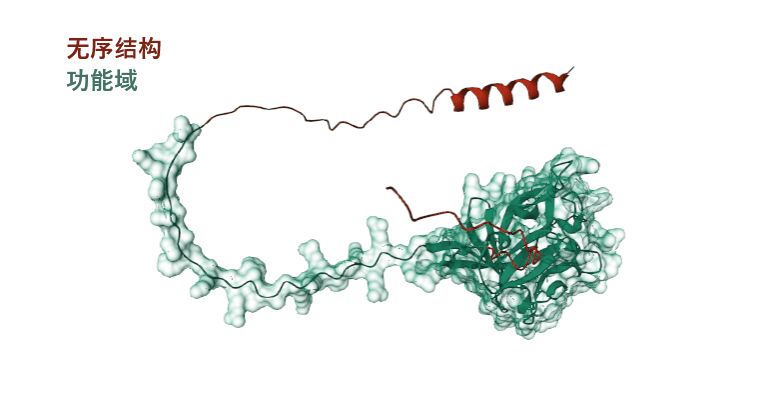
(Data source: AlphaFold)
Fibroblast growth factor 12 (FGF12) is a member of the FGF11 subfamily, composed of 243 amino acids. It is an intracellular non-secretory protein. The main functional domain is a full-length segment, which contains a disordered structure at the N-terminus and C-terminus respectively.

(Data source: Uniprot)
FGF12 research can be divided into two categories: the study of the regulatory mechanism of normal FGF12 and the clinical relevance of variant FGF12. Let's take a look at them one by one:
Loss of bone morphogenetic protein (BMP) signaling induces a phenotypic switch in pulmonary artery smooth muscle cells (PASMCs), which underlies the pathological development of pulmonary vascular remodeling in pulmonary arterial hypertension (PAH). FGF12, a novel regulator of BMP-induced phenotypic changes in PASMCs, may modulate this phenotypic switch and improve pulmonary vascular remodeling in PAH, suggesting potential therapeutic value.
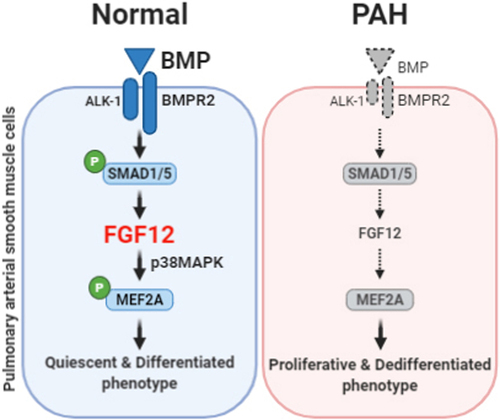
(Data source: Yeo Y, et al. Hypertension. 2020)
Chronic liver disease is associated with the development of liver fibrosis, which often leads to cirrhosis if left untreated. Deletion of FGF12 in macrophages protects against BDL-induced and CCL4-induced liver fibrosis. Inhibiting macrophage FGF12 may be a promising therapeutic approach to combat liver fibrosis in the future.

(Data source: Li S, et al. Hepatology. 2022)
FGF12 also regulates hair growth: FGF12 siRNA transfection downregulates FGF12 expression in the outer root sheath (ORS) of mice, delaying the transition from telogen to anagen, reducing hair length in vibrissa follicles, and inhibiting ORS cell proliferation and migration. Furthermore, FGF12 overexpression modulates the expression of PDGF-C, MDK, and HB-EGF, factors that promote hair growth. FGF12 adenovirus was also used for FGF12 overexpression. Overall, FGF12 promotes hair growth by inducing the anagen phase of hair follicles, suggesting its potential as a treatment for hair loss.
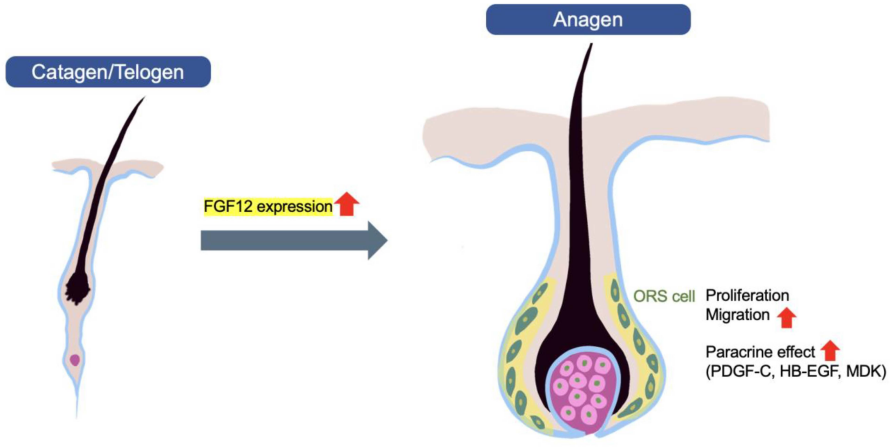
(Data source: JHS , et al. Int J Mol Sci. 2022)
FGF11 subfamily proteins interact with the C-terminal tail of NaVs (voltage-gated sodium channels (NaVs ) are the backbone of neuronal function. Mutations in the genes encoding NaVs( NaV1.1 [ SCN1A ], NaV1.2 [SCN2A], NaV1.3 [SCN3A] , and NaV1.6 [SCN8A]) are responsible for some of the most common and severe inherited epilepsy and epileptic encephalopathy ) to regulate both rapid and prolonged channel inactivation. Brugada syndrome (BrS) is a potentially life-threatening inherited cardiac arrhythmia caused by ion channel disorders resulting from mutations in the NaVs gene (NaV1.5). The Q7R mutant of FGF12 reduces binding to the C-terminus of the NaV1.5 protein, affecting Na+ channel trafficking and kinetics, providing multifaceted evidence supporting Q7R-FGF12 as a BrS-associated mutation.

(Data source: Hennessey JA, et al. Heart Rhythm. 2013)
Different FGF12 variants mediate differential regulation of NaV1.2 and NaV16 channels, implicating FGF12 in its complex effects on neuronal network activity and suggesting that FGF12 may be amenable to precision therapy with sodium channel blockers.

(Data source: Seiffert S, et al. EBioMedicine. 2022)
The activities involved in both normal FGF12 regulation and variant disorders are relatively complex. Although there are many aspects involved, further exploration is needed in terms of mechanism discovery.
