Service Support
Mabnus Bio was responsible for the customized development of EGFR (25-645), EGFR (669-1210) truncations, and NEK7 active enzyme proteins, as well as SPR interaction detection services in this study.

( Data source: Project Active Protein Development Data )
Background
Hepatocellular carcinoma (HCC), the most common type of liver cancer, ranks fourth globally as the sixth most common cancer and the leading cause of cancer-related death . Lenvatinib is a first-line treatment for patients with inoperable HCC. However, persistent lenvatinib can exert selective pressure on HCC cells, leading to HCC recurrence. Therefore, the mechanisms of lenvatinib resistance need to be investigated . Accumulating evidence suggests that lenvatinib resistance can be acquired in HCC cells through kinase rearrangement mechanisms .

Huang Qibo from the Liver Surgery Center of Wuhan Union Hospital of China, published an article titled " NEK7-induced phosphorylation of EGFR on serine 1070 drives the acquired lenvatinib resistance in hepatocellular carcinoma " in Hepatology (IF= 15.8). The study identified NEK7 as a driver of acquired lenvatinib resistance by directly binding to and phosphorylating EGFR at serine 1070. Inhibitory peptides designed targeting the domain from amino acids 979 to 1099 were shown to inhibit EGFR S1070 phosphorylation and therapeutically suppress the acquired anti-tumor effects of lenvatinib in mouse hepatocellular carcinoma cells.
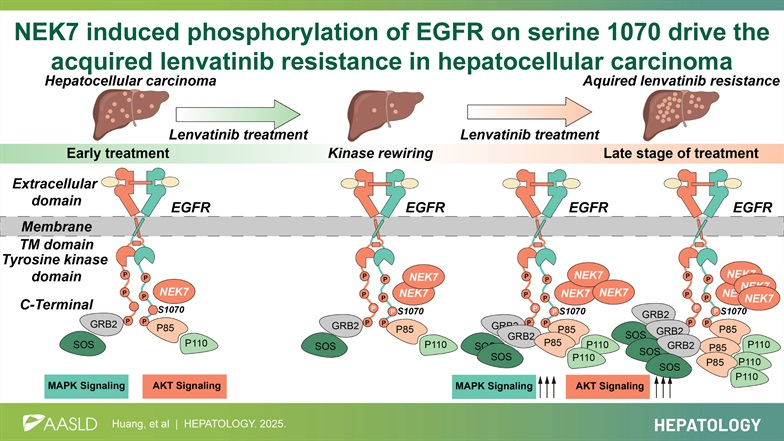
NEK7 is a gene that contributes to acquired resistance to lenvatinib in hepatocellular carcinoma
A patient-derived organoid model of acquired lenvatinib resistance (ALR-PDOX) was constructed, and NEK7 was identified as a key resistance gene using a CRISPR-Cas9 kinase knockout library. NEK7 is a key kinase that contributes to acquired lenvatinib resistance in HCC.

Inhibition of NEK7 protein can increase the sensitivity of hepatocellular carcinoma to lenvatinib
In both in vitro and in vivo models, NEK7 knockout significantly reduced the IC50 value of lenvatinib in HCC cells, significantly reduced the number of tumors and maximum tumor volume in mice , and NEK7 expression levels correlated with lenvatinib resistance. Specific knockout of the Nek7 gene in hepatocytes effectively suppressed acquired lenvatinib resistance in the Trp53 KO/MYC OE model. In a subcutaneously implanted tumor model, knockdown of the NEK7 gene inhibited tumor progression by enhancing the efficacy of lenvatinib. Knockdown or ablation of the NEK7 gene restored the efficacy of lenvatinib and inhibited the development of acquired resistance in both in vitro and in vivo experiments.

NEK7 promotes acquired lenvatinib resistance through the EGFR signaling pathway
NEK7 is a serine/threonine kinase that plays a key role by adding phosphate groups to specific serine or threonine residues on target proteins . NEK7 affects the EGFR signaling pathway and its downstream MAPK and PI3K/AKT signaling pathways. Knockdown of NEK7 inhibits activation of both the MAPK and PI3K/AKT signaling pathways.
Using Crispr/Cas9 technology, EGFR knockout cell lines were constructed in Hep3B LR and Huh7 LR cells to explore the relationship between NEK7 and EGFR. The results showed that EGFR knockout significantly inhibited the proliferation of Hep3B LR and Huh7 LR cells, and the half-inhibitory concentration of these cells after lenvatinib treatment was lower than that of the LR vector group. Overexpression of NEK7 was unable to rescue the loss of downstream inhibitory effects after EGFR knockout. In vivo experiments further confirmed that even under lenvatinib treatment, NEK7 overexpression could not rescue the growth of xenograft tumors after EGFR knockout. NEK7 promotes acquired lenvatinib resistance in liver cancer cells in an EGFR-dependent manner.

EGFR C -terminal tail residue S1070 is phosphorylated by NEK7
By constructing multiple truncated forms of EGFR, it was found that both full-length EGFR and truncated EGFR could interact with NEK7. In vitro GST pull-down experiments confirmed that the intracellular segment of EGFR (669-1210) directly binds to NEK7.
Phospho-mass spectrometry analysis revealed that NEK7 activates EGFR at the S1026 and S1070 phosphorylation sites. Immunoprecipitation experiments demonstrated that S1070 plays a key role in the pan-phosphorylation of EGFR induced by NEK7 overexpression. Furthermore, EGFR S1070 is highly conserved across species, and in vitro phosphorylation experiments confirmed that NEK7 can phosphorylate EGFR S1070.

Phosphorylation of S1070 in the EGFR tail promotes the interaction between EGFR, P85, and GRB2
The EGFR signaling pathway plays a key role in regulating the MAPK and PI3K/AKT signaling pathways . When the C-terminal tail of the GRB2 protein is phosphorylated, its SH2 domain binds and recruits the SOS complex, thereby activating the MAPK signaling pathway. Studies have shown that the SH2 domain of the PI3K regulatory subunit P85 also has similar functions.
Exogenous overexpression and quantitative immunoprecipitation of endogenous proteins confirmed that the S1070A mutation also weakened the ability of P85 and GRB2 to interact with EGFR. NEK7 overexpression led to membrane aggregation of P85 and GRB2 after EGF stimulation. NEK7 knockdown, however, inhibited EGF's promotion of P85 and GRB2 membrane recruitment. The EGFR S1070A mutation may limit the formation of the P85-EGFR complex. Phosphorylation at the EGFR S1070 site plays a key role in the lenvatinib resistance mechanism mediated by the EGFR-P85 and EGFR-GRB2 pathways, with both pathways influencing each other .

NEK7-induced phosphorylation of EGFR C-terminal tail residue S1070 promotes acquired resistance to lenvatinib
The expression levels of EGFR S1070 and NEK7 are closely associated with the efficacy of neratinib treatment and the progression of liver cancer. The EGFR S1070 phosphorylation site can be used as a prognostic marker for patients treated with neratinib.

Pharmacological blockade of EGFR S1070 alleviates acquired lenvatinib resistance in HCC
By utilizing the Alphafold database to obtain structural information from the 979-1099 region of the EGFR C-terminal tail, two cell-penetrating peptides, TAT, were designed. In vitro and in vivo experiments demonstrated that TAT-pep10 significantly inhibited activation of the MAPK and PI3K/AKT signaling pathways in Hep3B LR cells. Quantitative co-immunoprecipitation experiments demonstrated that, following treatment of cells with TAT-pep10, the interaction of EGFR with NEK7, P85, and GRB2 was inhibited in a concentration-dependent manner in 293T cells. TAT-pep10 also inhibited the phosphorylation of EGFR S1070 and its downstream pathways in Hep3B LR cells in a concentration-dependent manner.
Surface plasmon resonance (SPR) detection showed that TAT-pep10 could interact with the EGFR intracellular membrane residues (positions 669-1210).
NEK7-mediated EGFR S1070 phosphorylation plays a key role in acquired lenvatinib resistance, so targeting EGFR C-terminal tail residue S1070 may be a potential strategy for treating this resistance.

Summarize
This study discovered a new EGFR phosphorylation site, S1070. NEK7 plays a crucial role in the development of acquired resistance to lenvatinib. In addition, NEK7 -mediated phosphorylation of the S1070 site on EGFR is essential for the stimulation of GRB2 and P85 on the cell membrane, which are regulatory subunits of the MAPK and PI3K/AKT signaling pathways. NEK7 -mediated EGFRS1070 phosphorylation is not only associated with acquired resistance to lenvatinib, but is also positively correlated with poor overall survival. Pharmacological inhibition of the EGFR C- terminal tail enhances the effectiveness of lenvatinib treatment and inhibits the progression of HCC. In summary, this study expands the understanding of NEK7- mediated EGFR phosphorylation in HCC. This study improves our understanding of acquired resistance to lenvatinib and highlights the potential of the EGFR C- terminus as a target for the treatment of acquired lenvatinib resistance.
