Fibroblast growth factor 6 (FGF6) is an important regulatory member of the mammalian muscle regeneration process. It belongs to the cytokine family that controls cell proliferation, cell differentiation and morphogenesis events.

(Data source: AlphaFold)
FGF 6 is composed of 208 amino acids and is a secreted extracellular protein. Its main functional domain is the 38-208 segment, which includes one N-type glycosylation and a pair of disulfide bond modifications.
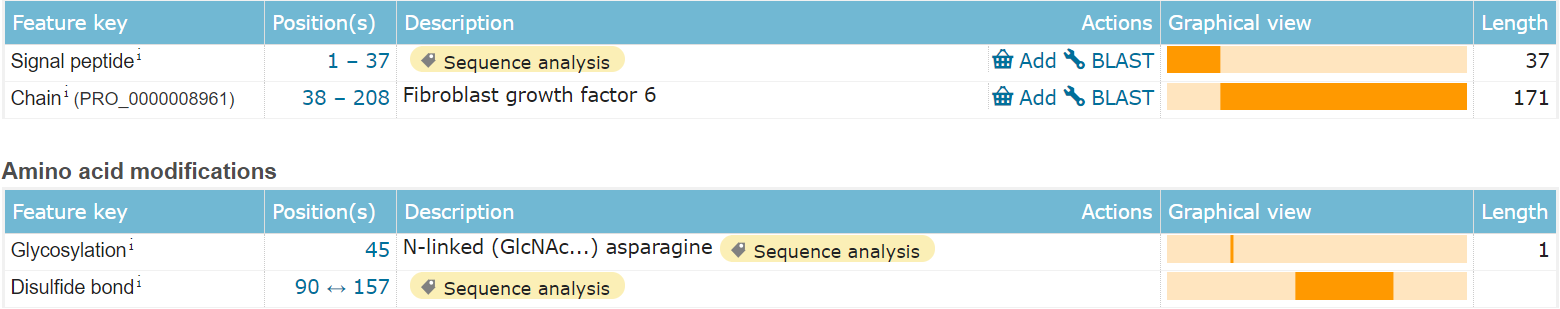
(Data source: Uniprot)
FGF6 exhibits a restricted expression profile and is primarily expressed in the myogenic lineage.
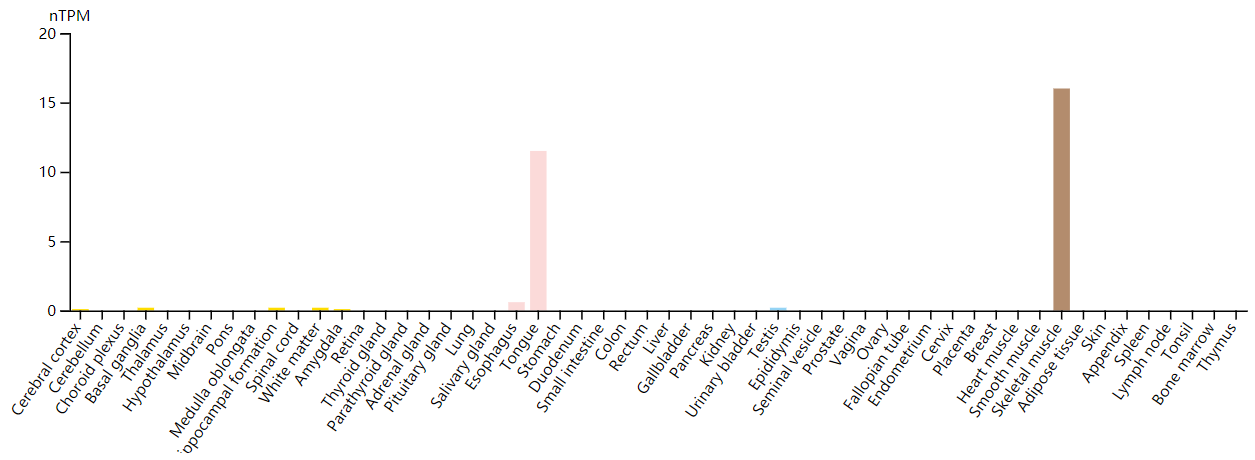
(Data source: Protein Atlas)
Physiologically, exercise and cold upregulate FGF6/9 expression in fat, and FGF6/9 is a potent inducer of uncoupling protein 1 (UCP1) expression in adipocytes and preadipocytes, thereby mediating energy dissipation in brown adipose tissue (BAT) and playing a role in regulating whole-body energy metabolism.
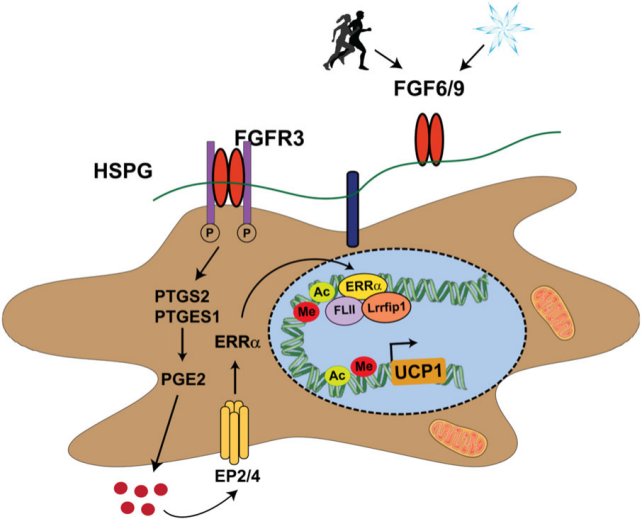
(Data source: Shamsi F, et al. Nat Commun. 2020)
Overexpression of FGF6 stimulates protein synthesis in mouse skeletal muscle and prevents weight gain and the development of insulin resistance in mice fed a high-fat diet, suggesting its potential in strategies for the prevention and treatment of energy metabolic diseases.
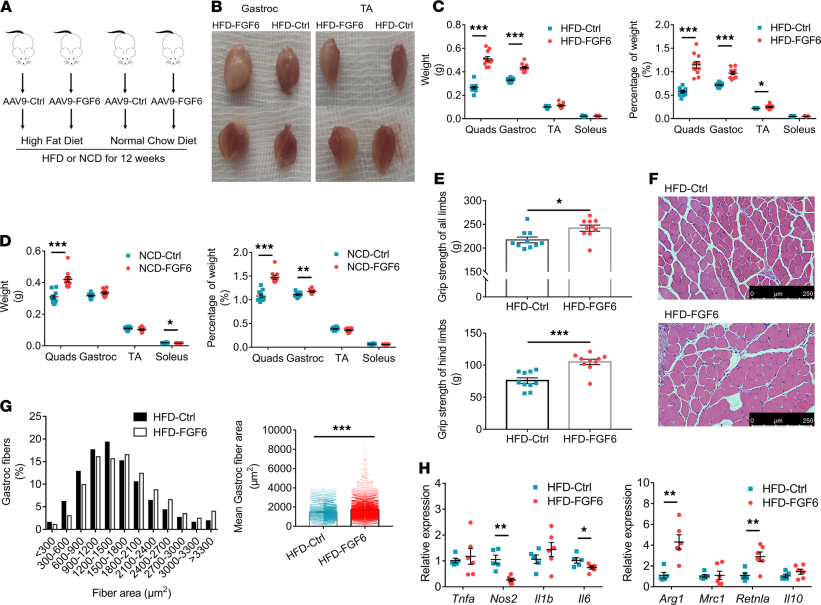
(Data source: Bo Xu , et al. JCI Insight. 2021)
On the other hand, FGF6 promoted the viability and migration of C2C12 and primary myoblasts in a dose-dependent manner through FGFR1-mediated upregulation of cyclin D1. Low concentrations of FGF6 promoted myoblast differentiation through FGFR4-mediated activation of ERK1/2, thereby upregulating the expression of MyHC, MyoD, and myogenin, thereby promoting the functional recovery of regenerated skeletal muscle after innervation.

(Data source: Cai Q, et al. Life Sci. 2020)
At the same time, FGF6 prevents myocardial infarction damage by inhibiting the Hippo pathway and promoting the nuclear accumulation of YAP, thereby promoting myocardial cell regeneration and cardiac repair.
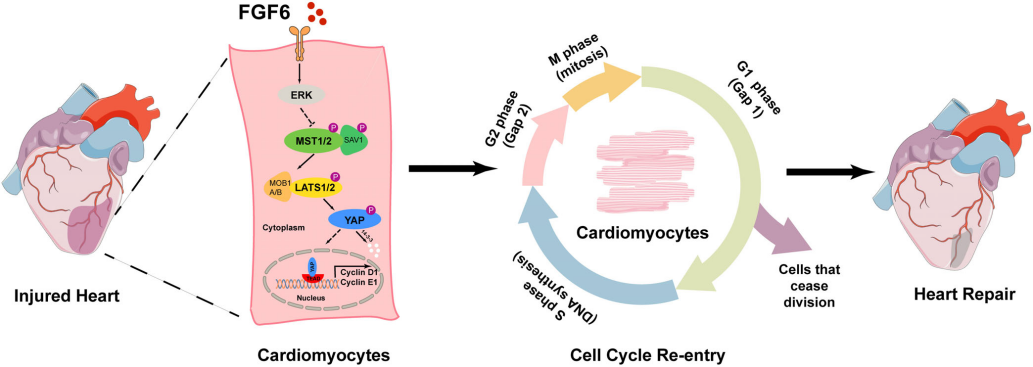
(Data source: Hu Z, et al. Cell Prolif. 2022)
FGF6 is also associated with hemochromatosis: loss-of-function FGF6 variants silence the FGF6-FGFR pathway, increase free heparin, and reduce hepcidin expression, thereby reducing the inhibition of ferroportin-mediated iron transfer from intracellular compartments to the blood (i.e., increasing plasma iron levels).
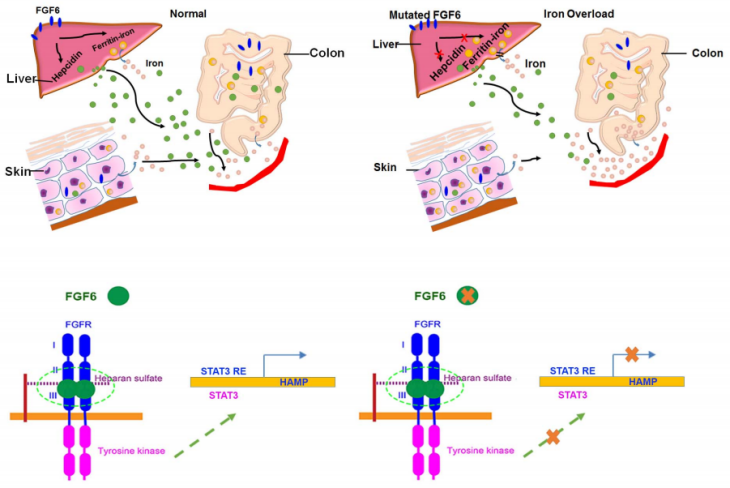
(Data source: Guo S, et al. Blood. 2019)
These studies not only demonstrate the regulatory role of FGF6 in energy metabolism but also in the regeneration of myogenic cells, providing new clues and ideas for potential therapeutic approaches to repair muscle damage. The FGF6 & FGFR signaling axis is also a crucial mechanism in iron metabolism.
