Background
The Fc region of IgG isotype therapeutic monoclonal antibodies is often engineered to optimize their pharmacology and pharmacokinetic (PK) properties, with safety also a key development consideration. Fc engineering can enhance antibody effector functions (e.g., ADCC, ADCP, and CDC) primarily by improving the Fc region's ability to bind to activating FcγRs or complement component C1q. Furthermore, enhancing Fc-Fc hexamerization or FcγRIIB interactions through specific amino acid substitutions can achieve receptor clustering-based agonist activity. Alternatively, reducing FcγR and C1q binding can achieve (partial) "silencing" of the Fc domain. Multimerization of the Fc domain is also an effective strategy for blocking Fcγ receptors (FcγRs) and the neonatal Fc receptor (FcRn).

On July 7, 2025, Professor Brennan's research team published an article titled "Impact of antibody Fc engineering on translational pharmacology, and safety: insights from industry case studies" in mAbs vol. 1. The article details that engineering the Fc region to achieve desired pharmacology and pharmacokinetics is a widely adopted strategy in drug development. The Fc region can be modified through amino acid substitutions and glycoengineering, resulting in enhanced or reduced effector function, preferentially immunobinding to specific FcR subtypes or pH-dependent binding to FcRn. These alterations in mAb binding and effector activity may also potentially be associated with undesirable effects or safety concerns. For Fc-modified mAbs, the selection of in vitro and in vivo nonclinical pharmacology and safety models should consider species differences in FcR expression and function, potential differential effects of Fc modifications in humans and nonclinical species, the impact of targeting and cognate ligand expression patterns, and the potential impact of emerging antidrug antibodies against mAbs.
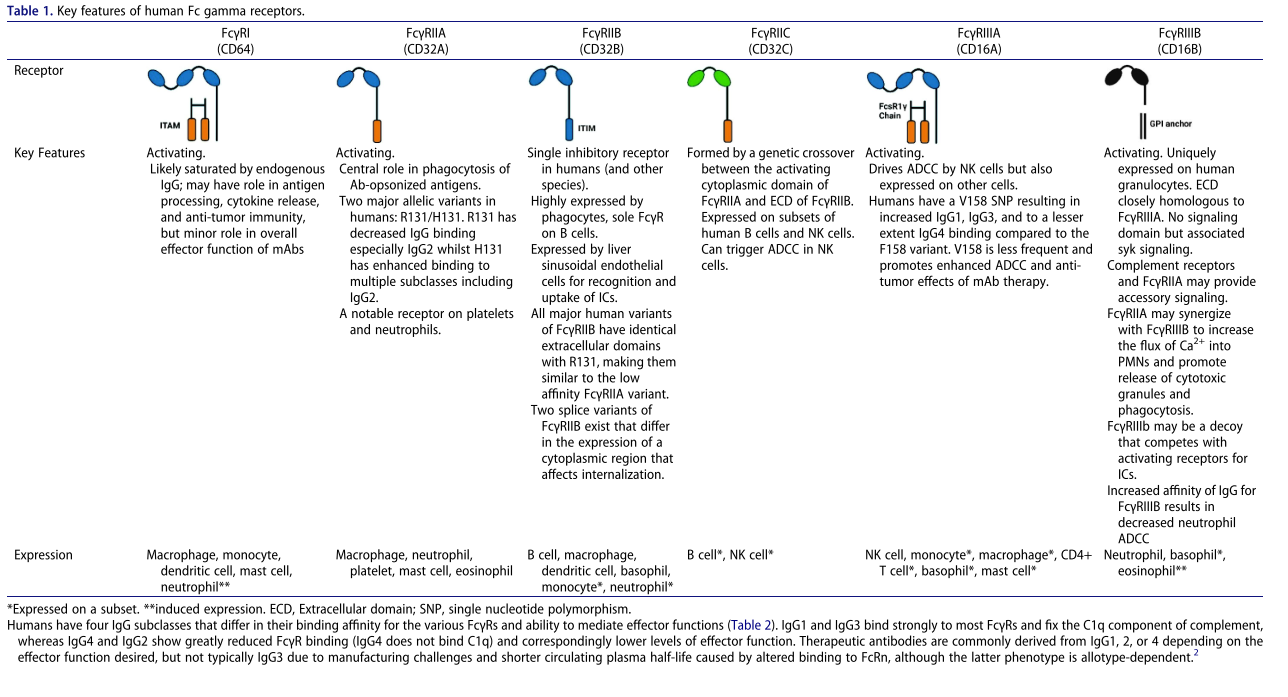
Expression and function of FcγR in humans
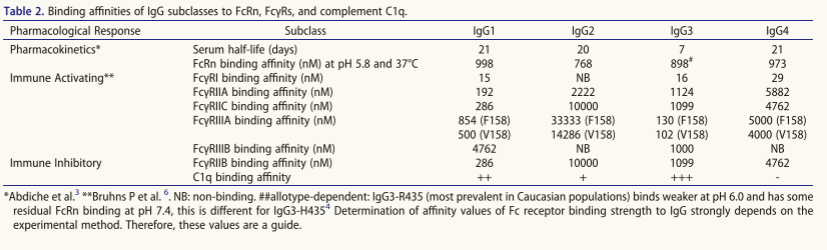
Human FcγRs are functionally classified as activating and inhibitory receptors, respectively, which transmit signals through intracellular ITAM or ITIM motifs, regulating cellular function following IgG antibody cross-linking. Among these, hFcγRI (CD64), hFcγRIIA (CD32a), hFcγRIIC (CD32c), and hFcγRIIIA (CD16a) are activating receptors, while FcγRIIB (CD32b) is the only inhibitory receptor (Table 1). With the exception of the high-affinity FcγRI, which can bind monomeric IgG, most FcγRs have a lower affinity for IgG1Fc (Table 2). Their potent functional effects rely on immune complex-mediated FcγR cross-linking, which is crucial for downstream signaling. FcγRIIIB (CD16b) lacks a transmembrane/cytoplasmic domain and is therefore GPI-anchored to the membrane, rendering it incapable of direct signaling. However, its high expression on neutrophils, homology to FcγRIIIA, and absence in rodents and most NHPs warrant careful evaluation.
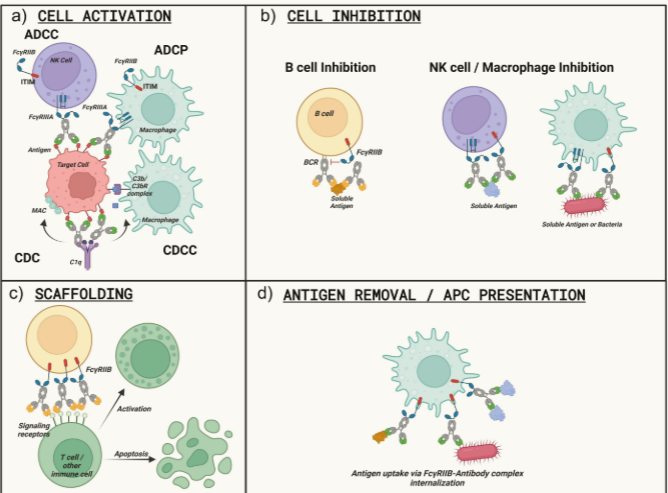
FcγRIIIA on NK cells mediates ADCC killing. FcγRIIA/FcγRIIIA on macrophages and FcγRIIIB on neutrophils promote ADCP killing. FcγRs (such as FcγRIIB) can also enhance target cell activation or inhibition through Fab arm crosslinking:
FcγRIIB-mediated "super-crosslinking" can induce target cell apoptosis/activation. FcγRIIA on platelets/neutrophils can trigger mediator secretion and coagulation. FcγRIIIB has a dual nature: although it normally clears circulating immune complexes without activating neutrophils, it can synergize with FcγRIIA to mediate IC activation under specific conditions.
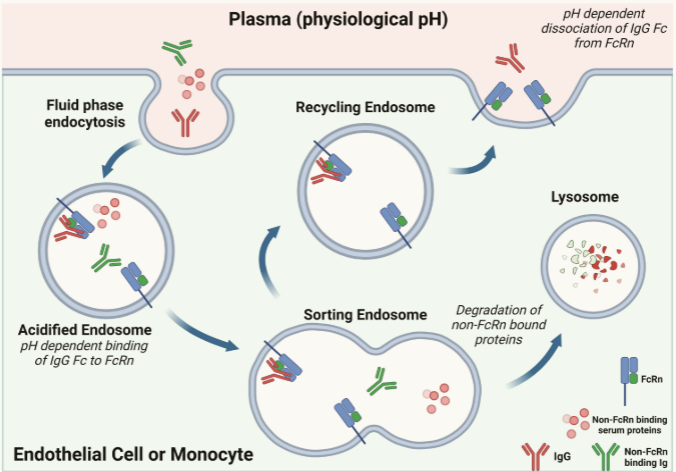
Expression and function of FcRn
FcRn is expressed in immune and non-immune tissues and is crucial for the recycling and transcellular transport of IgG . Circulating plasma proteins (e.g., antibodies, albumin, cytokines) can undergo fluid-phase endocytosis into endosomes. Following endosome entry, maturation, and acidification, IgG antibodies (red) can bind to FcRn in a pH-dependent manner. Once formed, the FcRn-IgG complex can recycle to the plasma membrane, where exposure to neutral pH in the circulation promotes dissociation. Antibodies and serum proteins that do not bind to FcRn or are unable to bind to FcRn at all are targeted for lysosomal degradation, where catabolic enzymes break down the protein into peptides and amino acids.
Adverse effects of Fc effector function in humans
Excessive FcγR/complement activation may pose safety risks for Fc-containing therapeutics. FcγR-mediated ADCC/ADCP and C1q/CDC can lead to non-target cell killing and cytokine release (e.g., tumor lysis syndrome), as seen with OKT3/rituximab. Cellular dysfunction: Aberrant FcγR cross-linking can induce activation-related apoptosis, ligation, proliferation inhibition, or internalization (depending on target biology). Complex inflammation: Cross-linking of circulating multivalent molecules forms soluble immune complexes, driving inflammatory responses. Thrombotic risk: Platelet FcγRIIA binding can promote activation and thrombosis (as seen with anti-CD40L mAbs). In the complement cascade, C3a/C5a anaphylatoxins activate mast cells/basophils/endothelial cells (C3aR/C5aR-mediated).

Enhancement or elimination of antibody Fc function through protein and glycoengineering
Many effector functions of IgG Fc contribute to the therapeutic efficacy of therapeutic mAbs and related Fc-containing scaffolds. Depending on the therapeutic goal, antibody engineering can enhance or inhibit effector function and half-life. When targeting inflammatory diseases, Fc-mediated immune activation (such as complement activation and cytokine release) should be avoided unless cell depletion is required (such as activation of NK/T cells in anticancer therapy). Non-activated IgG is often modified to inert IgG2/IgG4 subtypes or Fc silencing (such as IgG1/4 engineering). The core areas of Fc engineering include: (1) effector function enhancement/elimination ; (2) half-life regulation (mostly extension); (3) direct blockade of FcγR/FcRn.
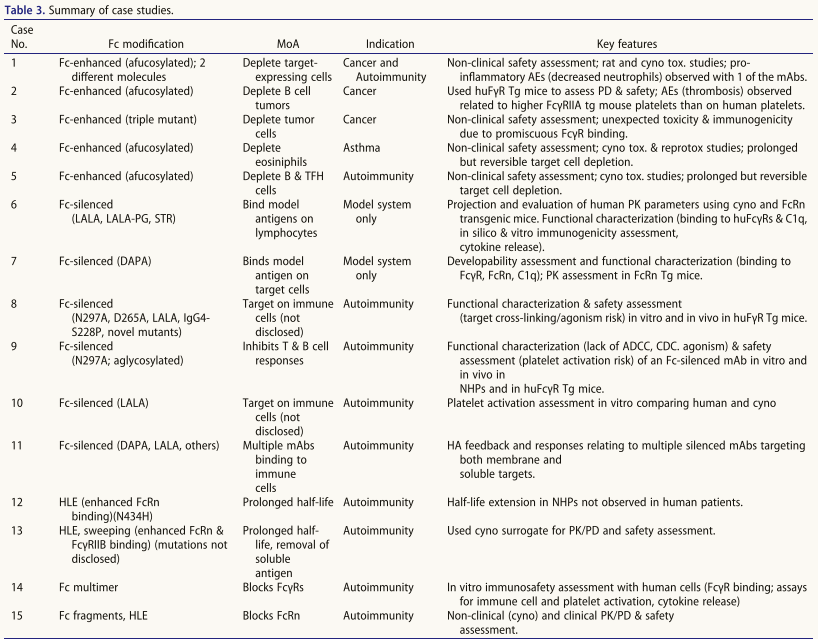
Amino acid substitutions that enhance Fc effector function
Amino acid substitutions can also be made within the IgG Fc to alter activation . Research methods include: 1. Alanine scanning (US2006173170A1) to locate binding domains: a) FcγR/C1q site: hIgG1 lower hinge region and proximal CH2 domain; b) FcRn site: the "elbow" region. 2. Computational modeling and high-throughput screening to identify novel FcγR binding-enhancing variants (Table 5); 3. Specific mutation strategies: a) E345K/E430G mutations promote hexamerization after target binding, enhancing complement activation (CDC/ADCC); b) Q311R/M428E/N434W triple mutations synergistically improve CDC activity and half-life, and can be combined with defucosylation to enhance ADCC.
Strategies to enhance FcγRIIB binding
The strategy of enhancing FcγRIIB binding was initially used to inhibit autoreactive B cells and later expanded to targeted agonism (particularly proximal B cell-driven). Typical examples include: 1. an anti-DR5 agonistic mAb , the first tumor-killing mAb requiring FcγRIIB crosslinking; 2. a TNF superfamily-targeted mAb targeting CD40/4-1BB/OX40 (preclinical validation). Table 6 lists relevant Fc variants, with "SELF" and "V11" mutations being commonly used. Several Fc variant mAbs with enhanced FcγRIIB binding have entered clinical trials (Table 4).

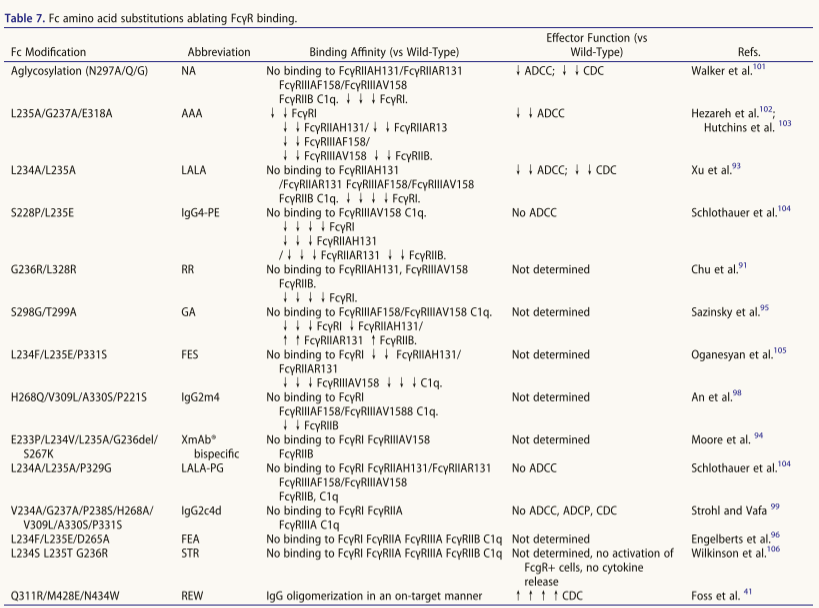
Glycoengineering and amino acid substitution for Fc silencing
The N297A/Q/G single-point mutation achieves deep Fc silencing by eliminating glycans, significantly reducing low-affinity FcγR/C1q binding (loss of ADCC/CDC activity) while retaining FcγRI binding and decreasing thermal stability. Several aglycosylated IgG1s have entered clinical trials.
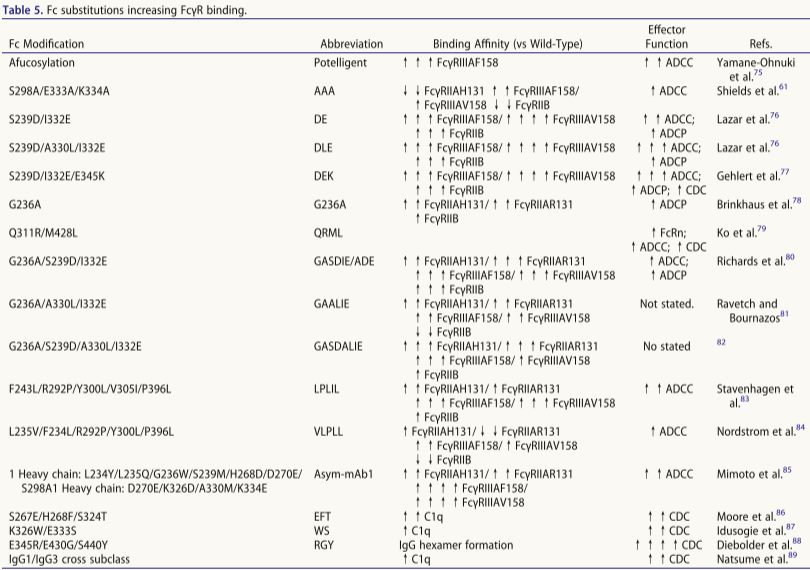
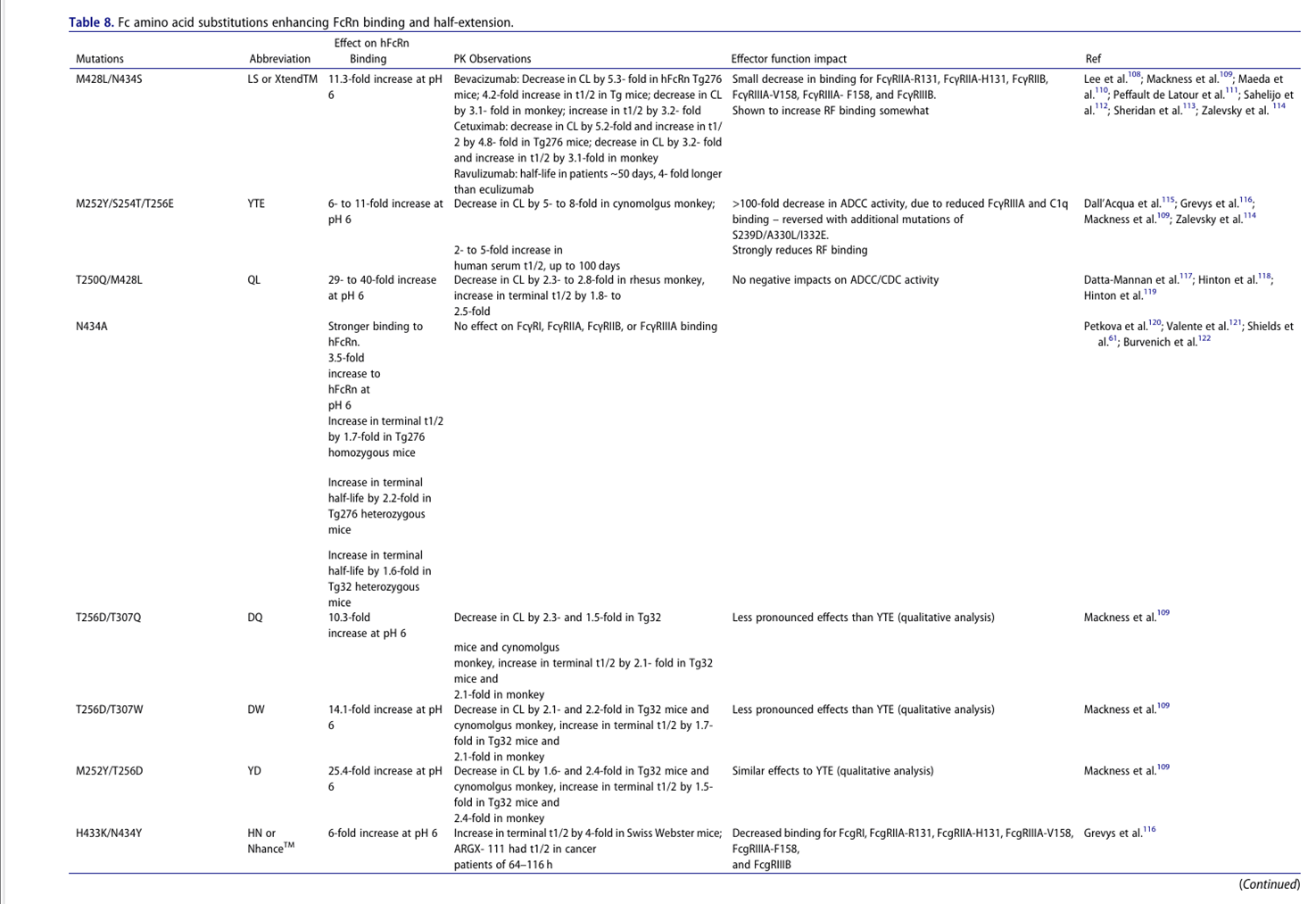
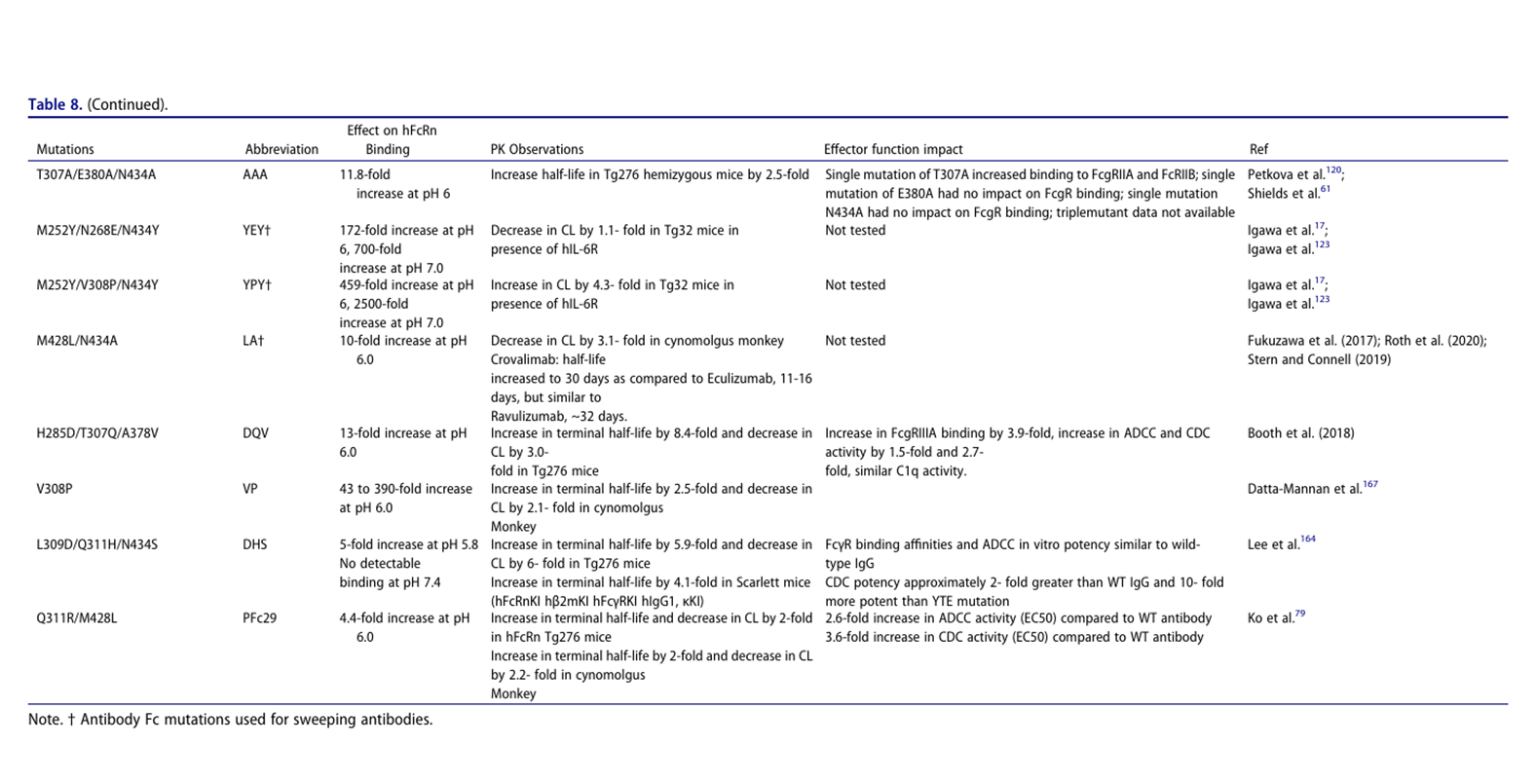
Extended half-life
Half-life extension (HLE) can increase mAb exposure, prolong pharmacological activity, and reduce dosing frequency and cost, but risks should be considered: 1. Limitations: a) Agonist mAbs may trigger adverse events due to prolonged target binding; b) They are ineffective against targets with high target-mediated elimination or rapid turnover (case study 12); c) They may affect FcγR and rheumatoid factor (RF) binding. 2. Engineering principle: Enhanced FcRn binding at pH 6 (not pH 7.4). 3. Sweeper antibody design: Eliminate soluble targets through dual mechanisms: a) Conditional antigen binding: Binding to plasma targets at neutral pH (avoiding endosomal binding); b) Fc engineering: Enhanced endosomal uptake of mAb-target complexes (FcγRIIB/FcRn/PI regulation), achieving a "net influx" of antigens for endosomal degradation. This is particularly suitable for targets with high concentrations (such as complement) or high turnover rates (such as cytokines).
FcγR and FcRn blockade
In autoantibody diseases, IVIG works by blocking FcγR/FcRn and complement regulation, but supply constraints, high costs, and inconvenient administration are challenges. This has driven the development of recombinant high-affinity Fcs: 1. Fc mutants (representative modifications). 2. Fc multimer engineering: a) Stradomers™: IgG1 hinge-Fc + IgG2 hinge region, broadly bind to FcγRs (including NHP homologs), and are effective in CIA/ITP models (GL-2045 Phase I terminated/NCT03275740); b) Fc hexamer: IgG1 hinge-Fc + IgM tail ± L309C, high-affinity blockade of FcγR/C1q: IgG1 type: triggers cytokine/platelet/complement activation in human whole blood; IgG4 type: has no activation effect; c) Fc trimer: binds to FcγRs but does not activate them, inhibiting IC function (effective in ITP models, Phase I terminated). 3. FcRn blockade therapy: non-selectively enhances IgG degradation.
Summarize
Fc engineering can and has already brought tangible clinical benefits and value to patients, but it also raises a complex and intertwined set of considerations that can pose risks if not properly addressed. A deepening understanding of these complexities, coupled with the continued accumulation of clinical experience and the iterative development of therapeutic application concepts, has driven the widespread application of key Fc modifications such as "LALA," "LS," and "YTE," as well as their derivatives.
