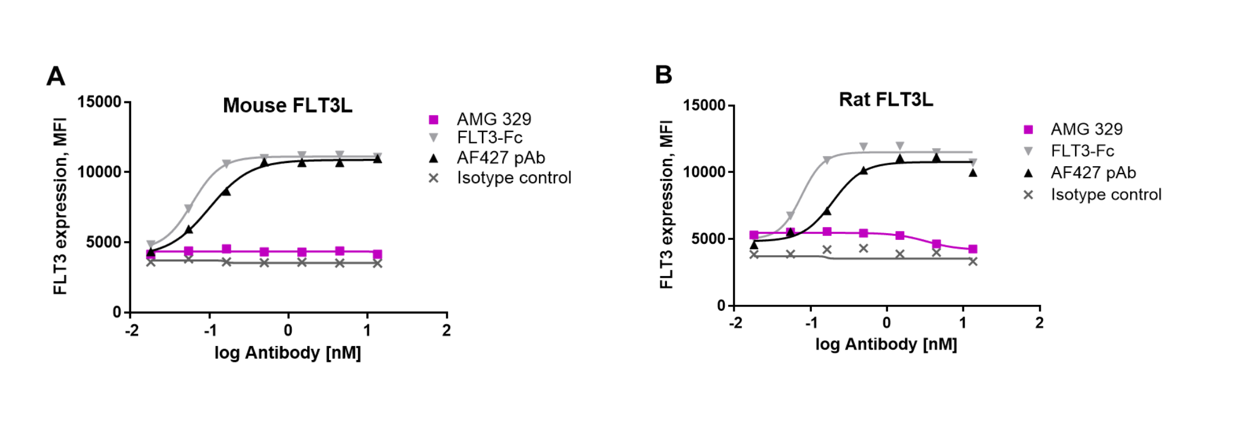
AMG 329 specifically neutralized both soluble and cell-bound human FLT3L , whereas AMG 329 did not neutralize either mouse or rat FLT3L, suggesting that FLT3L neutralization is species-specific.
AMG 329 specifically binds to FLT3L without binding to or neutralizing other class III tyrosine kinase receptor ligands (SCF, macrophage colony-stimulating factor (M-CSF), or platelet-derived growth factor (PDGF)).
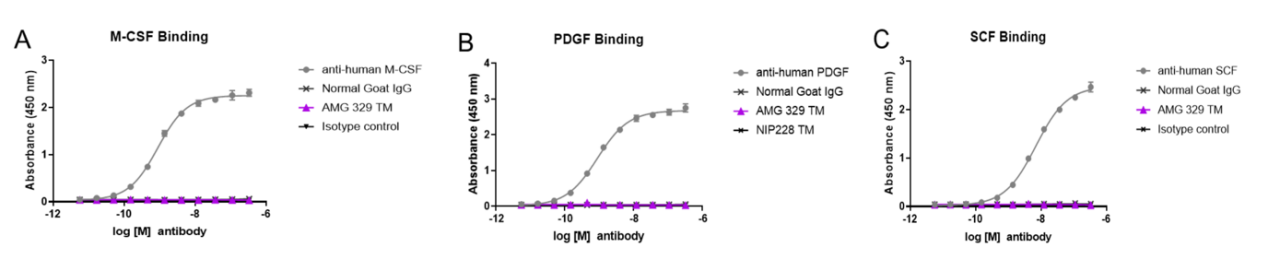
FcR binding, antibody-dependent cellular cytotoxicity (ADCC), and C1q binding
In the presence of allogeneic peripheral blood mononuclear cells, AMG 329 did not induce cell death in CD4+ T cells treated with IL-7 to upregulate FLT3L expression , indicating that AMG 329 lacks antibody-dependent cellular cytotoxicity. Binding to the Fc neonatal receptor (FcRn) was unaffected by the triple mutations in the AMG 329 Fc. ELISA analysis revealed that AMG 329 did not bind to human C1q in vitro.
AMG 329 does not induce ADCC and is unlikely to induce complement-dependent cytotoxicity, suggesting a low likelihood of Fc-related off-target effects.


Pharmacokinetic and pharmacodynamic analysis
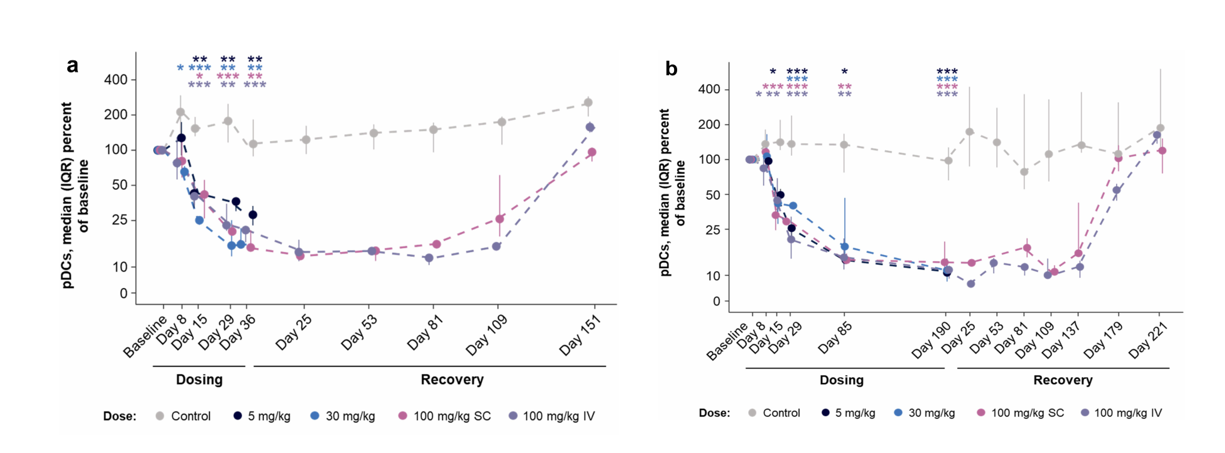
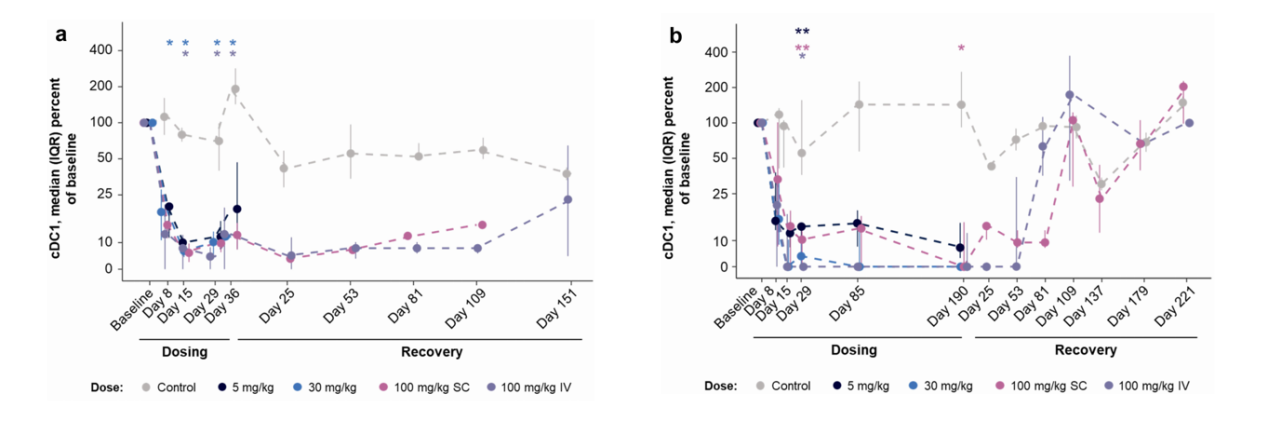
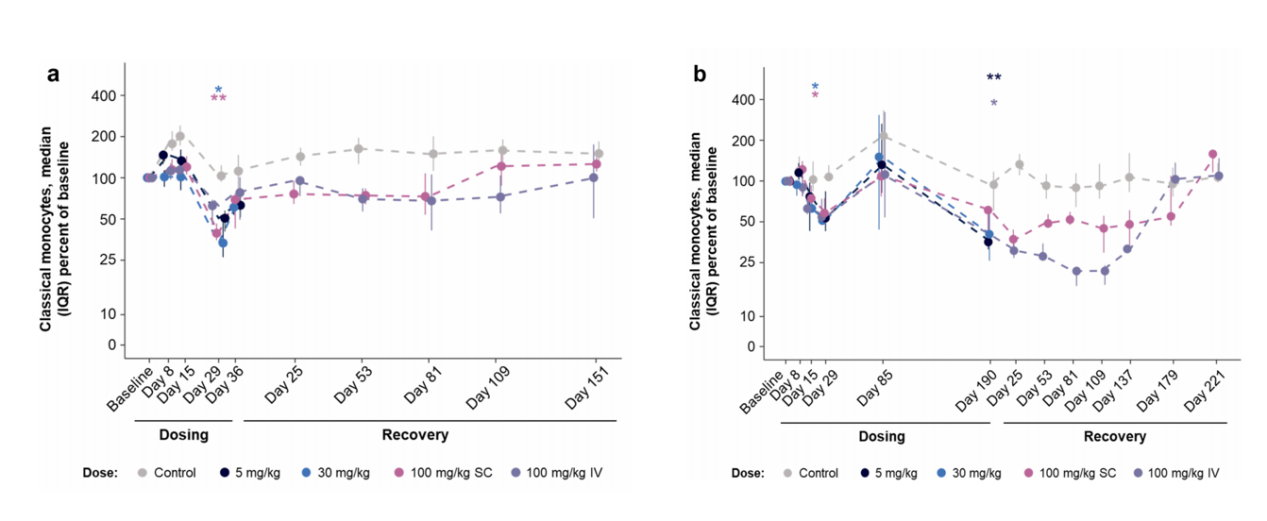
A toxicity study involving five doses over five weeks and 14 doses over 27 weeks revealed that AMG 329 administration reduced the relative proportions of pDCs, cDC1s, cDC2s, and classical monocytes in a dose-independent manner. The relative proportions of pDCs, cDC1s, and cDC2s were maintained during the recovery phase and generally returned to baseline levels by the end of the recovery period. These results suggest that the AMG 329-associated reductions in peripheral blood pDCs, cDC1s, cDC2s, and classical monocytes in cynomolgus monkeys are consistent with the expected pharmacology of AMG 329, with durable responses observed in many of these subsets during the recovery phase; recovery to control values 224 days after dosing suggests reversibility.
T cell-dependent antibody response (TDAR) to KLH
In rhesus macaques treated with KLH, no differences in anti-KLH IgM or IgG titers were observed in response to AMG 329; the magnitude and duration of the KLH response were similar across dose groups compared with the vehicle control group.
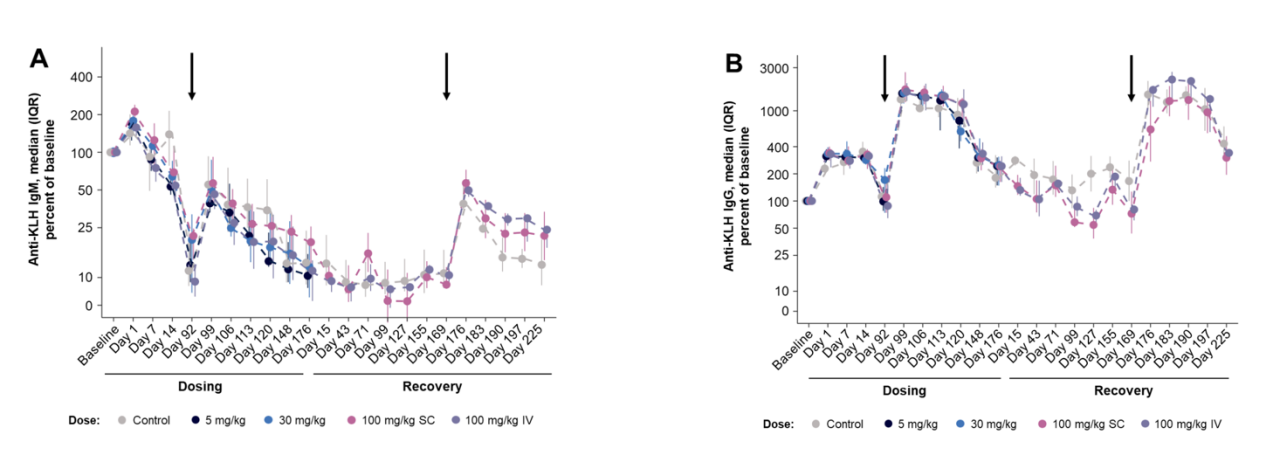
Despite a substantial reduction in pDCs, cDCs, and classical monocytes, the TDAR response to KLH in cynomolgus monkeys suggests intact immune function. Collectively, these findings support advancing AMG 329 into clinical development for the treatment of inflammatory or autoimmune diseases.
Summarize
FLT3 is primarily expressed in dendritic cells (DCs). Inhibiting FLT3/FLT3L signaling through pharmacological or genetic means can reduce DC (dendritic cell) numbers. In vitro characterization, pharmacokinetics, and pharmacokinetic analysis of AMG 329 revealed that the antibody does not induce ADCC, and the likelihood of Fc-related off-target effects is low. Following administration, the proportions of pDCs, cDC1s, cDC2s, and classical monocytes decreased. AMG 329 is currently being studied in a Phase 2 clinical trial as a potential treatment candidate for various autoimmune or inflammatory diseases (NCT06312020).
