Fibroblast growth factor 23 (FGF23) belongs to the FGF19 subfamily and is synthesized by osteoblasts, osteocytes and bone marrow. It is an important regulator of phosphate and calcium homeostasis and is also related to iron homeostasis, inflammation and erythropoiesis.
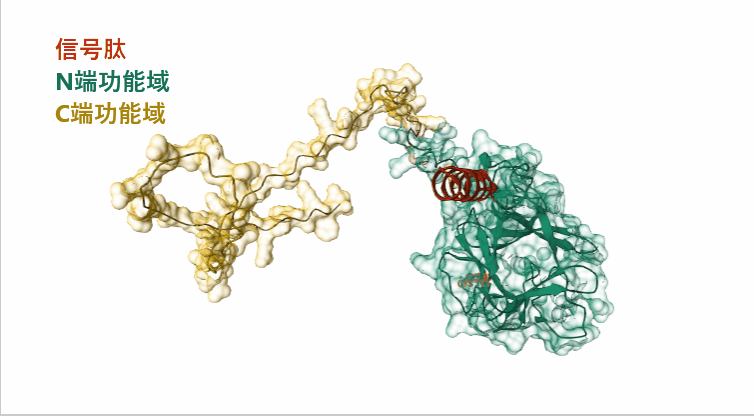
(Data source: AlphaFold)
FGF23 is composed of 251 amino acids and is an endocrine extracellular protein. The 1-24 segment is a signal peptide that guides secretion. The main functional domain is the 25-251 segment, which is also the full-length form of wild-type FGF23, including a pair of disulfide bonds, 2 glycosylation sites and 1 phosphorylation site amino acid modification.
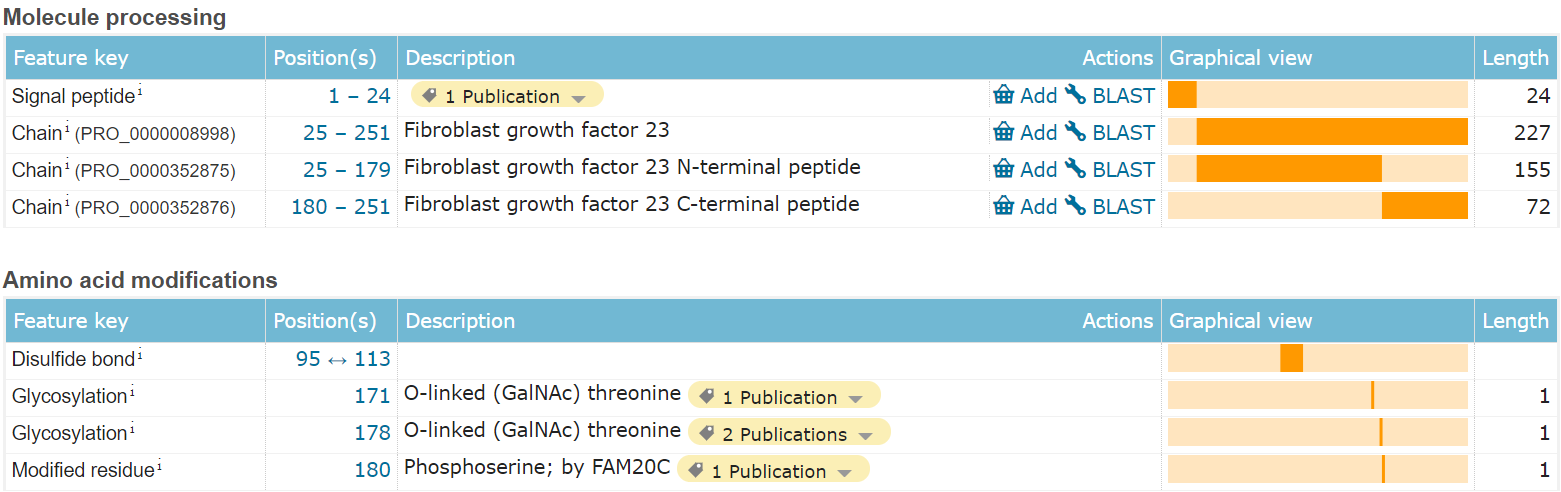
(Data source: Uniprot)
Many of FGF23's important structural and physiological significance are linked to its C-terminus. Circulating levels of physiologically active full-length FGF23 are regulated by a highly conserved proteolytic cleavage site at the C-terminus ( a furin-converting proenzyme recognition site located at Arg176 to Arg179 ). FGF23 also undergoes multiple post-translational modifications that influence its stability and susceptibility to proteolysis. Phosphorylation at Ser180 by extracellular kinase family member 20C (FAM20C) prevents post-translational O-glycosylation by polypeptide N-acetylgalactosaminyltransferase 3 (GALNT3). This lack of O-glycosylation predisposes FGF23 to hydrolysis into two inactive segments, 25-179 and 180-251.
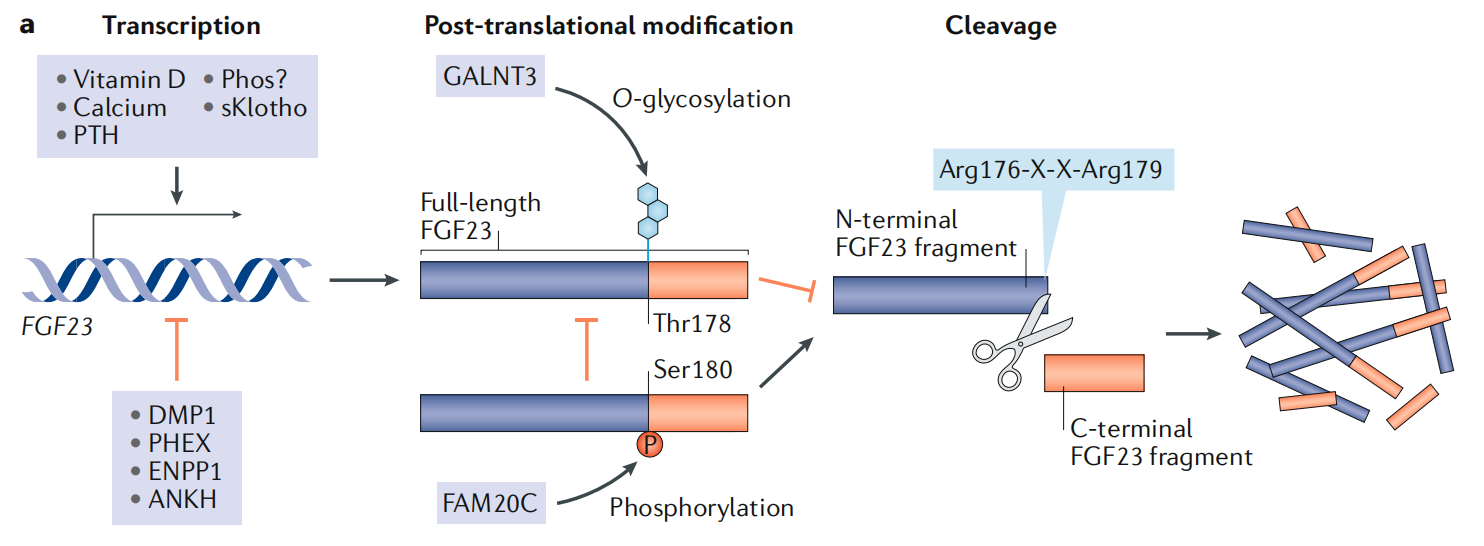
(Data source: Edmonston D, et al. Nat Rev Nephrol. 2020)
In terms of signal transduction, FGF23 regulates the co- receptor α-Klotho (KL ) activates FGFR1c, FGFR3, and FGFR4 in a Klotho-dependent or -independent manner. In most cases, FGFR1c is the major endocrine receptor for FGF23 and is mediated by α - Klotho.
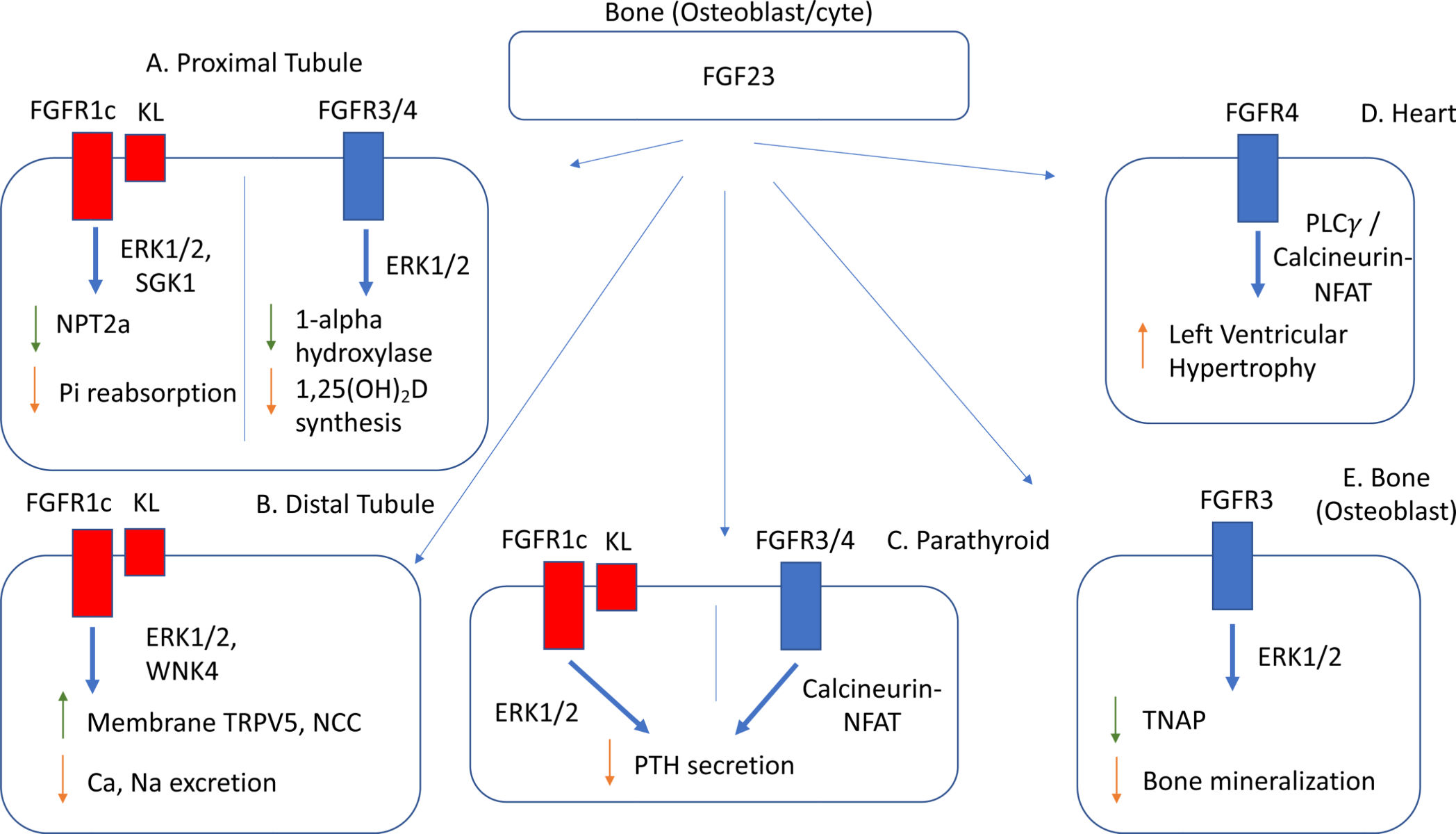
(Data source: Bryan B Ho , et al. J Mol Endocrinol. 2021)
α - Klotho increases the binding affinity of FGF23 to FGFR1c by 20-fold. The dimerization and receptor activation of the stable FGF23, FGFR1c extracellular domain and α - Klotho ternary complex still depend on heparan sulfate (HS), thereby activating downstream pathways.

(Data source: Chen G, et al. Nature. 2018)
As a bone and bone marrow-derived hormone, FGF23 is crucial for maintaining phosphate homeostasis. It primarily acts as a regulator of PTH metabolism through the MAPK (ERK1 and 2, and Akt) pathway, inhibiting vitamin D3 synthesis in the kidney to lower serum phosphate levels. FGF23 deficiency leads to hyperphosphatemia and ectopic calcification, while FGF23 excess can cause hypophosphatemia, bone defects, and kidney-related diseases.

(Data source: Phuc P , et al. Cells. 2021)
An increasing number of local and systemic factors regulate FGF23 transcription and cleavage to control overall FGF23 activity and content, involving many different organ systems. For example, in patients with chronic kidney disease (CKD) and acute kidney injury (AKI), FGF23 is affected through inflammation, iron deficiency, anemia, and metabolism-related pathways.
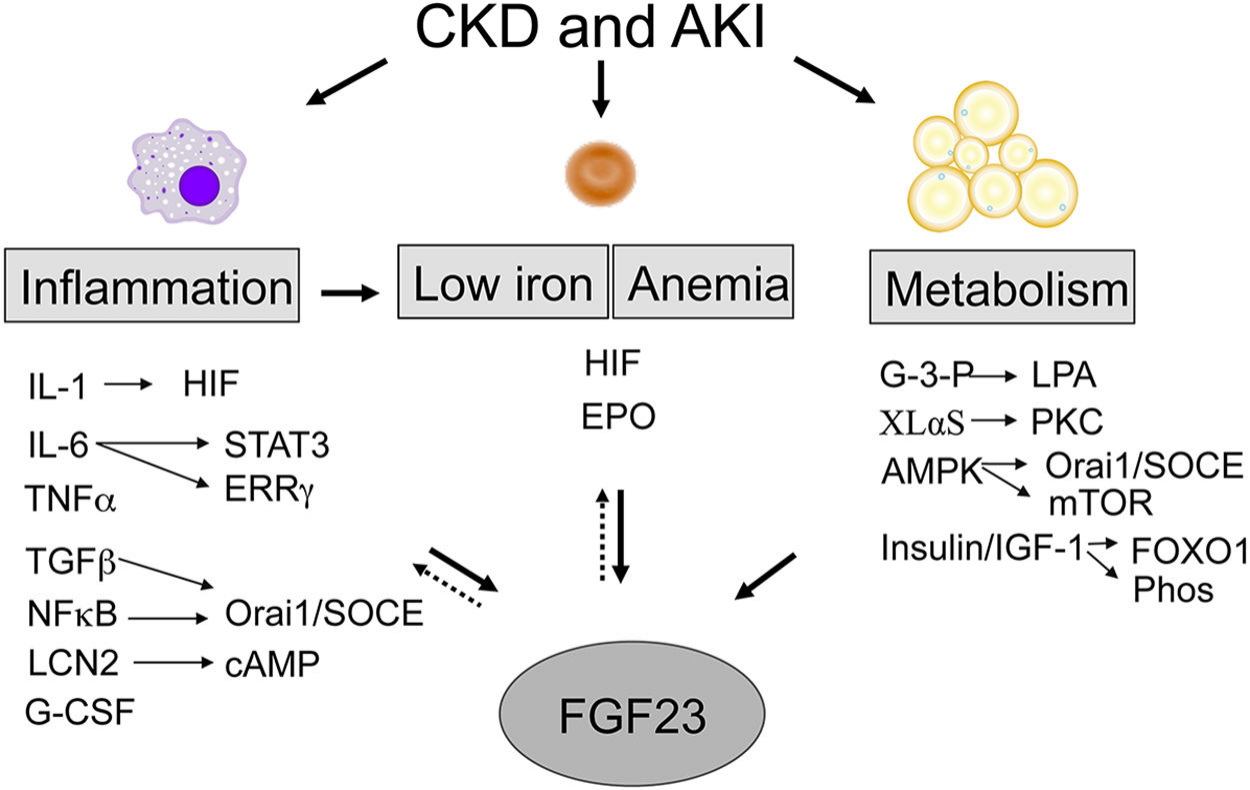
(Data source: Simic P, et al. Curr Osteoporos Rep. 2021)
Given the important role FGF23 plays in many human diseases, it has been an ideal target for therapeutic drug development, including the development of diagnostic antibodies that recognize multiple forms of FGF23.
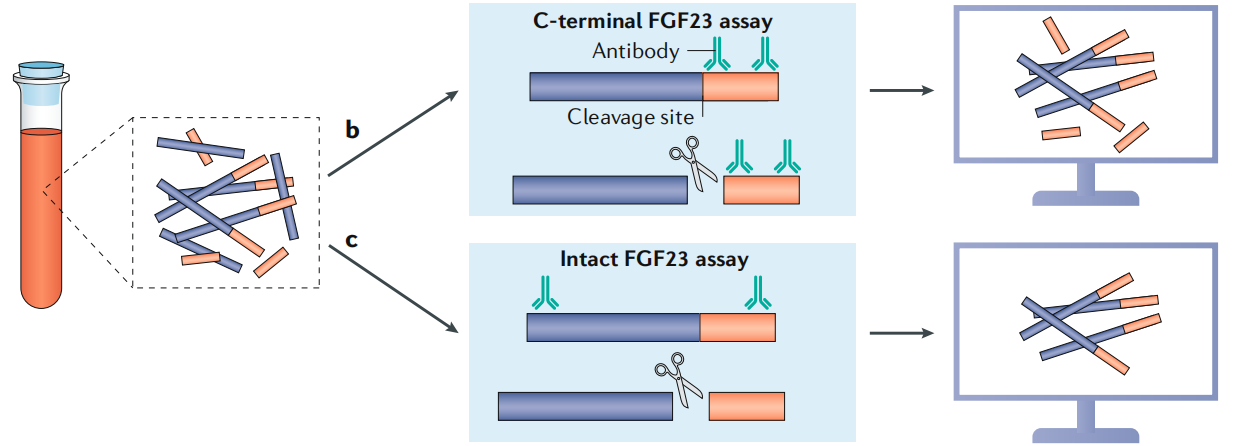
(Data source: Edmonston D, et al. Nat Rev Nephrol. 2020)
Currently, the most advanced FGF23 therapeutic drug is Kyowa Hakko Kirin's KRN23 (Burosumab, trade name Crysvita, a recombinant fully human anti-FGF23 IgG1 monoclonal antibody that binds to FGF23 and blocks its biological activity). It was approved by the FDA on April 18, 2018, for the treatment of X-linked hypophosphatemic rickets (XLH, the main feature of which is low serum phosphate levels, causing skeletal deformities and developmental disorders) in children one year and older and adults. It has significantly improved the severity of rickets and growth in children with XLH.
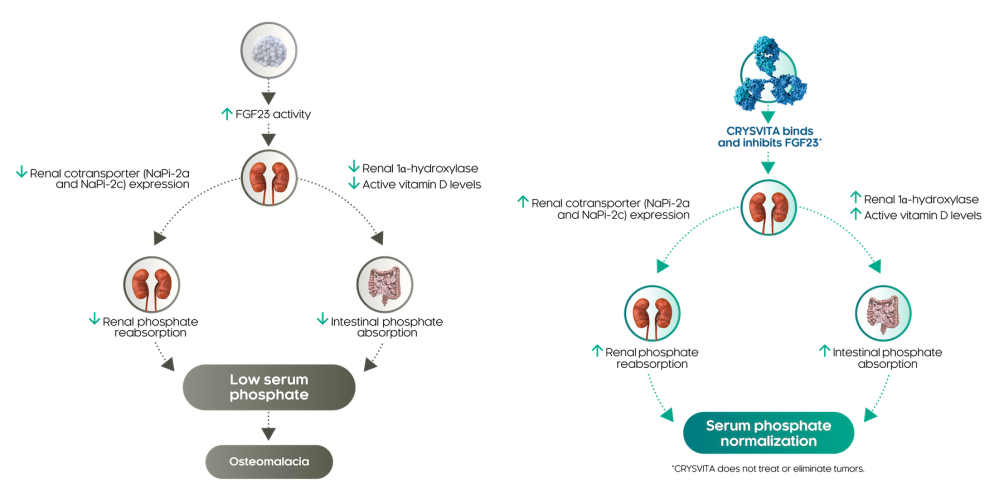
(Data source: Kyowa Hakko Kirin official website)
The endocrine effects of FGF23 are now well established and are regulated at three major steps: transcription, post-translational modification, and peptide cleavage. However, the multiple paracrine effects in different tissues are less well understood. Furthermore, the regulation of FGF23 under physiological and pathophysiological conditions, including transcriptional and post-transcriptional mechanisms, remains unclear. Further studies are necessary to better understand the full range of FGF23 regulatory significance.
