The fibroblast growth factor (FGF) 9 subfamily consists of FGF 9, FGF 16, and FGF 20. FGF 9 subfamily members are highly expressed in cell embryos to drive the formation of neural tissue and play an important role in the regulation of embryonic development, cell proliferation, cell differentiation, and cell migration.
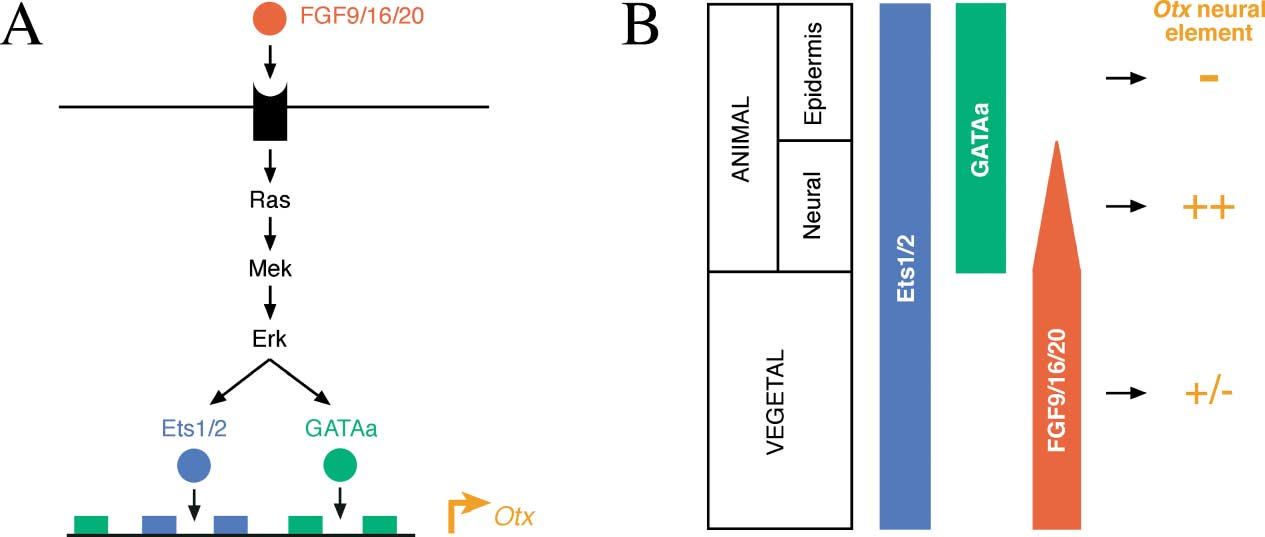
(Data source: Bertrand V, et al. Cell. 2003)
(Data source: AlphaFold)
FGF9 is composed of 208 amino acids and is a paracrine extracellular protein. It has no conventional signal peptide and its main functional domain is the 4-208 segment, which includes an N-type glycosylation modification.
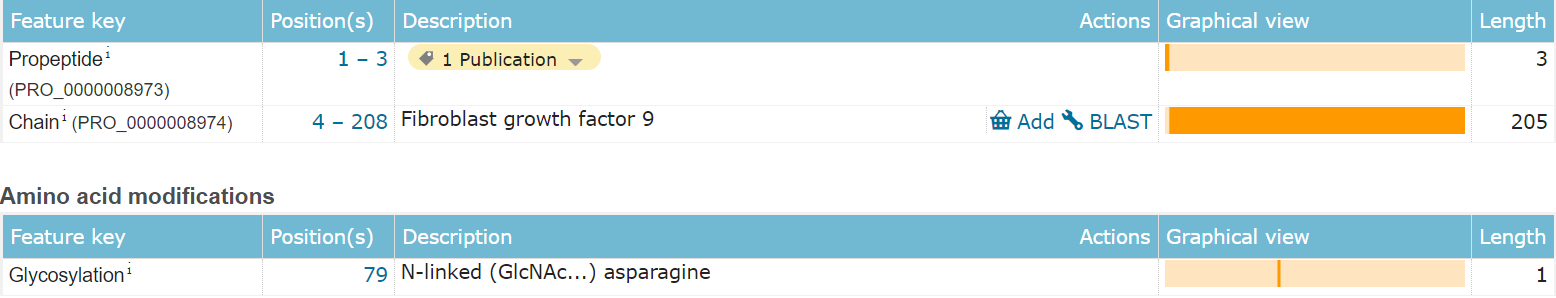
(Data source: Uniprot)
FGF9 is highly conserved among species, with minimal sequence differences.

Interesting research on FGF9 focuses on its role in regulating gonadal differentiation. Loss of FGF9 function in transgenic mice results in male-to-female sex reversal, while increased FGF9 copy number has been observed in human patients with 46,XX disorder of sex development (DSD). Specifically, SRY and SF1 regulate FGF9 expression in mice, whereas SRY-mediated FGF9 expression in humans is independent of SF1.

(Data source: Li YH, et al. Biol Reprod. 2020)
The same phenomenon and problem were also explained in an article in Science, which showed that intrachromosomal inversion of FGF9 in mole mice changed the regulatory landscape of gonadal expression patterns.

(Data source: M Real F, et al. Science. 2020)
Physiologically, exercise and cold exposure upregulate FGF9 expression in adipose tissue. FGF9 is a potent inducer of uncoupling protein 1 (UCP1) expression in adipocytes and preadipocytes, mediating energy dissipation in brown adipose tissue (BAT) and regulating systemic energy metabolism. More strikingly, FGFR3 expression in human white fat is negatively correlated with body mass index (a measure of obesity) and insulin resistance (a condition that can lead to type 2 diabetes). This suggests that activating the FGF9 & FGFR3 pathway could benefit patients with obesity, diabetes, and related metabolic diseases.
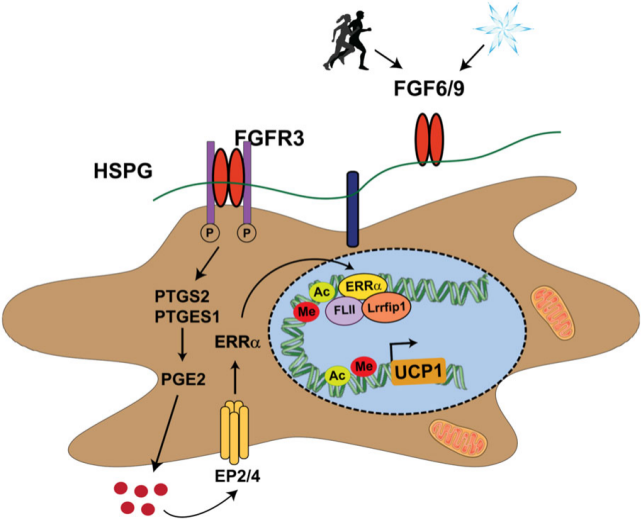
(Data source: Shamsi F, et al. Nat Commun. 2020)
FGF9 are also the main cause of multiple synostosis syndrome. As a negative regulator of bone homeostasis, FGF9 provides a potential therapeutic target for bone degenerative diseases by regulating osteogenesis and osteoclastogenesis.

(Data source: Tang L, et al. J Bone Miner Res. 2021)
In addition, the overexpression of FGF9 in various tumor tissues (prostate cancer @Huang Y, et al. Int J Biol Sci. 2015, gastric cancer @Wang R, et al. J Exp Clin Cancer Res. 2019, etc.) promotes tumor progression, which also provides the possibility of targeting FGF9 to treat tumor diseases. However, whether FGF9 has unlimited possibilities still needs in-depth exploration.
