The fibroblast growth factor (FGF) 8 subfamily consists of FGF8, FGF17, and FGF18. FGF8 subfamily members regulate embryonic development by controlling the growth and differentiation of progenitor cells and also play a role in adult tissue repair to maintain tissue homeostasis. By regulating multiple cellular signaling pathways, the FGF8 subfamily is involved in altering craniofacial development, odontogenesis, tongue development, and salivary gland branching morphogenesis.
(Data source: Francavilla C, et al. Open Biol. 2022)
FGF8 subfamily members have similar receptor binding specificity. They exhibit high binding affinity to the 3C variants of FGFR1-3 and non-splicing FGFR4. FGF8 and FGF17 have been shown to have higher binding affinity to FGFR3c and FGFR4, but weaker binding affinity to FGFR1c. Similarly, FGF18 preferentially binds to FGFR3c over FGFR2c.
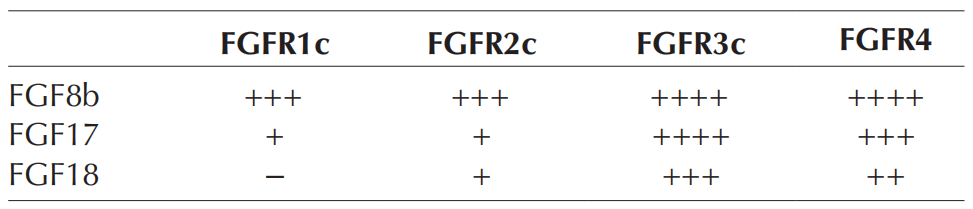
(Data source: Estienne A, et al. Reproduction. 2018)
Subfamily signaling caused by mutations, abnormal expression, or isoform splicing plays an important role in the development of oral diseases. Targeting FGF8 subfamily members provides a new and promising strategy for treating oral diseases. Below we take a closer look at the information about FGF8 members:
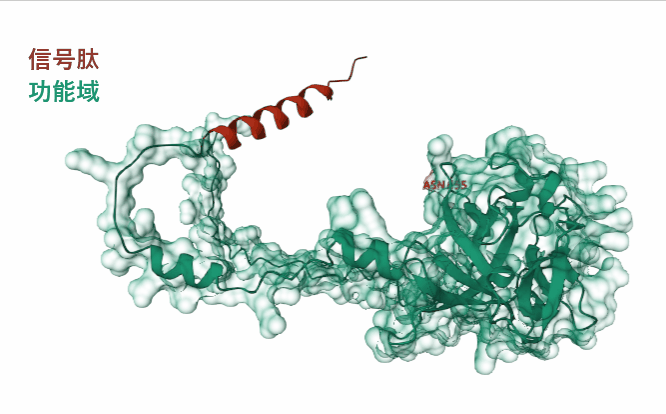
(Data source: AlphaFold)
FGF8 is composed of 233 amino acids and is a secreted extracellular protein. The main functional domain is the 23-233 segment, which includes an N-type glycosylation modification.
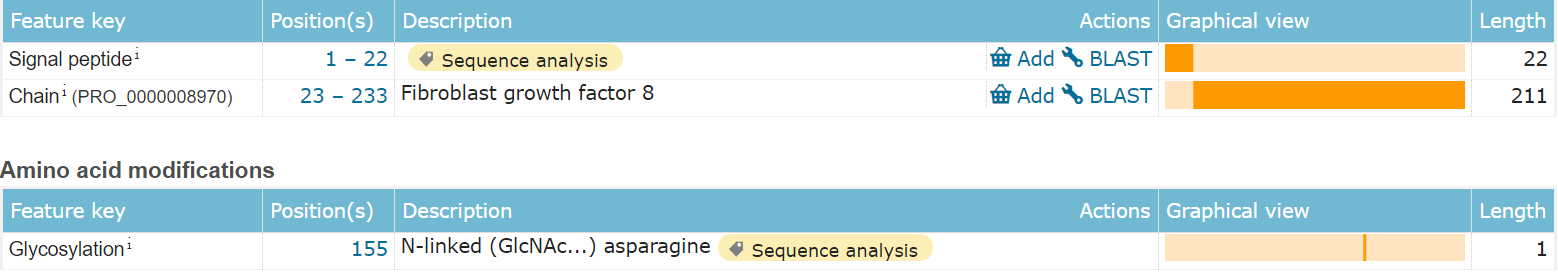
(Data source: Uniprot)
FGF8 isoforms work together to play diverse and important roles during vertebrate development. The human FGF8 gene has four alternatively spliced isoforms—a, b, e, and f. Common mutations include the H14N mutation in the signal peptide region and three heterozygous mutations in the constant region: K100E (K71E in FGF8b), R127G (R98G in FGF8b), and T229M (T200M in FGF8b).
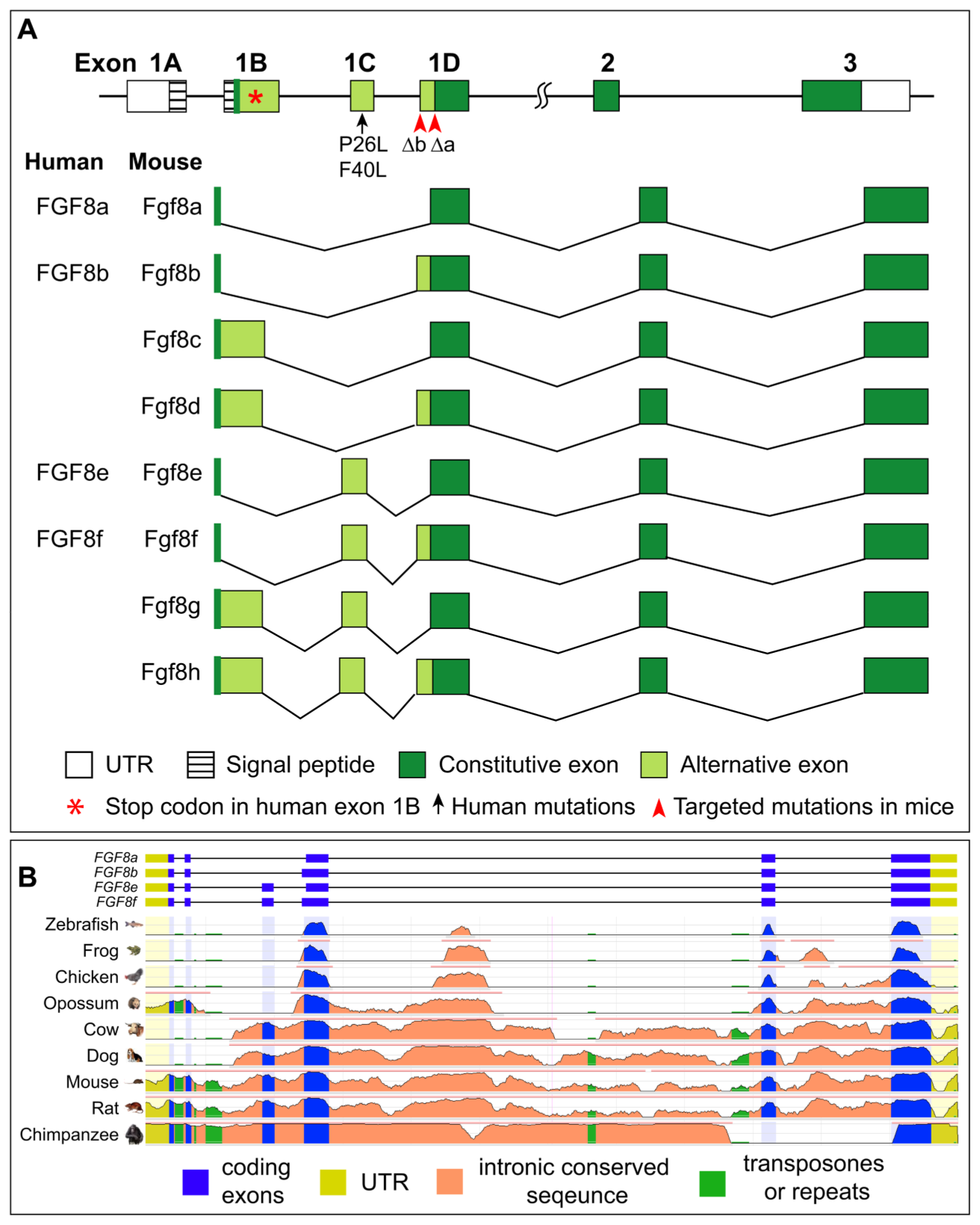
(Data source: Sunmonu NA, et al. J Cell Physiol. 2011)
Duplication of the FGF8 locus is associated with human femoral dysplasia, and the phenotype is likely the result of positional effects altering FGF8 expression.
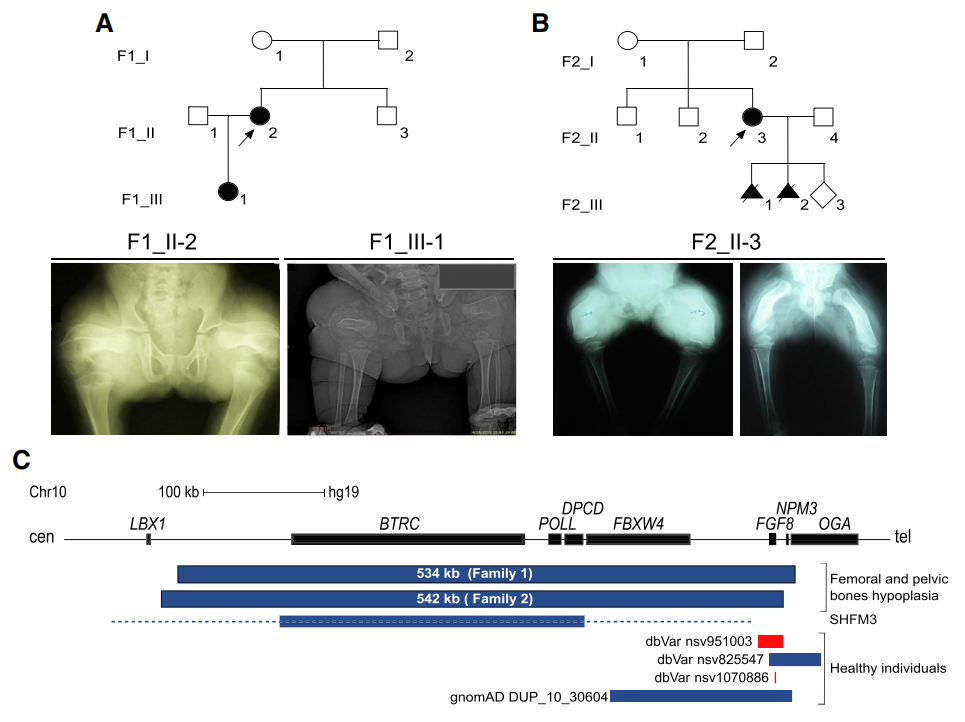
(Data source: Liu X, et al. Int J Biol Sci. 2021)
The D73H missense mutation of FGF8 is involved in the development of cleft lip or cleft palate (Riley BM, et al. Proc Natl Acad Sci USA. 2007); at the same time, in a high-arched jaw mouse model, reducing FGF8 can rescue maxillary hyperplasia and palate width in compound mutants, indicating that craniofacial malformations in early development are a characteristic of FGF8 overactivation syndrome.

(Data source: Tabler JM, et al. Dev Cell. 2013)
In addition, FGF8 plays an important role in tooth development and the establishment of dental epithelial stem cell progenitors: ectopically activated epithelial FGF8 changes the cell fate of the outer enamel epithelial cells of the incisor tongue by activating several key dental genes (including Pitx2, Sox2, Lef-1, p38, and Erk1/2), and inducing the formation of a new extra incisor crown parallel to the original incisor on the lingual side, leading to the formation of the extra incisor crown and its eventual fusion with the original incisor; at the same time, excessive epithelial FGF8 signaling significantly downregulates the expression of mesenchymal Bmp4, resulting in severe damage to enamel mineralization.

(Data source: Chen Y, et al. J Dent Res. 2022)
FGF8 also plays a promoting role in the development of oral tumor diseases, inducing epithelial-mesenchymal transition and promoting metastasis of oral squamous cell carcinoma.
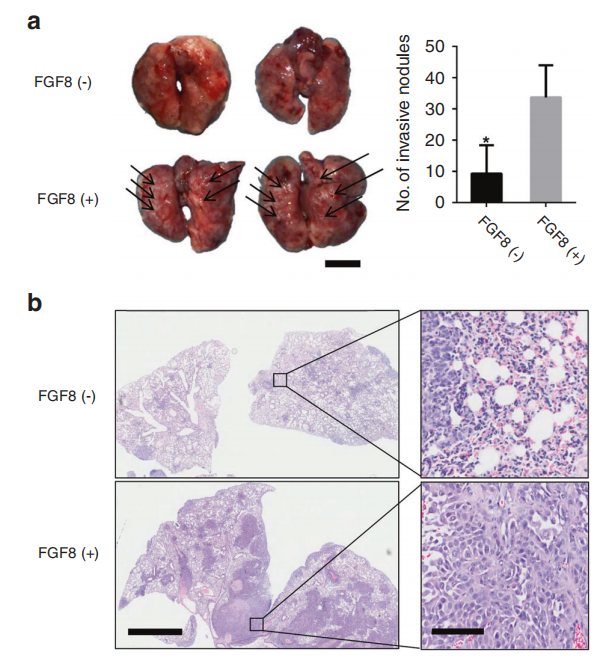
(Data source: Hao Y, et al. Int J Oral Sci. 2021)
Whether the development of oral and maxillofacial diseases is caused by FGF8 activating mutations, increased expression or abnormal isoform splicing, or the abnormal regulation of the FGF8 signaling pathway associated with the development of oral cancer, targeting FGF8 may provide a novel and promising strategy for the treatment of oral diseases. However, the detailed mechanism studies are currently relatively weak and further work is needed to clarify.
