Fibroblast growth factor receptor 2 (FGFR2) is an important transmembrane tyrosine kinase receptor that plays a key role in regulating cell proliferation, differentiation, migration and survival, especially in embryonic development and tissue homeostasis.
Expression distribution of FGFR2
FGFR2 is predominantly expressed in epithelial cells (such as skin, mammary gland, and gastrointestinal tract) and osteoblasts. Its distribution is regulated by splice variants (type IIIb is mainly in the epithelium, and type IIIc is mainly in the mesenchyme). It is a key receptor that mediates epithelial-mesenchymal interactions and bone development/homeostasis.

(Data source: Uniprot)
Function of FGFR2
1. As a fibroblast factor receptor, it regulates cell proliferation, differentiation, migration and apoptosis through tyrosine kinase activity and is essential for embryonic development (limb, lung, bone, skin formation) and osteoblast differentiation/apoptosis.
2. Core role of the signaling pathway: Phosphorylation of downstream targets (PLCG1, FRS2, PAK4), activating multiple cascade reactions: a) PLCG1 → production of DAG/IP3; b) FRS2 → recruitment of GRB2/GAB1/PIK3R1/SOS1 → activation of RAS-MAPK (ERK1/2) and AKT pathways.
3. Regulatory mechanism: a) Negative regulation through the ubiquitination-internalization-degradation pathway; b) Mutations leading to constitutive activation or degradation defects can cause signaling abnormalities; c) Overexpression can activate STAT1.
(Data source: Uniprot)
The structure of FGFR2
FGFR2 is a type I transmembrane protein encoded by the FGFR2 gene with a length of 821 AA and a molecular weight of approximately 92 kDa . It is a typical receptor tyrosine kinase (RTK).
Extracellular domain: consists of three Ig-like C2-type domains: a) C1 domain: a distal regulatory domain involved in receptor autoinhibition; b) C2 domain: a core ligand-binding domain that directly binds to FGF (fibroblast growth factor); c) C3 domain: determines ligand specificity (e.g., isoform IIIb or IIIc) and contains an acidic box that regulates binding affinity. The heparin-binding domain (HBD) is located between C1 and C2 and binds to heparan sulfate proteoglycans (HSPGs), stabilizing the FGF-FGFR2 complex.
Intracellular domain: Juxtamembrane domain: Contains regulatory sites (such as Tyr375) that inhibit kinase activity. Split tyrosine kinase domain (Split TK domain): a) TK1 domain (N-terminal kinase lobe): Responsible for ATP binding; b) TK2 domain (C-terminal kinase lobe): Responsible for substrate phosphorylation, containing the activation loop (Tyr656/657). C-terminal tail: Contains phosphorylatable tyrosines (such as Tyr769) that recruit downstream signaling molecules (such as PLCγand FRS2).
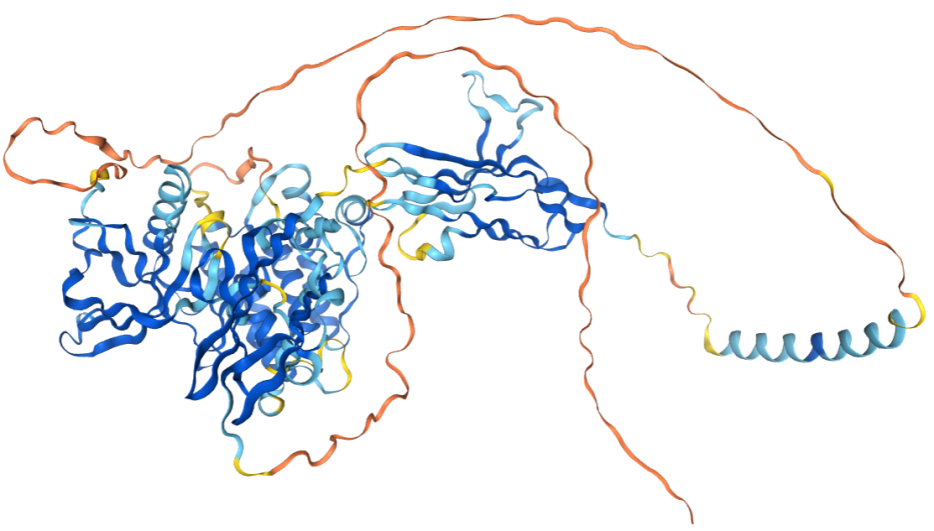
(Data source : AlphaFold)
FGFR2 signaling pathway
FRS2α is the core hub connecting FGFR2 with the RAS-MAPK and PI3K pathways. It mainly includes the following branches:
PLCγ (phospholipase Cγ) pathway: Phosphorylated FGFR2 recruits and phosphorylates PLCγ . Activated PLCγ hydrolyzes the membrane phospholipid PIP2 , producing the second messengers IP3 and DAG . IP3 releases Ca²⁺ from intracellular calcium stores , while DAG activates protein kinase C. Downstream effects: Calcium signaling and PKC activation are involved in cell migration and differentiation.
RAS-MAPK (RAS-mitogen-activated protein kinase) pathway: The adaptor protein FRS2α is a key hub of FGFR signaling and is phosphorylated by FGFR2 . Phosphorylated FRS2α recruits the Grb2-SOS complex. SOS , a guanine nucleotide exchange factor ( GEF ), activates the small G protein RAS . Activated RAS, in turn, activates RAF , MEK , and ERK/MAPK . Downstream effects: ERK phosphorylates multiple transcription factors (such as Elk-1, c-Fos, and c-Myc ), regulating gene expression and primarily driving cell proliferation, survival, and differentiation.
The PI3K-AKT (phosphatidylinositol 3-kinase-AKT) pathway: Phosphorylated FGFR2 or FRS2α recruits Gab1 , which in turn recruits and activates PI3K . Activated PI3K catalyzes PIP2 to generate the second messenger PIP3 . PIP3 recruits the PH domain-containing protein kinase PDK1 and AKT ( PKB ) to the plasma membrane. PDK1 phosphorylates and partially activates AKT , while mTORC2 is generally responsible for full AKT activation. Downstream effects: AKT phosphorylates multiple substrates, promoting cell survival, inhibiting apoptosis, regulating metabolism, and promoting proliferation.
STAT (Signal Transducer and Activator of Transcription) Pathway: Under certain conditions, phosphorylated FGFR2 can directly or indirectly recruit and phosphorylate STAT proteins (particularly STAT1, STAT3, and STAT5). Phosphorylated STATs form dimers and translocate to the cell nucleus. Downstream Effects: Regulate the transcription of specific target genes, participating in cell differentiation, survival, and immune responses.
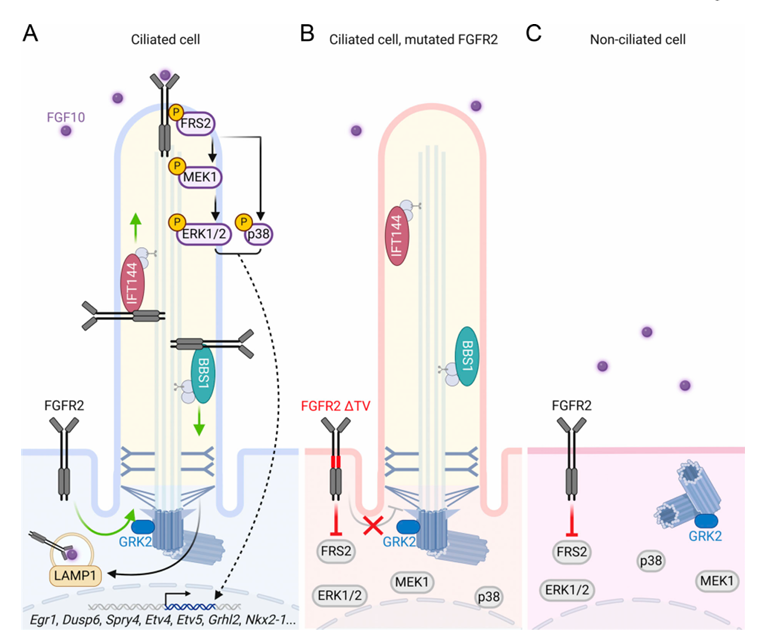
(Data source: Alexandru Nita1, et al. The Journal of cell biology vol. 2025)
FGFR2 and disease
Cholangiocarcinoma (especially intrahepatic cholangiocarcinoma, iCCA) : FGFR2 fusion: Approximately 10%-20% of iCCA patients have FGFR2 gene fusion (such as FGFR2 & BICC1 , FGFR2 & AHCYL1 , etc.), which leads to constitutive activation of the receptor and promotes tumor proliferation and metastasis.
(Data source: Arndt Vogel, et al. Annual Review of Medicine. 2023)
Gastric cancer: Amplification and overexpression Approximately 4% of gastric cancers have FGFR2 gene amplification, leading to protein overexpression, which is associated with diffuse gastric cancer, lymph node metastasis, and high N stage, indicating a poor prognosis . 27 Promotes lymphatic metastasis: The FGFR2/bFGF signaling pathway promotes lymphatic metastasis by increasing lymphatic microvessel density (LMVD).
Esophageal cancer: high expression and invasiveness: The positive rate of FGFR2 in esophageal squamous cell carcinoma reached 45.11% (significantly higher than 12.5% in normal tissues ), which was positively correlated with the depth of tumor infiltration and lymph node metastasis, and negatively correlated with the degree of differentiation.
Apert syndrome and Crouzon syndrome: caused by mutations in the FGFR2 extracellular domain (such as S252W and C278F ), which lead to abnormal activation of receptor dimerization and affect the differentiation of skull osteoblasts.
FGFR2-targeted therapy
Bemarituzumab is a monoclonal antibody targeting FGFR2, developed by Five Prime Therapeutics, Inc. Its primary mechanism of action is as an FGFR2 antagonist. By specifically binding to the FGFR2 extracellular domain, it blocks ligand-mediated signaling, inhibiting tumor growth and metastasis. It is indicated for the treatment of FGFR2b-positive gastroesophageal junction cancer. It received approval for Phase 3 clinical trials in China on March 7, 2022.
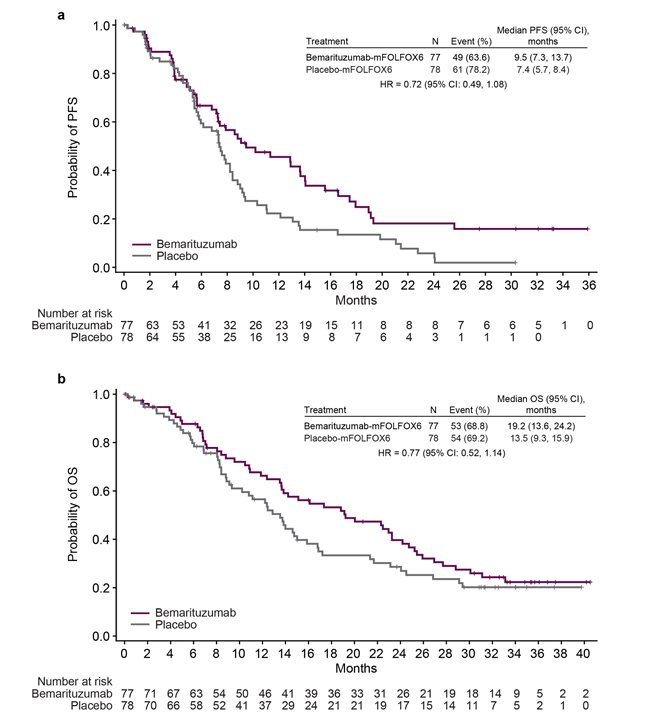
(Data source: Zev AW , et al. Gastric cancer. 2024)
TQB2210 is a monoclonal antibody targeting FGFR2b, developed by Chia Tai Tianqing Pharmaceutical Group Nanjing Shunxin Pharmaceutical Co. LTD. Its primary mechanism of action is as an FGFR2b regulator, intended for the treatment of solid tumors and advancedcancers. Phase 1 clinical trials were approved in China on May 16, 2025.
HWS116 is a monoclonal antibody targeting FGFR2b, developed by Wuhan Humanwell Innovation Pharmaceutical R&D Center Co., Ltd. Its primary mechanism of action is as an FGFR2b inhibitor for the treatment of advanced malignant solid tumors. The first application for Phase 1 clinical trials in China was filed on February 9, 2025.
HDM-2020 is an ADC targeting FGFR2b, developed by Huadong Medicine Co., Ltd. Its primary mechanism of action is as an FGFR2b inhibitor for the treatment of FGFR2-positive gastric cancer. The first Phase 1 clinical trial in China was approved on July 16, 2025.
