LY6/PLAUR Domain Containing 3 (LYPD3/C4.4A) is a member of the LY6/uPAR protein family. LYPD3 is expressed in the placenta, skin, and urothelium. PLAUR encodes the urokinase-type plasminogen activator receptor (uPAR), a glycolipid-anchored cell surface protein that plays an important role in various physiological and pathological processes. Studies have found that LYPD3 is present in the majority of primary and metastatic transitional cell carcinomas (TCCs) and breast cancer tissues, but is absent in adjacent normal tissues.
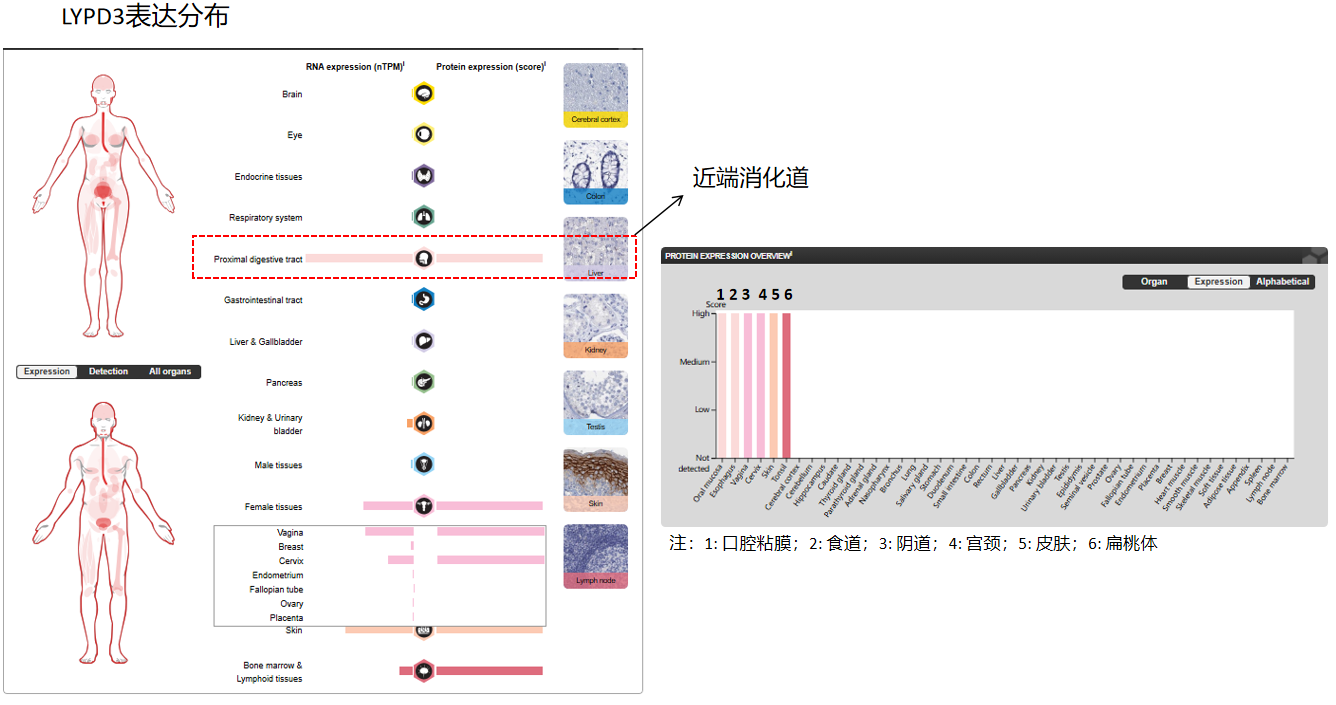
LYPD3 expression distribution
LYPD3 is primarily expressed in the placenta, skin, and urothelium. It is found in suprabasal keratinocytes of chronic wounds. Weak expression is observed in the esophagus and peripheral blood mononuclear cells. It is found in most primary and metastatic transitional cell carcinomas (TCCs) and breast cancer tissues but not in adjacent normal tissues. High expression is observed in the tumor portion of some noninvasive superficial lesions and in invasive and metastatic urothelial carcinomas.
(Data source: Uniprot)
Function of LYPD3
Supports cell migration. May be involved in urothelial cell-stroma interactions. May be involved in tumor progression.
Promote cell migration and adhesion: Participate in wound healing in normal tissues (such as skin and lung epithelium) and mediate cell spreading and migration by binding to matrix components such as laminin.
Driving tumor malignant progression: by activating PI3K/AKT and EGFR signaling pathways.
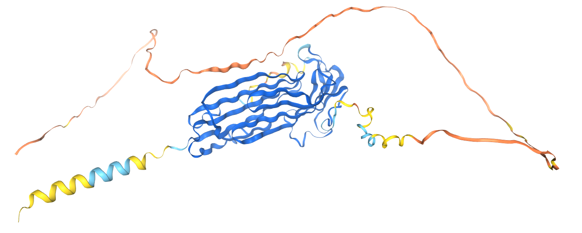
Structure of LYPD3
LYPD3 , encoded by the LYPD3 gene, is a 346-aa GPT-type membrane protein with a molecular weight of approximately 35.9 kDa . It belongs to the LY6/uPAR family. It comprises three functional domains (DI, DII, and DIII) connected in series by a flexible linker. From the N-terminus to the C-terminus, the domains function as the core ligand-binding region, containing uPAR-binding epitopes (e.g., Arg58 and Ile63), whose mutations can block uPA binding; the domains stabilize the DI conformation and participate in interactions with integrins (e.g., αvβ3); the domains connect to the GPI anchor, mediating cell membrane localization; and the domains contain protease cleavage sites (e.g., Arg279-Ala280) that are cleaved by matrix metalloproteinases (MMPs) to release soluble uPAR.
(Data source : AlphaFold)
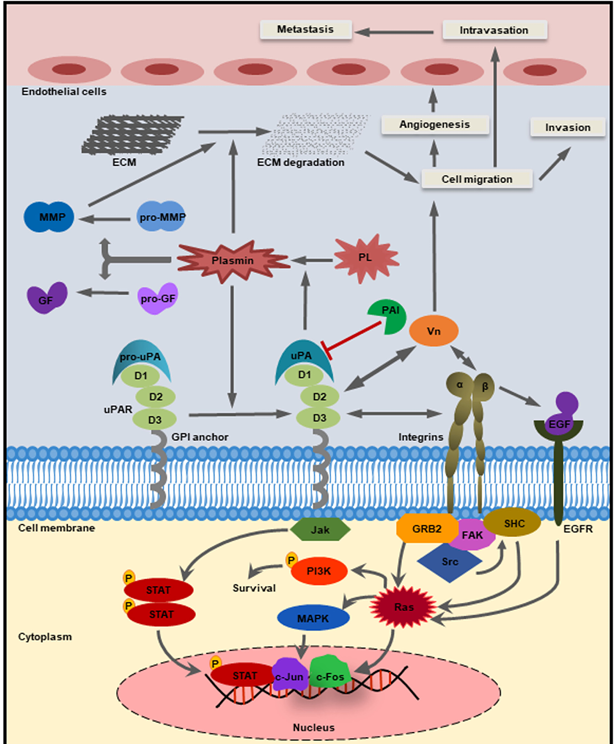
LYPD3 signaling pathway
Classic uPA/uPAR-mediated fibrinolysis and signal activation pathway: After uPA binds to uPAR (i.e., PLAUR), it activates plasminogen to convert into plasmin , which degrades extracellular matrix (ECM) components (such as fibrin and laminin) and promotes cell migration and invasion.
Integrin-mediated cell adhesion and migration signals: PLAUR forms a complex with integrins (such as αvβ3、α5β1), activating focal adhesion kinase ( FAK ) and Src family kinases, thereby regulating cytoskeleton reorganization and migration.
PI3K/AKT/mTOR pathway (promotes tumor progression): In clear cell renal cell carcinoma ( ccRCC ), high expression of PLAUR activates the PI3K/AKT/mTOR pathway, inhibiting cell apoptosis and promoting proliferation, migration and epithelial - mesenchymal transition ( EMT ).
Transcriptional regulation-mediated tumor invasion (such as breast cancer): The transcription factor TCF7L2 directly binds to the PLAUR promoter, upregulating its expression, thereby enhancing the proliferation and invasion ability of triple-negative breast cancer ( TNBC ) cells.
(Data source Mahmood N, et al. Front Oncol. 2018)
LYPD3 and disease
PLAUR is primarily implicated in disease pathogenesis by regulating tumor invasion and metastasis (e.g., renal cancer, breast cancer, and glioma), mediating cardiovascular disease risk (e.g., myocardial infarction), and promoting drug resistance. Its multiple roles as a transmembrane receptor make it an important target for cancer therapy (e.g., targeted drug or vaccine design) and cardiovascular risk assessment.
Clear cell renal cell carcinoma (ccRCC): PLAUR is significantly overexpressed in ccRCC, enhancing cell proliferation, migration, and invasion and inhibiting apoptosis by activating the PI3K /AKT/mTOR signaling pathway. Clinical analysis has shown that its high expression is associated with poor patient prognosis.
Triple-negative breast cancer (TNBC): The transcription factor TCF7L2 promotes TNBC cell proliferation, migration, and invasion by upregulating PLAUR expression . In animal studies, silencing PLAUR significantly inhibited tumor growth.
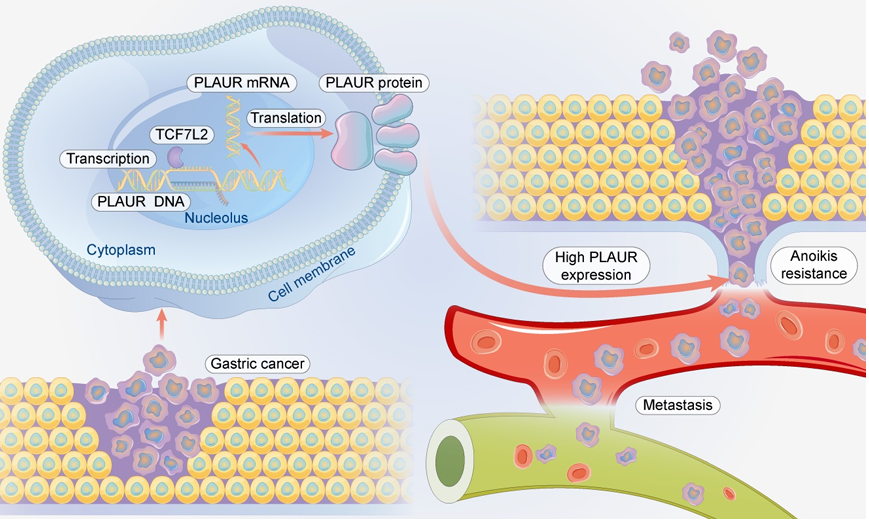
Gastric cancer: In the nuclei of gastric cancer cells, PLAUR DNA is transcribed and, with the participation of TCF7L2 , PLAUR mRNA is generated . The mRNA then exports to the cytoplasm and is translated into PLAUR protein. High PLAUR expression gives gastric cancer cells anoikis resistance , allowing them to survive even without the original extracellular matrix, helping the cells invade blood vessels and subsequently metastasize.
(Data source: Tao Zhang, et al. Int J Biol Sci. 2022)
LYPD3-targeted therapy
Lupartumab is a monoclonal antibody targeting LYPD3, developed by Bayer AG . Its primary mechanism of action is as a LYPD3 inhibitor , primarily for the treatment of advanced, malignant solid tumors. A Phase 1 clinical trial began on May 22, 2014, and has since been terminated.
FG-001 , a small molecule fluorescent dye developed by FluoGuide AS , targets uPAR . Its mechanism of action is as a uPAR modulator (urokinase-type plasminogen activator receptor modulator), primarily used for real-time visualization and localization of tumor tissue during neurosurgery. A Phase II clinical trial began on March 27, 2023.
WAL-0921 is a uPAR- targeting monoclonal antibody developed by Walden Biosciences, Inc. Its primary mechanism of action is as a uPAR inhibitor (urokinase-type plasminogen activator receptor inhibitor) for the treatment of diabetic nephropathy, glomerular disease, and IgA nephropathy. Phase II clinical trials began on July 2, 2024.
